Product Uses
- Measurement of LOX inhibition by compounds.
- Positive control in immunodiagnostics.
- Positive control as LOX enzyme in comparisons to LOXL enzymes.
- Identification/characterization of substrates of LOX
Material
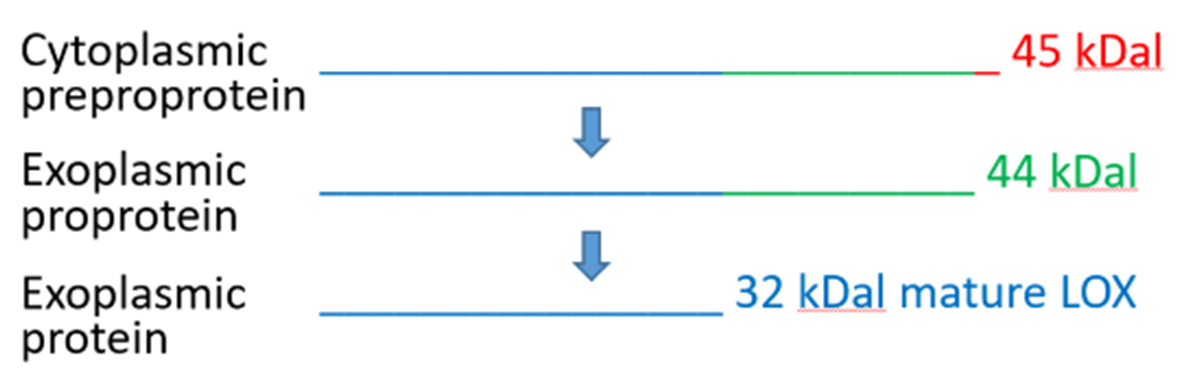
Lysyl oxidase (LOX, Acc.# NM_173932.4, similar to Human Acc.# NM_001178102, EC 1.4.3.13) is a copper containing 32 kDal protein found in structural tissues where it functions to promote crosslinking of structural proteins resulting in higher tensile strength of the biopolymer. Although concentrations are up to 500 µg/ml in some tissues (e.g. tendon), the yields are very low in the purified form, usually 1-2 mg per Kg of raw material (Kagan and cai, 1995). LOX is supplied as a white lyophilized powder.
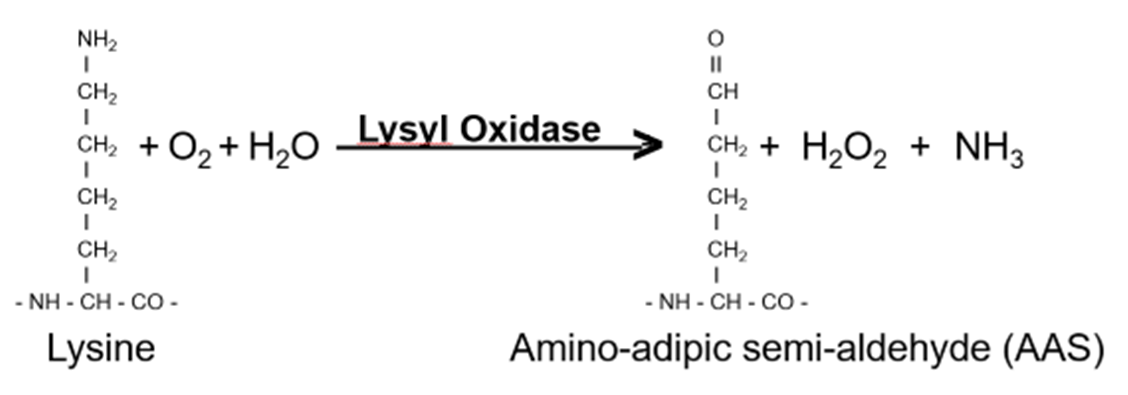
Figure 2: LOX will exchange the NH2 group with O, which creates a reactive entity for crosslinking to other tertiary amines. In the assays of LOX, H2O2 is used to oxidize reporter molecules with horse radish peroxidase as a catalyst.
Storage and Reconstitution
Briefly centrifuge to collect the product at the bottom of the tube. Reconstituting a 5 mg tube of CS-LXE01 with 100 ml of nanopure will generate a 50 µg/ml stock of LOX in the following buffer: 4M urea in PBS pH 7.4. The protein can be frozen in liquid nitrogen and stored at –70°C. It is not recommended to store at 4°C in liquid form. The lyophilized protein is stable at 4°C desiccated (<10% humidity) for 6 months.
Purity
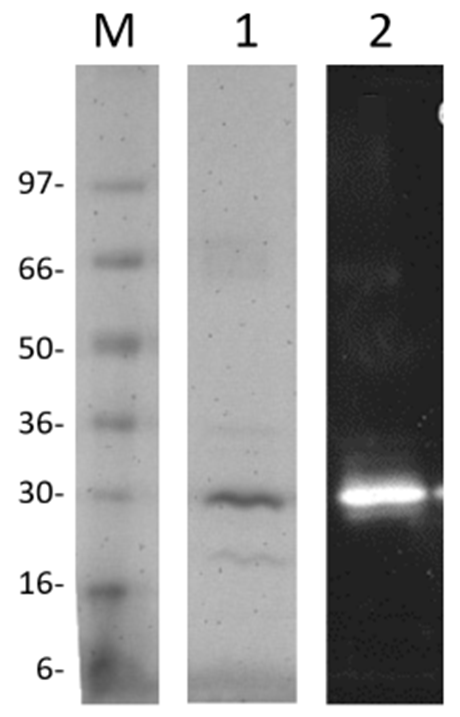
Protein purity is determined by scanning densitometry of Coomassie Blue stained protein on a 4-20% gradient polyacrylamide gel. LOX was determined to be >80% pure (see Figure 3 Below).
Figure 3: A 2 µg sample of LOX (lane 1) separated by electrophoresis using a 4-20% SDS-PAGE gel and stained with Coomassie Blue. In Lane 2, the immunoblotted protein was probed with anti-LOX (NBP100-2527) Protein quantitation was performed using the Precision Red™ Protein Assay Reagent (Cat.# ADV02). SeeBlue molecular weight markers are from Life Technologies Inc.
If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com
If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com