Citation Spotlight: Misshapen Kinase Alters Actin Dynamics in the Regulation of Ring Canal Size and Stability
- By Cytoskeleton Inc. - Actin News
- Aug 27, 2018

Kline et al. recently studied regulation of intercellular bridges in Drosophila egg chambers. Known as ring canals, the bridges transfer cytoplasmic materials between neighboring cells; in this case, from nurse cells to the oocyte over the course of oogenesis. Ring canals are essential for maintaining an organism’s fertility. Ring canal size and stability rely upon dynamic re-organization of F-actin. Here, the Ste20 family kinase misshapen (msn) has a novel role in regulating ring canal size and stability with changes in msn expression levels or localization altering the F-actin cytoskeleton. Phosphotyrosine (pTyr) signal in ring canals strongly overlaps with actin in control egg chambers. pTyr signal was used as a read-out to measure msn localization and activity in control vs msn-RNAi-treated egg chambers. In the latter, pTyr localized to nurse cell membranes and most ring canals. However, pTyr fluorescence in ring canals was variable, even within the same egg chamber, and did not overlap with actin signal. Hence, msn is not necessary for recruiting actin to the ring canal, but is essential for maintaining a ring canal’s actin-based structure. Cytoskeleton’s anti-phosphotyrosine antibody (Cat.# APY03; clone 11G2) was essential in the subcellular localization of pTyr signal in control and msn-depleted ring canals.
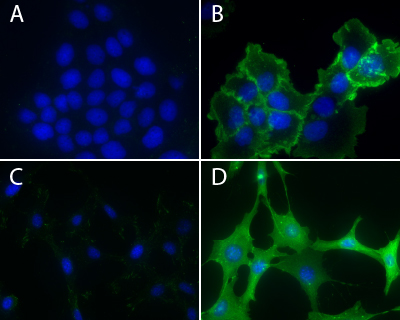
Immunofluorescence using Phosphotyrosine Antibody: Human epidermoid carcinoma A431 cells, untreated (3A) or treated (3B) with EGF (100 ng/ml for 3 minutes), and NIH3T3, untreated (3C) or treated (3D) with H2O2-activated sodium orthovanadate (100 µM for 10 minutes), were stained as described in the method. Phosphotyrosine and nuclei were visualized in green fluorescence and blue DAPI staining, respectively.
