Actin protein (a>95% pure): human platelet - Discontinued
Important Notice:
This product is no longer available. The 99% pure non-muscle Actin protein purified from human platelets is available for purchase (See Cat. # APHL99). The amounts and price listed above are for Cat. # APHL99.
Product Uses Include
- Identification and characterization of non-muscle actin binding proteins
- In vitro actin polymerization studies
- Antibody standard for Western blot analysis
Material
Non-muscle actin has been purified from human platelets. Each unit of platelets used in the preparation of non-muscle actin has been found to be non-reactive by an FDA approved test for HBsAg, HBcAb, HIV-1/2 ab, HIV-1 RNA, HTLV I/II ab, HCV ab, HCV RNA, and syphilis. Each unit of platelets has been ALT tested with results less than an established cutoff. The isotype composition of non-muscle actin is 85% β-actin and 15% γ-actin. Non-muscle actin has an approximate molecular weight of 43 kDa. The product is provided as a lyophilized white powder. The lyophilized protein is stable for 6 months when stored desiccated to <10% humidity at 4°C. The protein should be reconstituted to 10 mg/ml with distilled water. It will then be in the following buffer: 5 mM Tris-HCl pH 8.0, 0.2 mM CaCl2, 0.2 mM ATP, 5% sucrose, and 1% dextran.
Purity
Protein purity is determined by scanning densitometry of Coomassie Blue stained protein on a 12% polyacrylamide gel. APHL99 consists of >99% pure non-muscle actin while APHL95 is >95% pure (see Figure 1).
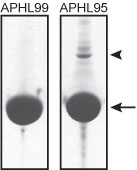
Figure 1: Purities of human platelet non-muscle actin protein. 100 µg of >99% pure (APHL99) and >95% pure (APHL95) non-muscle actin were run on SDS-PAGE gels and stained with coomassie blue. The arrow indicates actin protein (~43 kDa), the arrowhead a gelsolin contaminant (~90 kDa). The minor impurities in the purified actins are predominantly actin binding proteins such as gelsolin and α-actinin. Protein quantitation was determined with the Precision Red Protein Assay Reagent (Cat. # ADV02)
Biological Activity
The biological activity of muscle actinis determined by its ability to efficiently polymerize into filaments (F-actin) in vitro and separate from unpolymerized components in a spin down assay. Stringent quality control ensures that APHL99 produces >85% F-actin and APHL95 produces >75% F-actin in this assay.
For product Datasheets and MSDSs please click on the PDF links below. For additional information, click on the FAQs tab above or contact our Technical Support department at tservice@cytoskeleton.com
 For a guide to performing actin polymerizations with this actin product please click here.
For a guide to performing actin polymerizations with this actin product please click here.
Gohla, A., Birkenfeld, J. and Bokoch, G. M. (2005). Chronophin, a novel HAD-type serine protein phosphatase, regulates cofilin-dependent actin dynamics. Nat. Cell Biol. 7, 21-29.
Posern, G., Miralles, F., Guettler, S. and Treisman, R. (2004). Mutant actins that stabilise F-actin use distinct mechanisms to activate the SRF coactivator MAL. EMBO J. 23, 3973-3983.
Roger, B., Al-Bassam, J., Dehmelt, L., Milligan, R. A. and Halpain, S. (2004). MAP2c, but not tau, binds and bundles F-actin via its microtubule binding domain. Curr. Biol. 14, 363-371.
Chew, C. S., Chen, X., Parente, J. A., Jr., Tarrer, S., Okamoto, C. and Qin, H. Y. (2002). Lasp-1 binds to non-muscle F-actin in vitro and is localized within multiple sites of dynamic actin assembly in vivo. J. Cell Sci. 115, 4787-4799
Kuriyama, R., Gustus, C., Terada, Y., Uetake, Y. and Matuliene, J. (2002). CHO1, a mammalian kinesin-like protein, interacts with F-actin and is involved in the terminal phase of cytokinesis. J. Cell Biol. 156, 783-790.
Kessels, M. M., Engqvist-Goldstein, A. E. and Drubin, D. G. (2000). Association of mouse actin-binding protein 1 (mAbp1/SH3P7), an Src kinase target, with dynamic regions of the cortical actin cytoskeleton in response to Rac1 activation. Mol. Biol. Cell 11, 393-412.
Vartiainen, M., Ojala, P. J., Auvinen, P., Peranen, J. and Lappalainen, P. (2000). Mouse A6/twinfilin is an actin monomer-binding protein that localizes to the regions of rapid actin dynamics. Mol. Cell. Biol. 20, 1772-1783.
Zhou, D., Mooseker, M. S. and Galan, J. E. (1999). Role of the S. typhimurium actin-binding protein SipA in bacterial internalization. Science 283, 2092-2095.
Question 1: Do you have pyrene-labeled non-muscle actin for use in a polymerization assay?
Answer 1: Pyrene-labeled non-muscle actin has been shown to be unstable under normal storage conditions. To examine the polymerization of unlabeled non-muscle actin, please click here for a polymerization protocol that uses an excess of unlabeled non-muscle actin (Cat# APHL95) + a small amount of pyrene-labeled skeletal muscle actin (Cat. # AP05). The pyrene muscle actin will not polymerize efficiently on its own at the concentration used in this assay, so the reaction is dependent on unlabeled actin polymerization for F-actin formation. In this way, the pyrene-labeled muscle actin is taken up and polymerized to serve as a reporter for polymerization of the unlabeled non-muscle actin that is present at a much greater concentration.
Question 2: Are the actin products shipped as pure G-actin or a mixture of G- and F-actin?
Answer 2: Most of our actin proteins are sold in the monomer form (G-actin) because it is suitable for most applications in that form. After resuspension in A-buffer, we recommend incubating the actin on ice for 60 min before beginning the experiment to depolymerize any actin oligomers that might have formed during storage. Typically actin is first diluted to 0.2 or 0.4 mg/ml concentration and then this can be incubated on ice for 60 min to depolymerize actin oligomers. If you need nucleation depleted actin for longer filaments, we recommend the ice incubation followed by a high-speed centrifµgation (100,000 x g) for 60 min to pellet actin oligomers. Remove the top 80% of the supernatant by pipetting and use this as your G-actin stock. We also provide pre-formed actin filaments (Cat. # AKF99) that are also shipped lyophilized and upon resuspension, the filaments are ready for use and average 5-10 microns in length.
If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com



