Product Uses Include
- Determination of the activity and GTPase specificity of uncharacterized GAPs.
- Biochemical characterization of small GTPases and their associated GAPs.
- Examination of the regulation of GAP activity by different cofactors or protein domains.
- Screen the mutant protein of either GAPs or GTPases for activity and substrate specificity.
- Identification of GAP inhibitors in HTS (high throughput screen) format. Please inquire for significant discounts on large quantities of any reagents in this kit.
Introduction
The Ras superfamily of small GTPases (such as Ras, Rho, Rab, Arf and Ran proteins) serve as binary switches cycling between a GDP-bound “OFF state” and a GTP-bound “ON state”, which mediates signals downstream. In cells, the cycling between the two states is mainly controlled by two types of regulatory proteins: the activating Guanine nucleotide Exchange Factors (GEFs) and the inactivating GTPase Activating Proteins (GAPs).
Small G-proteins of the Ras superfamily typically have an inrinsic GTPase activity, meaning that after they get activated (GTP-bound) by GEFs, they eventually “turn themselves off”. The intrinsic GTPase rate, however, is normally very slow, so the “automatic” turning off process can take up to hours.
Because of the slow intrinsic GTPase activity in most Ras superfamily GTPases, their GAPs are important signaling molecules that function to terminate their signaling. GAPs function by binding to the active, GTP-bound, form of their target G-proteins and greatly stimulating the G-protein’s intrinsic GTPase activity, thereby quickly turning off the G-protein.
This GAP assay kit measures the amount of inorganic phosphate (Pi) that is produced as a result of G-protein dependent hydrolysis of GTP to GDP + Pi. The kit contains all the reagents needed for the assay including GTPases, a positive control RhoGAP, carefully optimized buffers and phosphate detection reagent. The assay can be performed in either 96-well plates or in 384-well plates. There is enough reagents for 80 assays (20 for each provided GTPase) in the 96-well format and 160 assays (40 for each provided GTPase) in the 384-well format
While the kit comes with Cdc42, Rac1, RhoA and Ras proteins, it can also be used for any other Ras superfamily GTPase. See our G-protein product family web page for other GTPases available from Cytoskeleton.
For a kit to measure GEF activity, see Cat. # BK100.
Kit contents
THe kit contains sufficient materials for 80-160 assays. The following components are included:
- Reaction Buffer
- His-tagged Cdc42 protein (Cat. # CD01)
- His-tagged Rac1 protein(Cat. # RC01)
- His-tagged RhoA protein (Cat. # RH01)
- His-tagged Ras protein (Cat. # RS01)
- GST-tagged p50RhoGAP, catalytic domain (Cat. # GAS01)
- GTP stock (Cat. # BST06).
- CytoPhos™ reagent.
- 96-well clear half area flat bottom plate
- Detailed instructions and troubleshooting manual.
Equipment needed
- 96-well or 384-well plate spectrophotometer capable of measuring absorbance at 650 nm wavelength
Example results
The GTPase activity of RhoA with and without the addition of the catalytic domain of p50RhoGAP was tested (Fig 1).
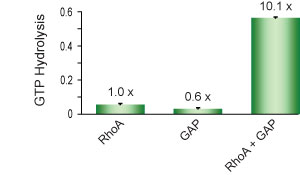
Figure 1. RhoGAP activity measured as GTP hydrolysis by RhoA protein. Each reaction contained reaction buffer + GTP with the addition of RhoA alone (RhoA), RhoGAP alone (GAP), or RhoA + RhoGAP (RhoA + GAP). Reactions were incubated at 37°C for 20 min. Phosphate generated by hydrolysis of GTP was measured by the addition of CytoPhos™ reagent and reading of absorbance at 650 nm.
For product Datasheets and MSDSs please click on the PDF links below. For additional information, click on the FAQs tab above or contact our Technical Support department at tservice@cytoskeleton.com
Question 1: How should I prepare my protein for testing in the GAP assay?
Answer 1: All protein samples and buffers must be free of phosphate prior to beginning the RhoGAP assay biochem kit (Cat. # BK105). If your protein is in PBS buffer then it must be dialyzed twice in 1000 volumes of 50 mM PIPES pH 7.0 or 50 mM Tris pH 7.5 buffer to reduce the phosphate content to non-detectable levels. For the assay itself, prepare an 8X concentration of protein or compound in 20 mM Hepes buffer pH 7.4 or similar or for compounds with low solubility in water, use Milli-Q water and 10% DMSO. We recommend a final concentration of 30 μM-100 μM for compound screening, or 10 to 1000 nM for proteins. A titration of your compound/protein is recommended when performing the GAP assay.
Question 2: Can I test cell or tissue lysates with the RhoGAP assay kit to screen for GAP proteins?
Answer 2: No, we do not recommend using lysates from cells or tissue samples with the RhoGAP assay kit (Cat. # BK105). In lysates there are too many complicating factors for a sensitive and accurate measurement of GAP activity. Instead, we recommend using a purified compound or protein that has been isolated with immunoprecipitation or purified by His-tagged chromatography.
Question 3: Does the RhoGAP protein work with Ras GTPase?
Answer 3: The Ras GTPase is not a good substrate for the p50 RhoGAP protein that is included with the RhoGAP assay kit (Cat. # BK105). We provide the Ras protein as a substrate to screen any potential Ras GAPs that you may wish to study. Please see these papers for characterization of RhoGAP and its lack of effect on Ras.
Garrett, M. D., Self, A. J., van Oers, C., and Hall, A . (1989) J. Biol. Chem. v 264, pp. 10-13.
Garrett, M. D., Major, G. N., Totty, N., and Hall, A. (1991) Biochem. J. v 276, pp. 833-336.
Lancaster, C. A., Taylor-Harris, P. M., Self, A. J., Brill, S., van Erp, H.E., and Hall, A. (1994) J. Biol Chem. v 269, pp. 1137-1142.
If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com











