Citation Spotlight: Rho-Family GTPase Activity: Key Components of Anti-Inflammatory and Neuroprotective Pathway
- By Cytoskeleton Inc. - Small G-Protein News
- Dec 21, 2016

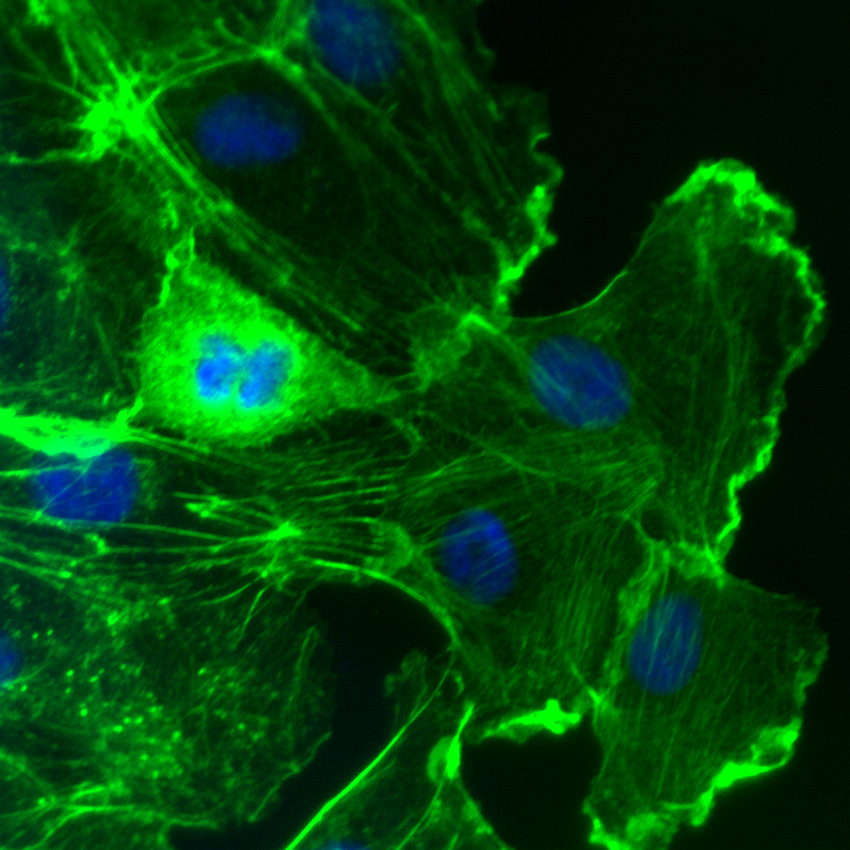
Recently, Rom et al. examined molecular pathways involved in leukocyte-mediated neuroinflammation given its causative role in neuronal dysfunction associated with brain injuries and diseases. Neuroinflammation involves a compromised blood-brain barrier (BBB) as leukocytes need to engage brain endothelial cells. To do so, leukocytes utilize integrin adhesion receptors for rolling, arrest, adhesion, and transendothelial migration (TEM), processes requiring Rho-family GTPase-mediated rearrangement of the actin cytoskeleton. Here, the activation of VLA-4 and LFA-1 leukocyte integrins following inhibition of PARP (poly(ADP-ribose) polymerase 1) activity in leukocytes was studied with the goal of preventing BBB breakdown. Using primary human brain microvascular endothelial cells to model the BBB, PARP inhibitors reduced leukocyte adhesion and TEM, concomitant with decreased activation of the two integrins and RhoA and Rac1 GTPases, as well as a reduced F-/G-actin ratio. Cytoskeleton’s RhoA and Rac1 G-LISA activation assays (Cat.# BK124 and BK128, respectively), Acti-stain 488 phalloidin (Cat.# PHDG1), and cell-permeable Rho inhibitor (Cat.# CT04) and Rho/Rac/Cdc42 activator (Cat.# CN04) were essential reagents, allowing for sensitive and reliable quantification of RhoA and Rac1 activation under control and experimental conditions while also measuring dynamic actin cytoskeletal changes. These results suggest novel therapies for protecting BBB integrity following brain disease and injury.
