| Author | Title | Journal | Year | Article Link |
|---|
| Grzywa, Renata et al. | Isothiocyanates as Tubulin Polymerization Inhibitors—Synthesis and Structure–Activity Relationship Studies | International Journal of Molecular Sciences | 2023 | ISSN 1422-0067 |
| Hou, Zhenyan et al. | S-72, a Novel Orally Available Tubulin Inhibitor, Overcomes Paclitaxel Resistance via Inactivation of the STING Pathway in Breast Cancer | Pharmaceuticals | 2023 | ISSN 1424-8247 |
| Liang, Baoxia et al. | Novel Indole-Containing Hybrids Derived from Millepachine: Synthesis, Biological Evaluation and Antitumor Mechanism Study | Molecules | 2023 | ISSN 1420-3049 |
| Wang, Chao et al. | Design, synthesis, and biological evaluation of 4-aryl-9H-carbazoles as tubulin polymerization inhibitors with potent anticancer activities | Arabian Journal of Chemistry | 2023 | |
| Horgan, Mark James et al. | Identification of Novel β-Tubulin Inhibitors Using a Combined In Silico/In Vitro Approach | Journal of Chemical Information and Modeling | 2023 | ISSN 1549-960X |
| Li, Yuan-Yuan et al. | Tetrabromobisphenol A-bis(2,3-dibromopropyl ether) impairs Postnatal Testis Development in Mice: The Microtubule Cytoskeleton as a Sensitive Target | Environment & Health | 2023 | ISSN 2833--8278 |
| Dong, Haiyang et al. | Design, synthesis and biological evaluation of tetrahydroquinoxaline sulfonamide derivatives as colchicine binding site inhibitors | RSC Advances | 2023 | ISSN 2046-2069 |
| Morishita, Jun et al. | Identification of a small RhoA GTPase inhibitor effective in fission yeast and human cells | Open Biology | 2023 | ISSN 2046-2441 |
| Yao, Yongfang et al. | Angiogenesis and anti-leukaemia activity of novel indole derivatives as potent colchicine binding site inhibitors | Journal of Enzyme Inhibition and Medicinal Chemistry | 2022 | ISSN 1475-6374 |
| Wang, Zhan et al. | Design, synthesis and biological evaluation of colchicine glycoconjugates as tubulin polymerization inhibitors | Bioorganic and Medicinal Chemistry | 2022 | ISSN 1464-3391 |
| Ma, Juan et al. | Discovery of Novel 3,4-Dihydro-2(1H)-Quinolinone Sulfonamide Derivatives as New Tubulin Polymerization Inhibitors with Anti-Cancer Activity | Molecules | 2022 | ISSN 1420-3049 |
| Liu, Chao et al. | Synthesis and biological evaluation of BU-4664L derivatives as potential anticancer agents | Bioorganic and Medicinal Chemistry Letters | 2022 | ISSN 1464-3405 |
| Kumar N, Manoj et al. | Benzothiazole-[1,2,3]triazolo[5,1-a]isoindoles: Synthesis, anticancer activity, bioavailability and in silico studies against Gama-Tubulin protein | Journal of Molecular Structure | 2022 | ISSN 0022-2860 |
| Wang, Guangcheng et al. | Design, synthesis and biological evaluation of novel 2-phenyl-4,5,6,7-tetrahydro-1H-indole derivatives as potential anticancer agents and tubulin polymerization inhibitors | Arabian Journal of Chemistry | 2022 | ISSN 1878-5352 |
| Woo, Jung AA et al. | β-arrestin1 promotes tauopathy by transducing GPCR signaling, disrupting microtubules and autophagy | Life science alliance | 2021 | ISSN 2575--1077 |
| Lead optimization of novel quinolone chalcone compounds by a structure-activity relationship (SAR) study to increase efficacy and metabolic stability | | 2021 | Article Link |
| Yang, Mei et al. | C118P, a novel microtubule inhibitor with anti-angiogenic and vascular disrupting activities, exerts anti-tumor effects against hepatocellular carcinoma | Biochemical Pharmacology | 2021 | ISSN 0006--2952 |
