* Limited stock available. If stock is not available, Cytoskeleton will produce a new batch upon request. Minimum order will apply. Inquire for more information.
Product Uses
- Measurement of microtubule-activated ATPase/GTPase assays.
- Identification/characterization of proteins or small molecules that affect motor ATPase/GTPase activity.
- Identification/characterization of proteins or small molecules that affect motor/microtubule interactions.
Material
Cytoplasmic dynein was purified from porcine brain by the procedure of B.M. Paschal et al. (1). The preparation contains dynein heavy chain (520 kDa), intermediate chain (54, 59, and 74 kDa) and light chains (c.20 kDa). The protein has been determined to be biologically active in a microtubule-activated ATPase assay (see below). The protein is supplied as a lyophilized white powder.
Storage and Reconstitution
The protein is supplied in 50 µg amounts which are designed for single use experiments. The powder is resuspended on ice with 50 µl of ice cold water to create a 1 mg/ml solution of dynein with a final buffer composition of 20 mM Tris-HCl pH 7.5, 50 mM KCl, 2 mM MgCl2, 0.5 mM EGTA plus 20% (w/v) sucrose. In this formulation the dynein will remain active for 3 days when stored at 4°C. Do not refreeze the protein as this will irreversibly denature the protein.
Purity
Protein purity is determined by scanning densitometry of a Coomassie-stained SDS-PAGE gradient gel. Figure 1, lane 9 shows 10 mg of CS-DN01 protein and purity was determined to be c.80%. The total protein in each tube will therefore be approximately 20% greater than the amount shown on the tube. Contaminants include kinesin, dynamin, dynactin and tubulin.
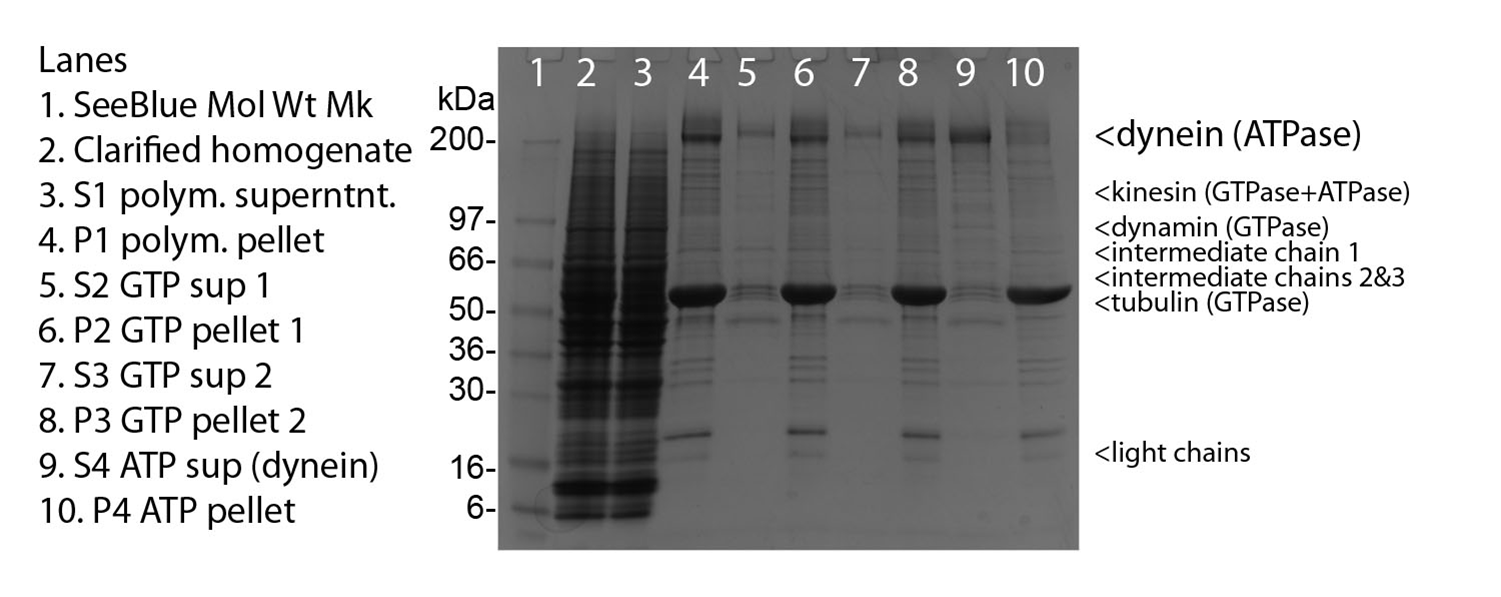
Figure 1. Dynein Motor Purification.
