Actin Binding Protein Spin-Down Assay Biochem Kit: rabbit skeletal muscle actin
Product Uses Include
- To determine whether a protein binds to filaments or monomers of actin.
- To determine whether a protein bundles F-actin
- To test various conditions (e.g. pH optima) or requirements (e.g. divalent cation requirement) for binding to actin.
Introduction
The actin binding protein spin-down assay kit provides G- or F-actin plus positive (α-actinin ) and negative (Bovine Serum Albumin, BSA) binding control proteins. Actin binding occurs when there is an affinity for any site of actin. F-actin binding can be measured by using a spin down method. In this way centrifugation is used to separate F-actin from G-actin by differential sedimentation. F-actin binding proteins will co-sediment with actin filaments and form a pellet at the bottom of the centrifugation tube. Proteins with F -actin bundling activity can be detected since they will cause the F-actin to sediment at lower centrifugal forces than normal (14,000 x g vs 150,000 x g). F-actin severing proteins, G-actin binding proteins or non-actin binding proteins will stay in the supernatant. Severing proteins will be expected if more G-actin remains in the supernatant than in the negative control sample, and this activity should be tested further by measuring F-actin length distributions before and after adding the test protein. G-actin binding proteins can be measured by adding the test protein to G-actin and inducing polymerization, if the test protein sequesters G-actin then during the spin-down assay more actin will be left in the supernatant compared with the control.
Actin can exist in two forms: Globular subunit (G-actin) and Filamentous polymer (F-actin) (See the About Actin page for more information). Both forms of actin interact with a plethora of proteins in the cell. To date there are over 50 distinct classes of Actin-Binding Proteins (ABPs), and the inventory is still far from complete. Actin Binding Proteins allow the actin cytoskeleton to respond rapidly to cellular and extracellular signals and are integral to cytoskeletal involvement in many cellular processes. These include cell shape and motility, muscle contraction, intracellular trafficking, cell pathogenesis and signal transduction.
This kit contains skeletal muscle actin (Cat. # AKL99). The same kit is also available with non-muscle actin (Cat. # APHL99), see Cat. # BK013. The non-muscle actin spin-down kit may be more appropriate to use to study actin binding proteins from non-muscle tissues.
Kit contents
The kit contains sufficient materials for 30-100 assays depending on assay volume. The following reagents are included:
- 8 x 250 µg Skeletal muscle actin (Cat. # AKL99).
- α-Actinin, positive control (Cat. # AT01).
- BSA, negative control
- General Actin Buffer (Cat. # BSA01).
- Actin Polymerization Buffer (Cat.# BSA02)
- F-actin Cushion Buffer.
- ATP, 100 mM (Cat. no. BSA04)
- EGTA, 0.5 M
- MgCl2, 100 mM
- Tris-HCl pH 6.5, 1.0 M
- Tris-HCl pH 7.5, 100 mM
- Manual with detailed protocols and extensive troubleshooting guide.
Equipment required
- Centrifugation set-up capable of 150 000 xg at 4°C and 24°C, 50 -200 µl volume capacity.
- SDS-PAGE system.
- Detection system for protein of interest (coomassie is good for purified proteins, Western blot or silver stain for less pure or low abundance test proteins).
- Gel scanner for densitometric determinations.
Example results
The actin binding protein spin-down assay kit was used to study the actin binding and bundling activities of α-actinin (cat. # AT01). α-Actinin was shown to bundle F-actin and make the F-actin sediment at 14,000 x g (Fig. 1).
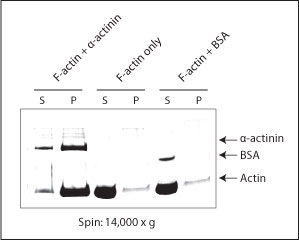
Figure 1. Actin bundling assay using kit BK001. F-actin was incubated alone or together with α-actinin or BSA. Bundled F-actin was pelleted by a 14,000 x g centrifugation and pellets (P) and supernatants (S) were run on an SDS-PAGE gel. Only in the presence of the F-actin bundling protein α-actinin is actin pelleted at this centrifugation speed.
