Custom Services
Cytoskeleton, Inc. has been a reliable source of compound screening services in the areas of pre-clinical drug development programs and early compound screening in primary HTS projects, as well as secondary screening and compound target validation. We also have extensive experience in gene design and expression with an eye for producing highly purified, biological active proteins. Our expertise in protein purification is the basis for the complementary skill of assay design. We have produced many functional assays for GTP exchange factors (GEFs) (e.g., Dbs, LARG, RasGRF, SOS1, Tiam1, Vav1, Vav2), kinesins (e.g., Eg5, CenPE, MKLP2), myosins (e.g., cardiac, smooth, skeletal, and non-muscle isoforms), small G-proteins (e.g., Rho, Arf, Ral families), tubulins (e.g., tumor, plant, and fungal origins) and actin binding proteins; many of them are multi-protein assays that might have protein complexes of 3 or more subunits, e.g., a soluble sarcomere format and the Arp2/3 complex based assay. A particular speciality of Cytoskeleton's design formats is the ability to rapidly create lyophilized formulations in appropriate daily use or weekly use amounts. This enables HTS use and reproducible daily performance. Our experiences in antibody and ELISA technology complement the cytoskeletal and signal transduction focus. We support all of our services with a dedicated technical services department and years of laboratory experience in the fields of cell biology, cancer biology, cardio-vascular research, and neuroscience.
For more details on these four main areas click on one of the following:
Compound Screening and Drug Development
ELISA and antibody development
If you have an immediate question please select from the buttons above or e-mail us at tservice@cytoskeleton.com
Exceptional and Dedicated Technical Support:
- 40+ years of combined experience
- Expertise in: Protein biochemistry, cell biology, cancer biology, cardio-vascular research, and neuroscience.
Product and Service Performance:
- Modules with clearly defined deliverables
- Activity tested to specification
- Shipped in desiccated chambers & guaranteed for one year
- Proven results (See citation tab above for examples)
- Satisfied clients (See examples below)
Current Clients Include:
- Merck & Co., Inc.
- Eli Lilly & Co.
- Amgen, Inc.
- Abbott Laboratories
- Pfizer, Inc.
- Astra-Zeneca plc
- GlaxoSmithKline plc
- Genentech, Inc.
- Johnson & Johnson
- Bristol-Myers Squibb
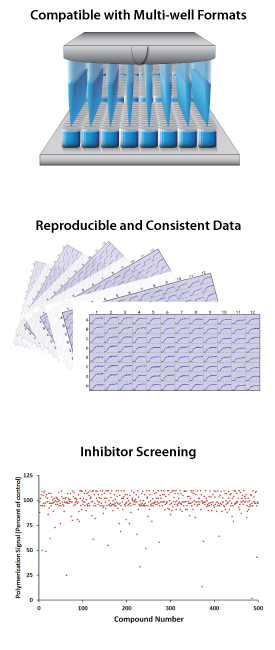
Cytoskeleton, Inc. has been a reliable source of compound screening data, custom protein production, assay design & development and antibody/ELISA development for past two decades. In 90% of cases, reports and products are delivered on time and within specifications, some projects have unpredictable outcomes and might take more time to understand, modify protocols and fulfill requirements. Over the past twenty years we have achieved a 97% customer satisfaction score. We support all of our services with a dedicated technical services department and years of laboratory experience in the fields of cell biology, cancer biology, cardio-vascular research and neuroscience. Below is a brief description of each area, for more information please click on the title eaction section:
Compound Screening Service
Examples of compound screening services:
1. SOS1 and Dbs GTP exchange factor assays using Mant-GTP or Bodipy-GTP nucleotide reporters, with monthly deliveries of 10 to 30 compounds, results turnaround within 1 week.
2. Calcium activated sarcomeric myosin S1 ATPase assays with tropomyosin/troponin coated F-actin (thin filaments) as substrate. Determine shift in pCa on addition of 24 compounds at two concentrations, results turnaround within 4 weeks.
3. CenPE and Eg5 kinesin motor IC50 determinations using microtubule stimulated ATPase assays, with monthly deliveries of 10 to 30 compounds, results turnaround within 1 week.