Legend: Porcine brain homogenate was used as a source of microtubules and dynein complex. Cycles of microtubule pelleting in nucleotide free, GTP, GTP and finally ATP released dynein from microtubules. Samples during purification were run on 4-20% gradient gel and stained with Coomassie blue. A 10 µg sample of cytoplasmic dynein protein is represented in lane 9. SeeBlue molecular weight markers are from Life Tech. The dyenin motor protein has an apparent molecular weight of 520 kDa, intermediate chains at 54, 59, 74 and light chains around 20 kDa. Protein amount was determined using Advanced Protein assay (Cat. # ADV02).
Microtubule Activated ATPase ASSAY
Cytoplasmic dynein ATPase and GTPase activities were measured by monitoring real time free phosphate generation using the Kinesin ATPase Assay Kit (Cat. # BK053). The assay is based upon an increase in absorbance at 650 nm. The absorbance at 650 nm is directly proportional to the amount of Pi generated in the kinesin ATPase/GTPase reaction. Under the conditions outlined below, an OD of 0.10 is equal to 400 pmoles of Pi (Figure 2).
Reagents
- Kinesin ATPase Endpoint Assay Kit (Cat. # BK053).
- CS-DN01 50 µg.
- 2.5 M sodium azide.
- PM12 buffer = 12 mM Pipes pH 7.0, 5 mM MgCl2.
Equipment
Monochromatic spectrophotometer (set to 650 nm) or a filter based spectrophotometer with a 650 nm filter and a bandwidth <50 nm.
Method (ATPase endpoint assay)
The reactions were conducted in half area 96 well plates (30 µl reaction volumes). Each reaction contains 0.8 µg of CS-DN01 protein, 2.0 µM taxol-stabilized microtubules (MTs), 10 µM taxol, 12 mM PIPES pH 7.0, 5 mM MgCl2, and 0.19 mM ATP. Control reactions were carried out in the absence of CS-DN01. These reactions gave readings of <0.1 OD over 20 min. The reaction mixture contains 4 mM azide which reduces kinesin ATPase. After 20 to 45 min incubation at 37 °C, reactions were measured in a SpectraMax 250 (Molecular Devices) to 650 nm absorbance wavelength. Typical assay results are shown in Figure 2. The nmoles of Pi was determined by the use of a phosphate standard curve (not shown).
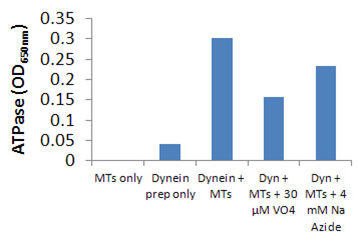
Figure 2. CS-DN01 microtubule-activated ATPase activity using the Kinesin ATPase Endpoint Assay Kit (Cat. # BK053). CS-DN01 ATPase was assayed in duplicate according to the method described. Control reactions without microtubules were compared to those with microtubules. 4 mM sodium azide mainly inhibits kinesin activity, whereas 30 µM sodium vanadate inhibits dynein.
