Citation Spotlight: Stability of HTLV-2 antisense protein is controlled by PML nuclear bodies in a SUMO-dependent manner
- By Cytoskeleton Inc. - Signal-Seeker News
- Aug 19, 2019

Dubuisson et al. recently identified a molecular, regulatory mechanism responsible for the instability of the antisense protein of HTLV-2 (APH-2). While human T lymphotropic virus type 1 (HTLV-1) has been linked to several diseases, the HTLV-2 virus is asymptomatic; thus, comparative studies have aimed to identify critical, functional differences. Both viruses have anti-sense proteins but dissimilarities exist between the two; for example, the HTLV-1 antisense protein, HBZ, plays a significant role in virus induced disease progression while the APH-2 protein is highly unstable and has a half-life of 20-30 minutes. Dubisson et al. sought to decipher the molecular mechanism responsible for this instability, and discovered that a post-translational modification, SUMOylation, regulates its stability. A critical first step in this break-through was the discovery that APH-2 is endogenously modified by SUMO 2/3 and localized to PML nuclear bodies. Additional molecular studies were performed to determine that SUMO 2/3 and PML are essential regulators of APH-2 stability, but had little effect on HBZ expression. Cytoskeleton’s SUMO 2/3 Detection Kit (Cat. # BK162) was an essential reagent that was utilized to investigate the endogenous SUMO 2/3 state of APH-2, which provided a key finding for understanding APH-2 stability. This study adds to the growing body of knowledge whereby viruses utilize cellular SUMOylation machinery to their benefit, and is complementary to data showing therapeutic viral treatments, such as interferon activate SUMO mechanisms as part of its mechanism of action to combat viruses.
Products used in this citation:
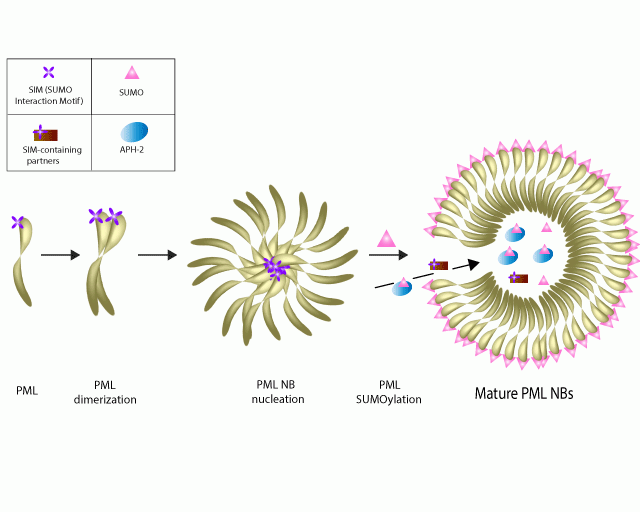
Schematic representation of SUMOylated APH-2 in a developed PML nuclear body.