| Sun, Chiao Yin et al. | LMBD1 protein participates in cell mitosis by regulating microtubule assembly | Biochemical Journal | 2021 | ISSN 1470-8728 |
| Liang, Dong et al. | Identification of anthelmintic parbendazole as a therapeutic molecule for HNSCC through connectivity map-based drug repositioning | Acta Pharmaceutica Sinica B | 2021 | ISSN 2211-3843 |
| Rahimzadeh Oskuei, Sara et al. | Design, synthesis and biological evaluation of novel imidazole-chalcone derivatives as potential anticancer agents and tubulin polymerization inhibitors | Bioorganic Chemistry | 2021 | ISSN 1090-2120 |
| Wang, Chao et al. | Design, synthesis and biological evaluation of 1-Aryl-5-(4-arylpiperazine-1-carbonyl)-1H-tetrazols as novel microtubule destabilizers | Journal of Enzyme Inhibition and Medicinal Chemistry | 2021 | ISSN 1475-6374 |
| Patel, Onisha et al. | Structural basis for small molecule targeting of Doublecortin Like Kinase 1 with DCLK1-IN-1 | Communications Biology | 2021 | ISSN 2399-3642 |
| Fareed, Momen R. et al. | New multi-targeted antiproliferative agents: Design and synthesis of ic261-based oxindoles as potential tubulin, ck1 and egfr inhibitors | Pharmaceuticals | 2021 | ISSN 1424-8247 |
| Würtz, Martin et al. | Reconstitution of the recombinant human γ-tubulin ring complex | Open Biology | 2021 | ISSN 2046-2441 |
| Zhernov, Ilia et al. | Intrinsically Disordered Domain of Kinesin-3 Kif14 Enables Unique Functional Diversity | Current Biology | 2020 | ISSN 1879-0445 |
| Baker, Stacey J. et al. | A Contaminant Impurity, Not Rigosertib, Is a Tubulin Binding Agent | Molecular Cell | 2020 | ISSN 1097-4164 |
| Skidmore, Lillian et al. | ARX788, a site-specific anti-HER2 antibody–drug conjugate, demonstrates potent and selective activity in HER2-low and T-DM1–resistant breast and gastric cancers | Molecular Cancer Therapeutics | 2020 | ISSN 1538-8514 |
| Wang, Yanming et al. | Antibody-drug conjugate using ionized CYS-linker-mmae as the potent payload shows optimal therapeutic safety | Cancers | 2020 | ISSN 2072-6694 |
| Morita, Ken et al. | Allosteric Activators of Protein Phosphatase 2A Display Broad Antitumor Activity Mediated by Dephosphorylation of MYBL2 | Cell | 2020 | ISSN 1097-4172 |
| Dillon, Gregory M. et al. | Acute inhibition of the CNS-specific kinase TTBK1 significantly lowers tau phosphorylation at several disease relevant sites | PLoS ONE | 2020 | ISSN 1932-6203 |
| Ullah, Imran et al. | An Antiparasitic Compound from the Medicines for Malaria Venture Pathogen Box Promotes Leishmania Tubulin Polymerization | ACS Infectious Diseases | 2020 | ISSN 2373-8227 |
| Song, Ming Yu et al. | Exploring diverse-ring analogues on combretastatin a4 (Ca-4) olefin as microtubule-targeting agents | International Journal of Molecular Sciences | 2020 | ISSN 1422-0067 |
| Chen, Shih Yin et al. | Exosomal 2′,3′-CNP from mesenchymal stem cells promotes hippocampus CA1 neurogenesis/neuritogenesis and contributes to rescue of cognition/learning deficiencies of damaged brain | Stem Cells Translational Medicine | 2020 | ISSN 2157-6580 |
| Zdioruk, Mykola et al. | A new inhibitor of tubulin polymerization kills multiple cancer cell types and reveals p21-mediated mechanism determining cell death after mitotic catastrophe | Cancers | 2020 | ISSN 2072-6694 |