1. Follow protocol to prepare MT solution at 2.0 mg/ml in PEM Buffer plus sequentially added taxol. 1 mg is enough for 96 assays.
a) Resuspend 1 mg >99% pure porcine tubulin (Cat. # T240) in 0.5 ml of ice cold PEM + 10% glycerol (adding extra GTP is not necessary).
b) Place at 37°C for 2 min.
c) Add 1 µl of 2 mM taxol (final conc = 4 µM), incubate for a further 2 min.
d) Add 1 µl of 2 mM taxol (final conc = 4+4 µM), incubate for a further 2 min.
e) Add 3 µl of 2 mM taxol (final conc = 4+4+12 µM), incubate for a further 2 min.
f) Place on the bench (not on ice or at 4°C) and the taxol-stabilized MTs are now ready. Shelf life is 3 days if not contaminated with bugs or other chemicals.
Note: For screening purposes, MTs are made and used the same day, do not use if more than 6 h old because phosphate will build up due to continued hydrolysis. This will add to background and reduce the S/N ratio.
2. Prepare compounds at 20X in 100% DMSO solution.
10 µl of stock solution at 2 mM compound is recommended.
3. Prepare motor mix as follows.
a) b) Prepare 10 ml PM12 buffer with 10 µM taxol final (50 µl of 2 mM taxol stock). Check reaction buffer for precipitate, make a new solution if precipitation is present.
c) Defrost 100 µg vial of DN01 by placing in a room temperature water bath until melted, then place on ice.
d) Mix the following in order at room temperature (RT) in a 20 ml polypropylene vial (not glass):
| # of assays | 384 | 192 | 96 | 30 | |
| 12800 | 6400 | 3200 | 800 | µl of PM12 +10µM taxol at RT. | |
| 1600 | 800 | 400 | 100 | µl of 2.0mg/ml MTs in PM12 plus 20uM taxol. | |
| 30 | 15 | 7 | 1.8 | µl of 100 mM ATP. | |
| 24 | 12 | 6 | 1.5 | µl of 2.5M sodium azide | |
| 400 | 200 | 100 | 25 | µl of DN01 1mg/ml. |
4. Reaction conditions.
a) Pipette 1.5 µl of compound into each well.
b) Pipette 30 µl of motor mix into each well.
c) Shake for 1 min at 400 rpm on a plate shaker to mix well.
d) Incubate at 37 °C for 20 min (enhancers) or 45 min (inhibitors).
e) Quench and develop signal by pipetting 120 µl of Malachite green solution (CytoPhos reagent in BK053 kit) into each well.
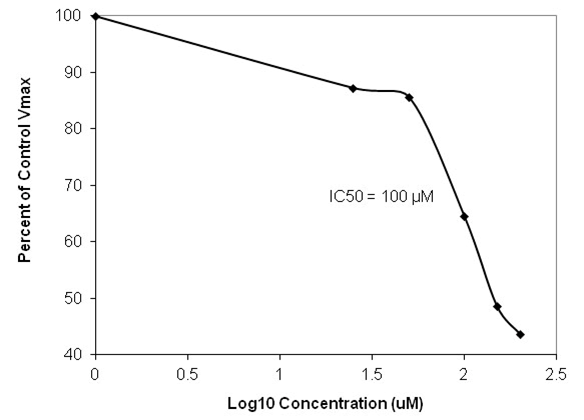
Figure 3. Dose response curve of CS-DN01 microtubule-activated ATPase in the presence of Ciliobrevin A using the Kinesin ATPase Endpoint Assay Kit (Cat. # BK053). CS-DN01 ATPase was assayed in duplicate according to the method described. 4 mM sodium azide was included to inhibit kinesin activity. Cilibrevin A inhibited with an IC50 of approx. 100 µM which is similar to that reported by Firestone et al. (2).
References
1. Paschal B.M. et al. 1991. Methods in Enzymology.196, 181-191.
2. Firestone A.J. et al. 2012. Nature. 484, 125-129.
For product Datasheets and MSDSs please click on the PDF links below.
Coming soon! If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com
