AAN01 has been discontinued, and has been replaced by AAN02 (link)
RRID
AB_10708070
Product Uses
This antibody is recommended for detection of actin in human, mouse, rat, xenopus, and bovine extracts (Fig. 1 and 2). The following protocols have been tested with this antibody:
| Western Blot | Immunocytochemistry | ELISA | Immunoprecipitation | |
| Yes | Yes | Yes | No |
Material
Rabbit polyclonal antibody against actin protein. Actin is the major protein of the microfilament cytoskeletal system and is a key protein in various cell motility processes. The immunogen used for antibody production was a peptide consisting of the 11 C-terminal amino acids of actin. Human platelet extract (Cat. # EXT01) is included as a positive control for Western blot analysis. A characteristic actin band at 43 kDa is identified on Western blots (see Fig. 1). Antiactin antibody is supplied as a lyophilized white powder.
Example Results
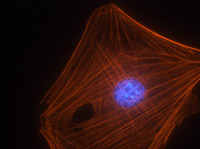
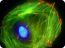
Figure 3. Immunofluorescence images of mouse Swiss 3T3 cells stained with anti-actin antibody. Swiss 3T3 cells were grown to 50% confluency and fixed with methanol. Immunofluorescence staining using 2 μg/ml (1:500 dilution) of anti-actin antibody is shown (red). The primary antibody was detected with a 1:500 dilution of goat anti-rabbit rhodamine conjugated antibody. DNA (blue) was stained with 100 nM DAPI in PBS. Photograph was taken with a 100X objective lens.

Figure 1. Western blot analysis of anti-actin antibody. Protein samples were separated by electrophoresis and transferred to PVDF membrane as described in the methods. Anti-actin antibody was diluted to 500 ng/ml (1:1000) for Western blot analysis. Actin was detected in 10 μg of platelet extract (43 kDa). Molecular weight markers are from Invitrogen.
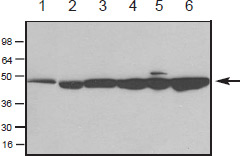
Figure 2. Western blot of purified actin and cell extracts probed with anti-actin antibody. Chemiluminescence detection of skeletal muscle actin (100 ng, lane 1), and in cell extracts of Xenopus A6 cells (lane 2), mouse Swiss 3T3 cells (50 μg, lane 3), rat NRK cells (50 μg, lane 4), human HeLa cells (50 μg, lane 5), and platelet cells (50 μg, lane 6). The actin band is indicated at 43 kDa (see arrow). The blot was probed with a 500 ng/ml (1:1000) dilution of antiactin antibody.
For product Datasheets and MSDSs please click on the PDF links below. For additional information, click on the FAQs tab above or contact our Technical Support department at tservice@cytoskeleton.com
Singh, K. et al. Non-muscle myosin II regulates aortic stiffness through effects on specific focal adhesion proteins and the non-muscle cortical cytoskeleton https://doi.org/10.1111/jcmm.16170
Lee, M., Kim, D. & Kwon, S. Hypergravity-induced changes in actin response of breast cancer cells to natural killer cells. Sci Rep 11, 7267 (2021). https://doi.org/10.1038/s41598-021-86799-7
Kurohmaru, M., Matsui, T., Igarashi, H., Hattori, S. & Hayashi, Y. Postnatal testicular development and actin appearance in the seminiferous epithelium of the Habu, Trimeresurus flavoviridis. Anat. Histol. Embryol. ahe.12628 (2020).
Delmotte, P., Han, Y. & Sieck, G. C. Cytoskeletal remodeling slows cross-bridge cycling and ATP hydrolysis rates in airway smooth muscle. Physiol. Rep. 8, e14561 (2020).
Dufek, B. et al. Pericyte abnormalities precede strial capillary basement membrane thickening in Alport mice. Hear. Res. 390, 107935 (2020).
Berkey, S. C. et al. EPAC1 and EPAC2 promote nociceptor hyperactivity associated with chronic pain after spinal cord injury. Neurobiol. Pain 7, 100040 (2020).
Xiong, Tian-Qing et al. The effects of calcineurin inhibitor FK506 on actin cytoskeleton, neuronal survival and glial reactions after pilocarpine-induced status epilepticus in mice. Epilepsy research vol. 140 (2018): 138-147. doi:10.1016/j.eplepsyres.2018.01.007
Gertz, Karen et al. The cytoskeleton in 'couch potato-ism': Insights from a murine model of impaired actin dynamics. Experimental neurology vol. 306 (2018): 34-44. doi:10.1016/j.expneurol.2018.04.004
Kandel, Judith et al. Mitochondrial DNA 3243A>G heteroplasmy is associated with changes in cytoskeletal protein expression and cell mechanics. Journal of the Royal Society, Interface vol. 14,131 (2017): 20170071. doi:10.1098/rsif.2017.0071
Hien, Tran Thi et al. Elevated Glucose Levels Promote Contractile and Cytoskeletal Gene Expression in Vascular Smooth Muscle via Rho/Protein Kinase C and Actin Polymerization. The Journal of biological chemistry vol. 291,7 (2016): 3552-68. doi:10.1074/jbc.M115.654384
Clin. Exp. Metastasis. 2015; 32, 301-311.