| Mirzaei, Salimeh et al. | Synthesis, structure-activity relationship and molecular docking studies of novel quinoline-chalcone hybrids as potential anticancer agents and tubulin inhibitors | Journal of Molecular Structure | 2020 | ISSN 0022-2860 |
| Palumbo, Valeria et al. | Drosophila Morgana is an Hsp90-interacting protein with a direct role in microtubule polymerization | Journal of Cell Science | 2020 | ISSN 1477-9137 |
| Barnes, Natalie G. et al. | A 2-step synthesis of Combretastatin A-4 and derivatives as potent tubulin assembly inhibitors | Bioorganic and Medicinal Chemistry | 2020 | ISSN 1464-3391 |
| Liu, Qian et al. | Identification of a lathyrane-type diterpenoid EM-E-11-4 as a novel paclitaxel resistance reversing agent with multiple mechanisms of action | Aging | 2020 | ISSN 1945-4589 |
| Abdel-Rahman, Somaya A. et al. | Cyclohepta[ b]thiophenes as Potential Antiproliferative Agents: Design, Synthesis, in Vitro, and in Vivo Anticancer Evaluation | ACS Pharmacology and Translational Science | 2020 | ISSN 2575-9108 |
| Mao, Bai Ping et al. | CaMSAP2 is a microtubule minus-end targeting protein that regulates BTB dynamics through cytoskeletal organization | Endocrinology | 2019 | ISSN 1945-7170 |
| Behbahani, Fatemeh Shaebani et al. | Synthesis and biological evaluation of novel benzo[c]acridine-diones as potential anticancer agents and tubulin polymerization inhibitors | Archiv der Pharmazie | 2019 | ISSN 1521-4184 |
| Cui, Ying Jie et al. | Synthesis of novel pyrazole derivatives and their tumor cell growth inhibitory activity | Molecules | 2019 | ISSN 1420-3049 |
| Xia, Xiaoyu et al. | Leukemia Cell Cycle Chemical Profiling Identifies the G2-Phase Leukemia Specific Inhibitor Leusin-1 | ACS Chemical Biology | 2019 | ISSN 1554-8937 |
| Wang, Yanming et al. | Novel silyl ether-based acid-cleavable antibody-MMAE conjugates with appropriate stability and efficacy | Cancers | 2019 | ISSN 2072-6694 |
| Shaik, Thoukhir B. et al. | Evaluation of Anticancer and Anti-Mitotic Properties of Quinazoline and Quinazolino-Benzothiadiazine Derivatives | Anti-Cancer Agents in Medicinal Chemistry | 2019 | ISSN 1871-5206 |
| Karimikia, Ehsan et al. | Colchicine-like β-acetamidoketones as inhibitors of microtubule polymerization: Design, synthesis and biological evaluation of in vitro anticancer activity | Iranian Journal of Basic Medical Sciences | 2019 | ISSN 2008-3874 |
| Eberle-Singh, Jaime A. et al. | Effective delivery of a microtubule polymerization inhibitor synergizes with standard regimens in models of pancreatic ductal adenocarcinoma | Clinical Cancer Research | 2019 | ISSN 1557-3265 |
| Zhai, Min'an et al. | 3,5-Diaryl-1H-pyrazolo[3,4-b]pyridines as potent tubulin polymerization inhibitors: Rational design, synthesis and biological evaluation | European Journal of Medicinal Chemistry | 2019 | ISSN 1768-3254 |
| Nixon, Gemma L. et al. | Repurposing and reformulation of the antiparasitic agent flubendazole for treatment of cryptococcal meningoencephalitis, a neglected fungal disease | Antimicrobial Agents and Chemotherapy | 2018 | ISSN 1098-6596 |
| Qi, Jianguo et al. | Synthesis and biological evaluation of N-substituted 3-oxo-1,2,3,4-tetrahydro-quinoxaline-6-carboxylic acid derivatives as tubulin polymerization inhibitors | European Journal of Medicinal Chemistry | 2018 | ISSN 1768-3254 |