4. Cancer tubulin (cell line and tumor derived) polymerization assays to determine IC50 of anti-tumor drugs, 4 to 22 compounds, results turnaround within 3 weeks.
5. Tubulin/microtubule polymerization assays with single concentration screening, 10 to 160 compounds, results turnaround within 2 weeks.
Purified, high biological activity recombinant and native source proteins
Examples of protein purification are:
1. Cardiac heavy merosmyosin produced 200 x 1 mg in 8 weeks with >90% purity, and delivered lyophilized product for high reporducibility.
2. Isolated 1.0-5.0 mg of 90% pure tubulin from 50 g of cells. Delivered a lyophilized formulation with greater than 85% polymerization efficiency. Cancer cell line tubulins are useful for developing drugs targeted toward cancer cells.
3. Small G-protein Arf1 and Arf6 within 8 weeks to >85% purity, and suitable for 10,000 GEF assays (50 x 1 mg).
We perform full quality control, which includes:
1. Biological activity determination depending on the requirements of the customer.
2. SDS-PAGE of purified proteins, stained with coomassie blue, and scanned with a densitometer, to estimate purity.
3. Full quantitation of yield, in mg per g tissue.
Enzyme Assay development
Examples of assay development are:
1. Calcium activated S1 myosin atpase assay on cardiac thin filament protein with EC50 = 6.0 µM calcium, within 8 weeks. Microliter scale (100 µl) and with a CV= 8%.
2. Small G-protein Arf1 and Arf6 GEF assays within 4 weeks with CV's less than 8%.
3.Microtubule stablization development for kinesin motor drug screen, resulting in one week room temperature stable preparation of microtubules. CV's less than 9% and day to day variation 8%.
Antibody and ELISA development
Examples of ELISA development are:
1. Developed a tubulin capture ELISA plate which utilizes a novel polyclonal antibody to capture all isoforms, PTM modified and all species of tubulin. This plate can be used in a sandwich format to probe for isoforms, PTMs or recombinant tubulins with the addition of an adjunct primary antibody. CV = 5.4%.
2. Development of a total Rho protein capture ELISA plate which is used to measure total Rho for comparison to activated Rho. CV = 5.2%.
If you have further interest, please send an e-mail describing your requirements to tservice@cytoskeleton.com
Cytoskeleton provides custom services on a confidential basis, and as such there are limited published citations of our work. Representative examples of publications coming from our services are 1) tubulin polymerization assays (ref.1) and plant tubulin purification (ref.2). We can also provide professional references of services performed upon request.
References:
1) In Vitro and In Vivo Anticancer Activities of Synthetic Macrocyclic Ketone Analogues of Halichondrin B1. CANCER RESEARCH 61, 1013–1021, February 1, 2001. Murray J. Towle, Kathleen A. Salvato, Jacqueline Budrow, Bruce F. Wels, Galina Kuznetsov, Kimberley K. Aalfs, Susan Welsh, Wanjun Zheng, Boris M. Seletsky, Monica H. Palme, Gregory J. Habgood, Lori A. Singer, Lucian V. DiPietro, Yuan Wang, Jack J. Chen, David A. Quincy, Ashley Davis, Kentaro Yoshimatsu, Yoshito Kishi, Melvin J. Yu, and Bruce A. Littlefield (2001).
2) The herbicide flamprop-M-methyl has a new antimicrotubule mechanism of action.Stefan Tresch, Ricarda Niggeweg and Klaus Grossmann. Pest Manag Sci 64: 1195–1203 (2008).
Cytoskeleton, Inc. has been a reliable source of protein, assays, and drug screening services for nearly two decades. Past clients include:
Merck Eli Lilly Eisai Amgen Abbott Labs Zentaris GlaxoSmithKline
Genentech Johnson & Johnson Bristol-Myers Squibb Pfizer Wyeth
Question 1: What information do you need to send a quotation on developing an assay?
Question 2: Who will be my contact during the project?
Question 1. What information do you need to send a quotation on developing an assay?
Answer 1. Contact information, gene accession number, assay type, assay quantity, and CV required.
Question 2. Who will be my contact during the project?
Answer 2. You will be placed in direct contact with the scientist who is managing your project.



