Actin: a critical player in cancer cell immunosurveillance
Introduction
Actin is a globular protein that can assemble into filamentous structures and is a foundational member of the cytoskeleton network in cells. Actin has well-established roles in muscle contraction, vesicle movement, cell motility, and many other biological processes. Emerging studies have highlighted its essential role in regulation of the immune system, whereby, mutations in actin and actin binding proteins (ABPs) lead to an array of inflammatory and/or immunodeficiency disorders(1). Actin related mutations that affect the immune system have been identified to affect hematopoietic cells leading to defective function of the immune cells(2). This is of particular interest as the growing literature suggests that the immune system plays a significant role in many diseases including cancer(3). Here, we highlight several new studies investigating actin’s function in cancer cells and its impact on immunosurveillance and cancer progression.
Actin Functions as a Tumor Neoantigen
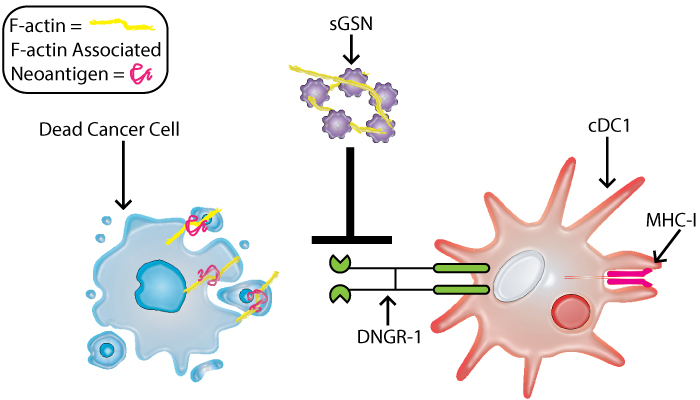
A specific subset of dendritic cells, known as Type 1 conventional dendritic cells (cDC1s), function by initiating T cell responses and enhancing T cell cytotoxicity; importantly, they are essential for effective anti-tumor response to adoptive T cell transfer or checkpoint immunotherapy, and their increased levels correlates with increased overall patient survival(4). cDC1s express high levels of the DNGR-1 receptor, which when activated promotes cross-uptake and activation of CD8+T cells. DNGR-1 has been shown to bind F-actin exposed on necrotic cell corpses and may be important during immunosurveillance(5). In a recent report, Giampazolias et al. showed that secreted gelsolin suppresses DNGR-1 binding to F-actin as a mechanism (Fig. 1) to suppress immunosurveillance(6). Using both cancer cell models and mice null for sGSN they showed that sGSN alters tumor growth, and augments response to immune checkpoint inhibitors. Finally, they observed that human cancers where mutations in proteins associated with the cytoskeleton were present, those that had lower levels of sGSN transcripts had better survival rates. These studies highlight the importance of the F-actin as a tumor antigen for cross-presentation in immunosurveillance.
Soluable Gelsolin inhibits DNGR-1 binding to f-actin and suppresses
immmunosurveillance.
Actin Alters Biophysical Properties of Cancer Cells
Natural killer (NK) cells are lymphocytes that also contribute to immunosurveillance and utilizes its cytotoxic activity to rid organisms of transformed or stressed cells(7). While much is known about how actin functions in NK cells, not much has been described about how actin in tumor cells regulate NK function. A study by Clement Thomas’ group determined that NK-cell attack produced a rapid and massive accumulation of F-actin near the immunologic synapse and had a protective effect for the cancer cell(8). The group found that resistant cancer cells produced a robust F-actin response while NK-susceptible cells did not. Interestingly, NK cell stimulation was maintained in response to this F-actin response; however, these cancer cells had much lower levels of internalized cytotoxic granzyme B. Importantly, suppressing upstream actin signaling, such as, depletion of CDC42 was sufficient to sensitize resistant cancer cells to NK cell mediated killing.
In contrast, Tello-Lafoz et al. recently published data showing that high expression of MRTF, an actin inducing transcription factor, sensitized melanoma and breast cancer cells to the immune system(9). The group showed that MRTF overexpression rigidified the F-actin cytoskeleton in cancer cells and made them more vulnerable to NK cells and T lymphocytes. This susceptibility translated to mouse cancer models; whereby, increased MRTF overexpression sensitized metastatic tumors to cytotoxic lymphocytes. They determined that the sensitization of cancer cells was not generalized to all apoptotic stimulants; rather, it was very specific to cytotoxic lymphocytes due to enhanced stimulation of these immune cells. Importantly, the investigators determined that the enhanced stimulation was promoted due to the biophysical properties of the cancer cells that were now expressing copious amounts of actin, which formed dense arrays of stress fibers that led to cell stiffening. They utilized DeAct to specifically depolymerize actin in cancer cells, which was sufficient to reverse the effects of MRTF enhanced susceptibility to cytotoxic lymphocytes. These results suggest that mechanical immunosurveillance may be a mechanism by which immune cells detect dysfunctional cancer cells even without specific cell surface biochemical features; however, the conflicting role of actin between these studies suggests that there is still much to be uncovered regarding it’s role in cancer cells as it relates to immune evasion.
