SUMO-2/3 Antibody Mouse Monoclonal (Clone 11G2)
Anti-SUMO-2/3 antibody is a mouse monoclonal antibody. The antibody was raised against full-length recombinant SUMO-2 protein (Uniprot: P61956) combined with a proprietary mix of peptides that include CQIRFRFDGQPINE. The antibody has been shown to immunoprecipitate a wide range of SUMO-2/3 targeted proteins in a HeLa cell lysate (Fig. 1A). A linear epitope has not been identified and ASM24 appears to recognize a conformational epitope. Each Lot of antibody is quality controlled to provide a high batch to batch consistency. The Lot specific µg per tube can be found in the Lot specific COA documents at www.cytoskeleton.com. ASM24 is purified by Protein G affinity chromatography and is supplied as a lyophilized white powder.
Each Lot of antibody is quality controlled to provide a high batch to batch consistency. The Lot specific µg per tube can be found in the Lot specific COA documents.

Validated Applications
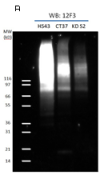
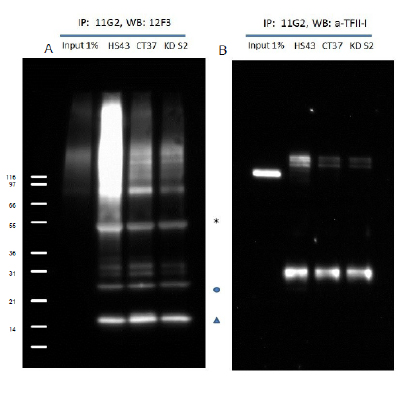
Figure 1: Immunoprecipitation using SUMO-2/3 Antibody Denatured cell lysates were prepared from HS43, CT37 and KD S210 (HS43: Heat Shock treated (43°C for 10min), CT37: untreated and KD S2: shRNA SUMO-2 knock down). 1mg of lysate was used for the immunoprecipitation of SUMO-2/3 conjugates. IP experiments were performed by the protocol presented in IP and WB Method. Western blots of immunopre-cipitated proteins were developed using anti-SUMO-2/3 (Cytoskeleon cat# ASM23) (A) or anti-TFII-I antibody (B). (A) Star (*) and circle (o) indicate heavy and light chains of antibodies. Un-conjugated free SUMO is denoted by a triangle. (B) Unconjugated TFII-I is visible near 120 kDa. Multiple bands indicate that TFII-I is SUMOylated by several SUMO-2/3 proteins. TFII-I has previously been reported to be a target for Sumoylation 10,11. To see the full Immunoprecipitation protocol, see the product datasheet. |
Validated Applications
HeLa cells were stained and visualized by widefield fluorescence microscopy as de-scribed in the IF method below. The cells were stained against α/β-tubulin (sheep anti-tubulin Ab, Cat# ATN02, green) and SUMO-2/3 (11G2, red). DNA was stained with DAPI. Mitotic cells in metaphase were imaged with a Zeiss Axio Observer.Z1 microscope (1.4 NA 63X objective). The localization of SUMO 2/3-conjugates at chromosomes can be observed during mitosis as has been previously reported12. To see the full Immunofluorescence protocol, see the product datasheet.