For product Datasheets and MSDSs please click on the PDF links below. For additional information, click on the FAQs tab above or contact our Technical Support department at tservice@cytoskeleton.com
Question 1: What are optimal conditions for preparing my actin binding test protein?
Answer 1: Make the test protein at the highest possible concentration in an actin compatible buffer such as a HEPES or phosphate buffer. The high concentration (preferably >20 μM) is necessary to optimize the opportunity to detect interactions between actin and the test protein. In some cases the Kd (dissociation constant) of an actin binding protein/actin interaction may be so low as not to be detectable by protein assay of the pellet; however, the affect on the actin in the reaction mix may be enough to be detected by differences in the amount of actin in the pellet and supernatant versus the negative control. We also recommend centrifuging the test protein at 150,000 x g for 1 h at 4°C to remove denatured proteins and cellular debris. This is not necessary if other steps (e.g., affinity column purification) have been taken to purify the test protein. After the high-speed centrifugation, remove the supernatant and place on ice. This is the test protein stock.
The total ionic strength should be below 50 mM, this is due to the character of actin association whereby it’s mainly ionic and hence will be affected by medium and high salt buffers. A good starting buffer is 20 mM Hepes pH 7.4 plus 20 mM KCl, plus protease inhibitors, phosphatase inhibitors and any co-factors that are necessary. A low salt actin polymerization 10x buffer can be made with 20 mM MgCl2 and 10 mM ATP thus allowing the salt to be kept low in the final reaction.
Additional issues to consider are that a (i) test protein cofactor may be necessary, (ii) a factor in the assay could be actively inhibiting interactions, (iii) the test protein could have a preference for a different type of actin, or (iv) the test protein may be at a low concentration in tissue extracts.
To address some of these potential problems, reagents can be added to remove inhibitors, such as EGTA (Part #BSEG-01) for calcium. In the case of motor proteins, the ATP/Mg2+ combination will dissociate the motor from the F-actin hence a non-hydrolyzable analog such as AMPPNP is used at 1 mM. An actin affinity column can be used to concentrate and isolate more of the test protein. Finally, different pH conditions may improve binding between the actin and actin binding protein (e.g. alpha-actinin’s pH optima is 7.0).
Question 2: Can this kit be used to determine binding affinities between actin and actin binding proteins?
Answer 2: Binding affinity (Kd) can be estimated from the spin-down assay by titrating the test protein concentration between 0.2 and 20 μM and finding the concentration where half the original protein is in the pellet.
Question 3: Can cell lysates be used with this kit as the source of a test protein?
Answer 3: Yes, cell lysates can be used as the source of the test protein. However, Cytoskeleton does not recommend this as the purity and concentration of the protein will often be too low to interact with actin. Also, the lysates will contain additional accessory proteins and multiple phosphatases and proteases that can interfere or alter the interactions between actin and actin binding proteins. If lysates are to be used, we recommend the following:
Although this kit is designed for use with pure proteins or compounds, some researchers have added extracts with good results. Generally researchers use over-expressed proteins and a wild-type control extract similarly over-expressed. It is necessary to make a 10 mg/ml protein extract and then use 1/3rd volume of this to 2/3rd volume of actin at 1 mg/ml. In this way there is a high enough concentration of protein to make a difference. The extraction buffer should be 20 mM Hepes pH 7.5, 20 mM KCl, plus any co-factors for your protein, and a protease inhibitor cocktail such as our Cat# PIC02. Phosphatase inhibitors can also be added. Rinse the cells with an ice cold buffer and lyse cells with a 25g bent over syringe needle or other device. The pH can alter F-actin binding properties of actin binding proteins (ABPs) by 0 to 100%. It is a good idea to test pH 6.5, 7.5 and 8.5 for extract preparation. The control cell line is very critical because potential ABPs could appear in the pellet by other routes; for example, denaturing or aggregation with other cellular proteins. The total ionic strength should be kept to below 50 mM in order not to salt off any potential ABPs.
If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com