Summary
With the growing success of immunotherapies for treating cancers, there is a significant interest in understanding the interplay between cancer cells and the immune system, especially as resistance mechanisms towards these therapies arise. These studies identified actin as an important player in immunosurveillance and its modified presence promoted cellular dysfunction that enhanced identification by immune cells. It was quite interesting that altering these actin dependent immune cell detection mechanisms had a profound effect on checkpoint inhibitor efficacy. As these studies progress it will be of interest to see if drugs targeting the actin cytoskeleton become viable combination therapies to overcome immunotherapy resistance. Cytoskeleton has an array of actin tools to aide investigators in this exciting area of research.
References
1.Papa R, Penco F, Volpi S, Gattorno M. Actin Remodeling Defects Leading to Autoinflammation and Immune Dysregulation. Front Immunol. 2020;11:604206.
2.Sprenkeler EGG, Webbers SDS, Kuijpers TW. When Actin is Not Actin' Like It Should: A New Category of Distinct Primary Immunodeficiency Disorders. J Innate Immun. 2021;13(1):3-25.
3.Waldman AD, Fritz JM, Lenardo MJ. A guide to cancer immunotherapy: from T cell basic science to clinical practice. Nat Rev Immunol. 2020;20(11):651-68.
4.Bottcher JP, Reis e Sousa C. The Role of Type 1 Conventional Dendritic Cells in Cancer Immunity. Trends Cancer. 2018;4(11):784-92.
5.Ahrens S, Zelenay S, Sancho D, Hanc P, Kjaer S, Feest C, et al. F-actin is an evolutionarily conserved damage-associated molecular pattern recognized by DNGR-1, a receptor for dead cells. Immunity. 2012;36(4):635-45.
6.Giampazolias E, Schulz O, Lim KHJ, Rogers NC, Chakravarty P, Srinivasan N, et al. Secreted gelsolin inhibits DNGR-1-dependent cross-presentation and cancer immunity. Cell. 2021;184(15):4016-31 e22.
7.Huntington ND, Cursons J, Rautela J. The cancer-natural killer cell immunity cycle. Nat Rev Cancer. 2020;20(8):437-54.
8.Al Absi A, Wurzer H, Guerin C, Hoffmann C, Moreau F, Mao X, et al. Actin Cytoskeleton Remodeling Drives Breast Cancer Cell Escape from Natural Killer-Mediated Cytotoxicity. Cancer Res. 2018;78(19):5631-43.
9.Tello-Lafoz M, Srpan K, Sanchez EE, Hu J, Remsik J, Romin Y, et al. Cytotoxic lymphocytes target characteristic biophysical vulnerabilities in cancer. Immunity. 2021;54(5):1037-54 e7.
Related Products
New Pan Actin Antibody
Anti-Pan Actin Mouse Monoclonal Antibody (Clone 7A8.2.1) (Cat. # AAN02)
Acti-Stain™ Products
Acti-stain™ 535 (Cat. # PHDR1)
Acti-stain™ 555 (Cat. # PHDH1)
Acti-stain™ 670 (Cat. # PHDN1)
Actin Live Cell Imaging Products
SiR-Actin Kit (Cat. # CY-SC001)
Cytoskeleton Kit (Includes SiR-Actin, SiR-Tubulin, and Verapamil) (Cat. # CY-SC006)
SiR700-Actin Kit (Cat. # CY-SC013)
SPY555-Actin (Cat. # CY-SC202)
Actin Products
Actin protein (>99% pure): bovine cardiac muscle (Cat. # AD99)
Actin protein (>99% pure): chicken gizzard muscle (Cat. # AS99)
Actin protein (pre-formed filaments): rabbit skeletal muscle (Cat. # AKF99)
Actin protein (>95% pure): rabbit skeletal muscle (Cat. # AKL95)
Actin protein (>99% pure): rabbit skeletal muscle (Cat. # AKL99)
Actin protein (>99% pure): human platelet (Cat. # APHL99)
Actin protein (rhodamine): human platelet (Cat. # APHR)
Actin protein (rhodamine): rabbit skeletal muscle (Cat. # AR05)
Actin Biochem Kits
Actin Binding Protein Spin-Down Assay Biochem Kit: rabbit skeletal muscle actin (Cat. # BK001)
Actin Binding Protein Spin-Down Assay Biochem Kit: human platelet actin (Cat. # BK013)
Actin Polymerization Biochem Kit (fluorescence format): rabbit skeletal muscle actin (Cat. # BK003)
G-Actin/F-actin In Vivo Assay Biochem Kit (Cat. # BK037)
MOX Actin Products