| Simpkins, Scott W. et al. | Predicting bioprocess targets of chemical compounds through integration of chemical-genetic and genetic interactions | PLoS Computational Biology | 2018 | ISSN 1553-7358 |
| Sato-Kaneko, Fumi et al. | Discovery of a Novel Microtubule Targeting Agent as an Adjuvant for Cancer Immunotherapy | BioMed Research International | 2018 | ISSN 2314-6141 |
| Dilworth, David et al. | The prolyl isomerase FKBP25 regulates microtubule polymerization impacting cell cycle progression and genomic stability | Nucleic Acids Research | 2018 | ISSN 1362-4962 |
| Tian, Zhenhua et al. | Biological activity and interaction mechanism of the diketopiperazine derivatives as tubulin polymerization inhibitors | RSC Advances | 2018 | ISSN 2046-2069 |
| Huan, L C et al. | Exploration of Some Thiazolidine-2, 4-dione and 2-Oxoindoline Derivatives Incorporating 3, 4, 5-Trimethoxybenzyl Moiety as Novel Anticancer Agents | Letters in Drug … | 2018 | Article Link |
| Lukinavičius, Gražvydas et al. | Fluorescent dyes and probes for super-resolution microscopy of microtubules and tracheoles in living cells and tissues | Chemical Science | 2018 | ISSN 2041-6539 |
| Chen, Minfeng et al. | Pericyte-targeting prodrug overcomes tumor resistance to vascular disrupting agents | Journal of Clinical Investigation | 2017 | ISSN 1558-8238 |
| Wieczorek, Anna et al. | Synthesis and evaluation of biological properties of ferrocenyl-podophyllotoxin conjugates | Dalton Transactions | 2017 | ISSN 1477-9234 |
| Zollo, Massimo et al. | PRUNE is crucial for normal brain development and mutated in microcephaly with neurodevelopmental impairment | Brain | 2017 | ISSN 1460-2156 |
| Kowalczyk, Karolina et al. | Colchicine metallocenyl bioconjugates showing high antiproliferative activities against cancer cell lines | Dalton Transactions | 2017 | ISSN 1477-9234 |
| Tantak, Mukund P. et al. | Design and synthesis of bis(indolyl)ketohydrazide-hydrazones: Identification of potent and selective novel tubulin inhibitors | European Journal of Medicinal Chemistry | 2017 | ISSN 1768-3254 |
| Sun, Maolin et al. | Synthesis and bioevaluation of N,4-diaryl-1,3-thiazole-2-amines as tubulin inhibitors with potent antiproliferative activity | PLoS ONE | 2017 | ISSN 1932-6203 |
| Wu, Yue et al. | Design and synthesis of 5-aryl-4-(4-arylpiperazine-1-carbonyl)-2H-1,2,3-triazole derivatives as colchicine binding site inhibitors | Scientific Reports | 2017 | ISSN 2045-2322 |
| Xu, Qile et al. | Design, synthesis and structure-Activity relationship of 3,6-diaryl-7H-[1,2,4]triazolo[3,4-b][1,3,4]thiadiazines as novel tubulin inhibitors | Scientific Reports | 2017 | ISSN 2045-2322 |
| Senese, Silvia et al. | Microtubins: A novel class of small synthetic microtubule targeting drugs that inhibit cancer cell proliferation | Oncotarget | 2017 | ISSN 1949-2553 |
| Pearson, Brandon L. et al. | Identification of chemicals that mimic transcriptional changes associated with autism, brain aging and neurodegeneration | Nature Communications | 2016 | ISSN 2041-1723 |
| Fukuda, Yasunori et al. | Tubulin is a molecular target of the Wnt-activating chemical probe | BMC Biochemistry | 2016 | ISSN 1471-2091 |
| Kamal, Ahmed et al. | Design, synthesis and antiproliferative activity of the new conjugates of E7010 and resveratrol as tubulin polymerization inhibitors | Organic and Biomolecular Chemistry | 2016 | ISSN 1477-0520 |