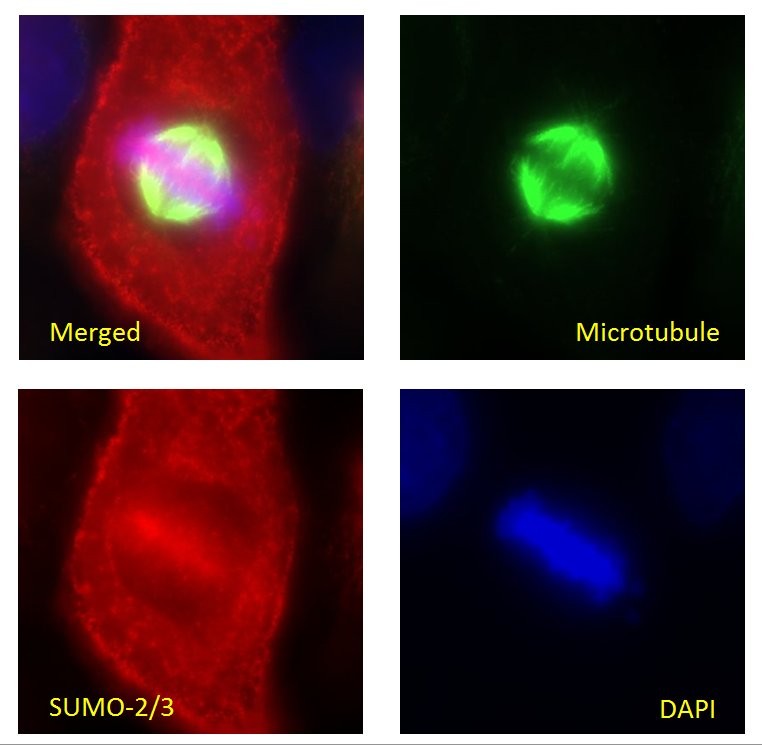
Validated Applications
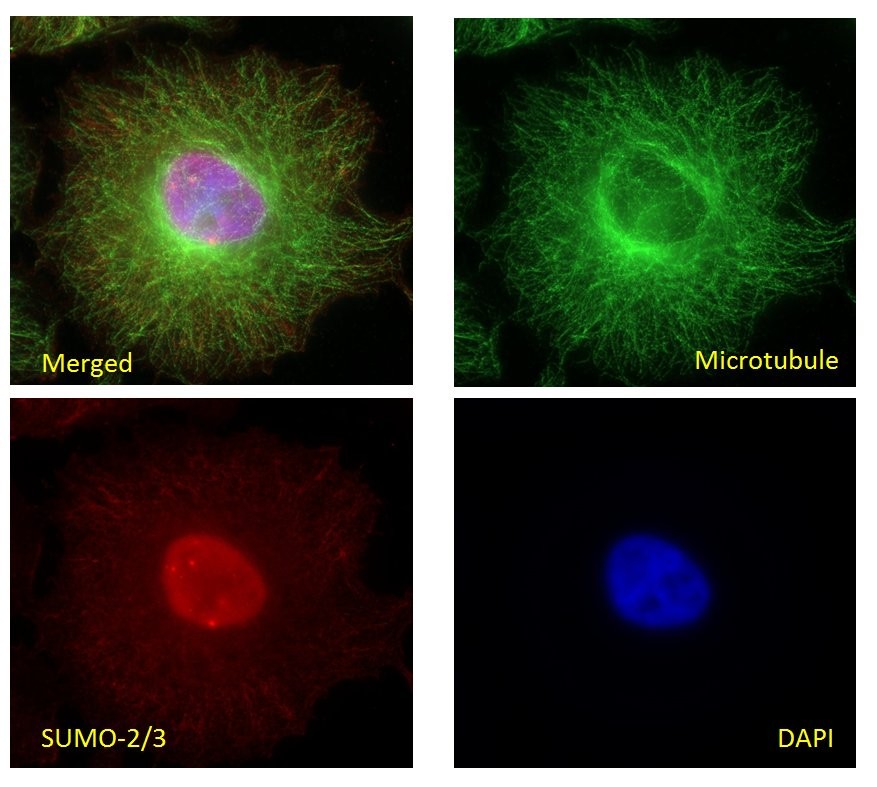
Figure 3: Immunofluorescence of HeLa cells in interphase with SUMO-2/3 Antibody
HeLa cells were stained and visualized by widefield fluorescence microscopy as de-scribed in the IF method below. The cells were stained against β-tubulin (sheep anti-tubulin Ab, Cat# ATN02, green) and SUMO-2/3 (11G2, red). DNA was stained with DAPI. Cells in interphase were imaged with a Zeiss Axio Observer.Z1 microscope (1.4 NA 63X objective). PML nuclear bodies (nuclear dots) were visible in SUMO-2/3 staining as has been previously reported5. To see the full Immunofluorescence protocol, see the product datasheet.
For more information contact: signalseeker@cytoskeleton.com
Associated Products:
Signal-Seeker™ SUMOylation 2/3 Detection Kit (Cat. # BK162)
Signal-Seeker™ SUMOylation 2/3 Affinity Beads (Cat.# ASM24-beads)
Signal-Seeker™: BlastR™ Rapid Lysate Prep Kit (Cat. # BLR01)
For product Datasheets and MSDSs please click on the PDF links below.
Sample Size Datasheet (Cat. ASM24-S): ![]()
Certificate of Analysis: Lot 011
Visit our Signal-Seeker™ Tech Tips and FAQs page for technical tips and frequently asked questions regarding this and other Signal-Seeker™ products click here
If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com