Product Uses
• Study actin-bundling activity.
• Identification of compounds that inhibit actin and fascin 1 actin-bundling activity.
• Biochemical characterization of fascin 1 protein interactions
• Western blot standard
Material
The wild-type human fascin 1 protein has been produced in a bacterial expression system. The recombinant protein contains no tag. The molecular weight of fascin is approximately 54 kDa and it is supplied as a white lyophilized powder.
Storage and Reconstitution
Before reconstitution, briefly centrifuge to collect the product at the bottom of the tube. The protein should be reconstituted to 5 mg/ml with the addition of 20 µl of Milli-Q water (100 µg size). When reconstituted, the protein will be in the following buffer: 20 mM Tris pH 8.0, 150 mM NaCl, 2 mM CaCl2, 5% (w/v) sucrose, and 1% (w/v) dextran. In order to maintain high biological activity of the protein, it is strongly recommended that the protein solution be supplemented with DTT to 1 mM final concentration, aliquoted into "experiment-sized" amounts, snap frozen in liquid nitrogen, and stored at -70°C. The protein is stable for six months if stored at -70°C. The protein should not be exposed to repeated freeze-thaw cycles. The lyophilized protein is stable at 4°C desiccated (<10% humidity) for one year.
Purity
Protein purity is determined by scanning densitometry of Coomassie Blue-stained protein on a 4-20% polyacrylamide gradient gel. Fascin 1 protein was determined to be >95% pure. (see Figure 1 Below).
Figure 1. Fascin 1 Protein Purity Determination. A 10 µg sample of recombinant fascin 1 protein (molecular weight approx. 54 kDa) was separated by electrophoresis in a 4-20% SDS-PAGE system and stained with Coomassie Blue. Protein quantitation was determined using the Precision Red Protein Assay Reagent (Cat. # ADV02). Mark12 molecular weight markers are from Life Technologies Inc.
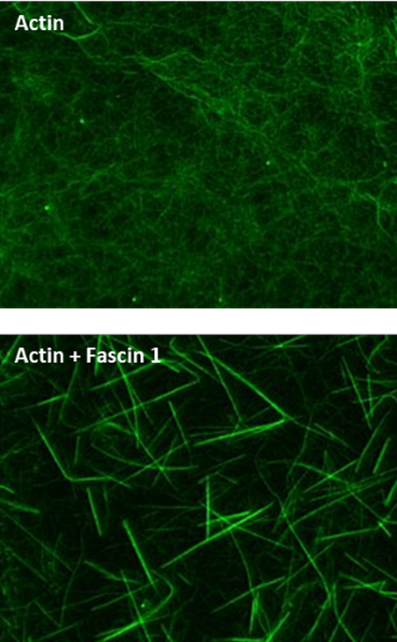
Fluorescence Microscopy Images of Actin-Bundling Assay with Fascin 1.
Figure Legend: Actin filaments alone and with fascin 1 stained with Acti-stainTM 488 Phalloidin as described in the method. Actin filaments were observed under a fluorescent microscope with a 480Ex/535Em filter set, a digital CCD camera, and 63x objective.
If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com
If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com