| Thapa, Pritam et al. | Far-Red Light-Activatable Prodrug of Paclitaxel for the Combined Effects of Photodynamic Therapy and Site-Specific Paclitaxel Chemotherapy | Journal of Medicinal Chemistry | 2016 | ISSN 1520-4804 |
| Yoshitake, Jun et al. | Modification of tau by 8-nitroguanosine 3′,5′-cyclic monophosphate (8-nitro-cGMP): Effects of nitric oxide-linked chemical modification on tau aggregation | Journal of Biological Chemistry | 2016 | ISSN 1083-351X |
| Hasanpourghadi, Mohadeseh et al. | Targeting of tubulin polymerization and induction of mitotic blockage by Methyl 2-(5-fluoro-2-hydroxyphenyl)-1H-benzo[d]imidazole-5-carboxylate (MBIC) in human cervical cancer HeLa cell | Journal of Experimental and Clinical Cancer Research | 2016 | ISSN 1756-9966 |
| Cai, De et al. | YSL-12, a novel microtubule-destabilizing agent, exerts potent anti-tumor activity against colon cancer in vitro and in vivo | Cancer Chemotherapy and Pharmacology | 2016 | ISSN 1432-0843 |
| Fu, Ying et al. | The contrasting catalytic efficiency and cancer cell antiproliferative activity of stereoselective organoruthenium transfer hydrogenation catalysts | Dalton Transactions | 2016 | ISSN 1477-9234 |
| Magalhaes, Luma G. et al. | Discovery of a series of acridinones as mechanism-based tubulin assembly inhibitors with anticancer activity | PLoS ONE | 2016 | ISSN 1932-6203 |
| Remers, William A. et al. | Synthesis and Antitumor Activity of Heterocycles Related to Carbendazim | Journal of Heterocyclic Chemistry | 2015 | ISSN 1943--5193 |
| Fawzy, Iten M. et al. | Newly Designed and Synthesized Curcumin Analogs with in vitro Cytotoxicity and Tubulin Polymerization Activity | Chemical biology & drug design | 2015 | ISSN 1747--0285 |
| Seashore-Ludlow, Brinton et al. | Harnessing connectivity in a large-scale small-molecule sensitivity dataset | Cancer Discovery | 2015 | ISSN 2159-8290 |
| Yan, Jun et al. | A novel synthetic compound exerts effective anti-tumour activity in vivo via the inhibition of tubulin polymerisation in A549 cells | Biochemical Pharmacology | 2015 | ISSN 1873-2968 |
| Sheldon, Jonathon E. et al. | Photoswitchable anticancer activity via trans-cis isomerization of a combretastatin A-4 analog | Organic and Biomolecular Chemistry | 2015 | ISSN 1477-0520 |
| Calles, Antonio et al. | Tivantinib (ARQ 197) efficacy is independent of MET inhibition in non-small-cell lung cancer cell lines | Molecular Oncology | 2015 | ISSN 1878-0261 |
| Xu, Qile et al. | Synthesis and biological evaluation of 3-alkyl-1,5-diaryl-1H-pyrazoles as rigid analogues of combretastatin A-4 with potent antiproliferative activity | PLoS ONE | 2015 | ISSN 1932-6203 |
| Wu, Shaoyu et al. | Bis-cyclopropane analog of disorazole C1 is a microtubuledestabilizing agent active in ABCB1-overexpressing human colon cancer cells | Oncotarget | 2015 | ISSN 1949-2553 |
| Mu, Yan et al. | The novel tubulin polymerization inhibitor MHPT exhibits selective anti-tumor activity against rhabdomyosarcoma in vitro and in vivo | PLoS ONE | 2015 | ISSN 1932-6203 |
| Choi, Bo Hwa et al. | Suprafenacine, an Indazole-Hydrazide Agent, Targets Cancer Cells Through Microtubule Destabilization | PLOS ONE | 2014 | ISSN 1932--6203 |
| Shigehiro, Tsukasa et al. | Efficient Drug Delivery of Paclitaxel Glycoside: A Novel Solubility Gradient Encapsulation into Liposomes Coupled with Immunoliposomes Preparation | PLOS ONE | 2014 | ISSN 1932--6203 |
