
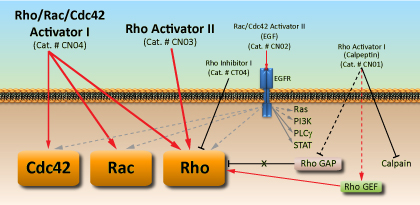
Activators
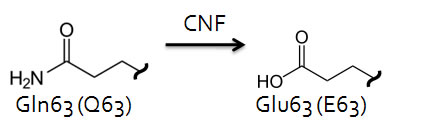
Direct activation of the Rho family small G-proteins is achieved by virulence factors from some pathogenic bacteria (e.g. Salmonella, Yersinia and uropathogenic E. coli). These small G-protein activators fall into two main classes: those that act as GEFs (e.g. SopE, SopE2, Map and IpgB1/2) and those that covalently modify Rho family GTPases conferring constitutive activity (e.g. CNF1/2/3, CNFy and DNT). The CNF/DNT activators function by deamidating Gln63 (Rho)/Gln61 (Rac/Cdc42) in the Switch II region of the small G-proteins, which blocks both intrinsic and GAP stimulated GTPase activity, resulting in the accumulation of GTP-bound active G-proteins. At Cytoskeleton, Inc., we have developed direct small G-protein activators that have different levels of selectivity for Rho GTPases [Rho Activator II (Cat# CN03) and Rho/Rac/Cdc42 Activator I (Cat. # CN04)] based on the catalytic activity of the CNF class of activators.
Several small G-protein activators that have classically been used to study Rho family small G-proteins transmit their signals through membrane bound receptors (e.g. EGF (Cat# CN02), PDGF, LPA, Sphingosine-1-phosphate and bradykinin). These molecules can be classified as both non-selective and indirect activators since they also activate other proteins (e.g. Ras, PI3K and PLC) and their activation of the Rho family GTPases is accomplished through the activation of GEFs.Calpeptin (Cat# CN01) is a more selective indirect activator of the Rho subfamily (RhoA, B and C) of GTPases. Calpeptin is a peptidic small molecule with multiple biological activities that has traditionally been used as a calpain (cysteine protease) inhibitor. The biological activity of calpeptin that relates to Rho GTPase activation is its ability to inhibit the SHP-2 tyrosine phosphatase. SHP-2 is known to dephosphorylate the Rho GTPase negative regulator p190RhoGAP, which enables it to stimulate the GTPase activity of Rho proteins leading to their inactivation. Current literature supports the hypothesis that calpeptin inhibition of the SHP-2 tyrosine phosphatase results in the accumulation of active Rho small G-proteins by keeping its negative regulator p190RhoGAP in its inactive state.
Available Activators and Inhibitors | |
| Product | Cat. # |
| Rho Activator II | (Cat. # CN03) |
| Rho Inhibitor I | (Cat. # CT04) |
| Rho Pathway Inhibitor I Rho kinase (ROCK) Inhibitor Y27632 | (Cat. # CN06) |
| Rho/Rac/Cdc42 Activator I | (Cat. # CN04) |
| Rho Activator I | (Cat. # CN01) |
| Rac/Cdc42 Activator II | (Cat. # CN02) |
| C3 Transferase Protein | (Cat. # CT03) |
Figure 2. Rho Activator II (CN03) induces actin stress fibers in serum starved 3T3 cells.
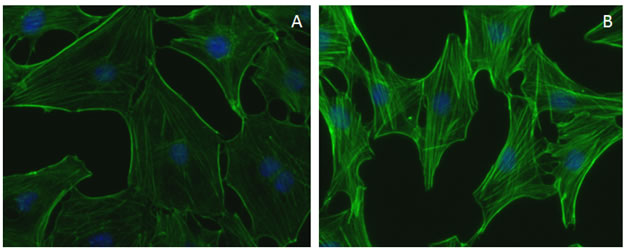
Figure 2. Actin cytoskeleton morphological changes induced by CN03 treatment of Swiss 3T3 cells. Swiss 3T3 fibroblasts plated on glass coverslips, grown for 2 days in DMEM plus 10% FBS, serum starved for 24 h in media containing 1% FBS followed by 24 h in serum free media. Cells were treated with a buffer control (A) or 1 µg/ml CN03 for 2 h at 37°C/5% CO2. Cells were then fixed, stained with Acti-stain™ 488 phalloidin (Cat.#PHDG1), and visualized by fluorescence microscopy. Images were taken at a magnification of 40x. The control cells (A) exhibited mainly edge ruffling, some lamellipodia and very few stress fibers, whereas treatment with CN03 (B) resulted in the development of abundant stress fibers with some edge ruffling. Under similar conditions, RhoA was activated ~10-fold as determined using the RhoA G-LISA® activation assay (Cat.# BK124).
Inhibitors
C3 Transferase from Clostridium botulinum is widely used to directly inhibit endogenous RhoA, B and C proteins by ADP-ribosylation on asparagine 41 in the effector binding domain of the GTPase. It will not inhibit other Rho family proteins such as Rac or Cdc42. The native exoenzyme typically takes 24h to inactivate Rho. To improve this reagent, scientists at Cytoskeleton, Inc. have covalently attached the C3 transferase to a proprietary cell penetrating moiety (Cat# CT04). CT04 can efficiently enter cells and inactivate cellular Rho proteins in as little as 2 h. CT04 has been used to inactivate Rho to an efficiency of 75-95% in fibroblasts, neurons, epithelial, endothelial, and hematopoietic cells as well as other primary and immortalized cell lines.
The small molecule Y-27632 inhibits the Rho signal transduction pathway by potently inhibiting the Rho effector proteins ROCK I and ROCK II. The Ki of 220 nM for ROCK I and 300 nM for ROCK II. The affinities for other Rho effector kinases , such as citron and PKN are at least 10X lower. Effector kinases in the Rho family pathway such as the Rac/Cdc42 effector, p21-activated kinase (PAK) are not affected. Cytoskeleton, Inc., supplies the ROCK inhibitor in quality controlled, cost effective aliquots (Cat# CN06). The uptake of cell permeable CN06 is time and temperature dependent, uptake has been shown to reach a plateau at 30 minutes at 37°C and is negligible at 4°C. ROCK proteins are ubiquitously expressed making this inhibitor applicable to the study of almost all cell lines and tissues.