"MTBP inhibits migration and metastasis of hepatocellular carcinoma"
Author(s): Bi Q. et al.
(See PubMed article)
Cell Death Dis. 2015; 6, e1767.
"A cellular screen identifies ponatinib and pazopanib as inhibitors of necroptosis"
Author(s): Fauster A. et al.
(See PubMed article)
Mol. Psychiatry. 2015; doi: 10.1038/mp.2014.172.
"Identification and functional characterization of rare SHANK2 variants in schizophrenia"
Author(s): Peykov S. et al.
(See PubMed article)
Biochem. Biophys. Res. Commun. 2014; 454, 335–340.
"Intracellular modifiers of integrin alpha 6p production in aggressive prostate and breast cancer cell lines"
Author(s): Kacsinta A.D. et al.
(See PubMed article)
Anti-actin antibody (Cat. # AAN01) was used as a Western blot loading control to confirm that differences in Erk activation were not due to differential protein loading in lysates from human mesenchymal stem cells.
Mol. Biol. Cell 2005; 16, 881-890.
"Laminin-5 induces osteogenic gene expression in human mesenchymal stem cells through an ERK-dependent pathway."
Author(s): Klees, R. F., Salasznyk, R. M., Kingsley, K., Williams, W. A., Boskey, A. and Plopper, G. E.
(See PubMed article)
Anti-actin antibody (Cat. # AAN01) was used in the study of muscle force regulation to measure changes in G and F-actin ratios as detected by Western blotting in swine carotid artery tissues.
Am. J. Physiol. 2005; 288, C633-639.
"Heat shock protein 20-mediated force suppression in forskolin-relaxed swine carotid artery."
Author(s): Meeks, M. K., Ripley, M. L., Jin, Z. and Rembold, C. M.
(See PubMed article)
Anti-actin antibody (Cat.# AAN01) was used in immunocytochemistry and Western blotting experiments using mouse pancreatic acinar cells to study the functional link between inositol (1,4,5)-trisphosphate receptors and actin in controlling calcium signaling.
J. Cell Sci. 2005; 118, 971-980.
"Inositol 1,4,5-trisphosphate receptor links to filamentous actin are important for generating local Ca2+ signals in pancreatic acinar cells."
Author(s): Turvey, M. R., Fogarty, K. E. and Thorn, P.
(See PubMed article)
J. Biol. Chem. 2004; 279, 14039-14048.
"N terminus is essential for tropomyosin functions: N-terminal modification disrupts stress fiber organization and abolishes anti-oncogenic effects of tropomyosin-1."
Author(s): Bharadwaj, S., Hitchcock-DeGregori, S., Thorburn, A. and Prasad, G. L.
(See PubMed article)
J. Biol. Chem. 2004; 279, 39705-39709.
"Protective effect of phosphatidylinositol 4,5-bisphosphate against cortical filamentous actin loss and insulin resistance induced by sustained exposure of 3T3-L1 adipocytes to insulin."
Author(s): Chen, G., Raman, P., Bhonagiri, P., Strawbridge, A. B., Pattar, G. R. and Elmendorf, J. S.
(See PubMed article)
Am. J. Physiol. 2004; 286, G645-652.
"Escherichia coli LPS induces heat shock protein 25 in intestinal epithelial cells through MAP kinase activation."
Author(s): Kojima, K., Musch, M. W., Ropeleski, M. J., Boone, D. L., Ma, A. and Chang, E. B.
(See PubMed article)
J. Cell Biol. 2004; 167, 545-554.
"Cranial neural crest recycle surface integrins in a substratum-dependent manner to promote rapid motility."
Author(s): Strachan, L. R. and Condic, M. L.
(See PubMed article)
Am. J. Physiol. 2003; 285, L643-653.
"Aberrant lung structure, composition, and function in a murine model of Hermansky-Pudlak syndrome."
Author(s): Lyerla, T. A., Rusiniak, M. E., Borchers, M., Jahreis, G., Tan, J., Ohtake, P., Novak, E. K. and Swank, R. T.
(See PubMed article)
Mol. Biol. Cell 2003; 14, 2492-2507.
"Caenorhabditis elegans UNC-98, a C2H2 Zn finger protein, is a novel partner of UNC-97/PINCH in muscle adhesion complexes."
Author(s): Mercer, K. B., Flaherty, D. B., Miller, R. K., Qadota, H., Tinley, T. L., Moerman, D. G. and Benian, G. M.
(See PubMed article)
J. Cell Sci. 2003; 116, 2073-2085.
"Specific requirement for two ADF/cofilin isoforms in distinct actin-dependent processes in Caenorhabditis elegans."
Author(s): Ono, K., Parast, M., Alberico, C., Benian, G. M. and Ono, S.
(See PubMed article)
Question 1: What is the antigen that this antibody was raised against?
Answer 1: The antigen used for antibody production was a peptide consisting of the 11 C-terminal amino acids of actin (Ser-Gly-Pro-Ser-Ile-Val-His-Arg-Lys-Cys-Phe).
Question 2: What species has this antibody been tested against?
Answer 2: This antibody is recommended for detection of actin in human, mouse,
rat, Xenopus laevis, and bovine cell/tissue extracts.
If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com