| Senese, S. et al. | Chemical dissection of the cell cycle: Probes for cell biology and anti-cancer drug development | Cell Death and Disease | 2014 | ISSN 2041-4889 |
| Bio, Moses et al. | Far-red light activatable, multifunctional prodrug for fluorescence optical imaging and combinational treatment | Journal of Medicinal Chemistry | 2014 | ISSN 1520-4804 |
| Mäki-Jouppila, Jenni H.E. et al. | Centmitor-1, a novel acridinyl-acetohydrazide, possesses similar molecular interaction field and antimitotic cellular phenotype as rigosertib, on 01910.Na | Molecular Cancer Therapeutics | 2014 | ISSN 1538-8514 |
| Mei, Mei et al. | A new 2α,5α,10β,14β-tetraacetoxy-4(20),11-taxadiene (SIA) derivative overcomes paclitaxel resistance by inhibiting MAPK signaling and increasing paclitaxel accumulation in breast cancer cells | PLoS ONE | 2014 | ISSN 1932-6203 |
| Bio, Moses et al. | Site-specific and far-red-light-activatable prodrug of combretastatin A-4 using photo-unclick chemistry | Journal of Medicinal Chemistry | 2013 | ISSN 0022-2623 |
| Zhang, Qiu et al. | Anti-tumor selectivity of a novel Tubulin and HSP90 dual-targeting inhibitor in non-small cell lung cancer models | Biochemical Pharmacology | 2013 | ISSN 1873-2968 |
| Sidhaye, Venkataramana K. et al. | A Novel Role for Aquaporin-5 in Enhancing Microtubule Organization and Stability | PLOS ONE | 2012 | ISSN 1932--6203 |
| Zach, Frank et al. | The retinitis pigmentosa 28 protein FAM161A is a novel ciliary protein involved in intermolecular protein interaction and microtubule association | Human molecular genetics | 2012 | ISSN 1460--2083 |
| Fitzgerald, Daniel P. et al. | TPI-287, a new taxane family member, reduces the brain metastatic colonization of breast cancer cells | Molecular Cancer Therapeutics | 2012 | ISSN 1535-7163 |
| Nguyen, Tam Luong et al. | Evading Pgp activity in drug-resistant cancer cells: A structural and functional study of antitubulin furan metotica compounds | Molecular Cancer Therapeutics | 2012 | ISSN 1535-7163 |
| Dyrager, Christine et al. | Inhibitors and promoters of tubulin polymerization: synthesis and biological evaluation of chalcones and related dienones as potential anticancer agents | Bioorganic & medicinal chemistry | 2011 | ISSN 1464--3391 |
| Hwang, Ji Hwan et al. | Induction of tubulin polymerization and apoptosis in malignant mesothelioma cells by a new compound JBIR-23 | Cancer letters | 2011 | ISSN 1872--7980 |
| Kim, Insook et al. | Zinc stimulates tau S214 phosphorylation by the activation of Raf/mitogen-activated protein kinase-kinase/extracellular signal-regulated kinase pathway | NeuroReport | 2011 | ISSN 0959-4965 |
| Kawaratani, Yasuyuki et al. | New microtubule polymerization inhibitors comprising a nitrooxymethylphenyl group | Bioorganic and Medicinal Chemistry | 2011 | ISSN 0968-0896 |
| Oussenko, Irina A. et al. | Effect of on 01910.Na, an anticancer mitotic inhibitor, on cell-cycle progression correlates with RanGAP1 hyperphosphorylation | Cancer Research | 2011 | ISSN 0008-5472 |
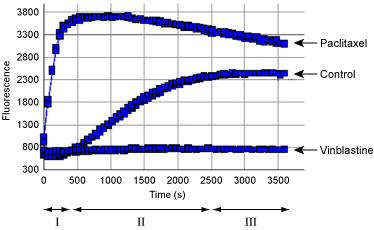
 For our Tubulin Polymerization (Fluorescence) Excel Template please download here.
For our Tubulin Polymerization (Fluorescence) Excel Template please download here. For our IC50 from Vmax Polymerization Excel Template please download here.
For our IC50 from Vmax Polymerization Excel Template please download here.



