Flipper-TR Kit for fluorescence cell membrane microscopy
Flipper-TR® probe is a fluorogenic probe that works by specifically targeting the plasma membrane of cells and reports membrane tension changes through its fluorescence lifetime output. It is the most advanced member of the Flipper probes family (see Citation tab with Refs. 1,2,3, and 4). Flipper-TR® spontaneously inserts into the plasma membrane of cells and is only fluorescent when inserted into a lipid membrane in contrast to aqueous solutions where it has very little fluorescence i.e. it is fluorogenic. The probe senses changes in the organization of lipid bilayer membranes, in particular the tension between lipid layers, through the twist angle and polarization between two twisted dithienothiophenes contained in its structure. (see adjacent figure).
Flipper-TR® use requires Fluorescence Liftime Imaging Microscopy (FLIM) which is a standard technique in modern microscopic set-ups, for more details see FAQ tab.
For Flipper-TR® datasheet click here.
Optical properties
λabs 488 nm
λem 600 nm
εmax 1.6x104 mol-1·cm-1
For more detailed information see the About tab.
Flipper-TR is a registered trademark of UNIGEM, Switzerland. Cytoskeleton, Inc. is the exclusive provider of Spirochrome, Ltd. products in North America and non-exclusive Worldwide distributor.
Schematic representation of the mechanism of action of Flipper-TR tension reporting properties
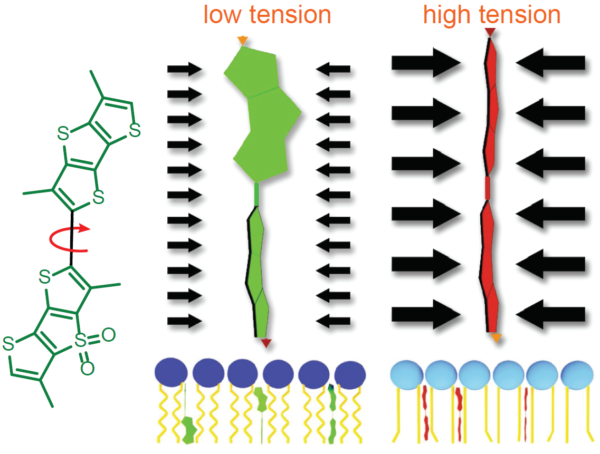
Figure legend: On the left; schematic image of the Flipper-TR molecule. In the middle a low tension lipid bilayer with Flipper twisted, on the right the probe is planar.
Flipper-TR®: A Revolutionary New Fluorescent Probe for Measuring Membrane Tension in Cells and Tissues
Flipper-TR® is a live cell fluorescent membrane tension probe which breaks down the technical barriers hitherto circumvented by technical feats known only to biophysicists with custom equipment. The Flipper-TR membrane tension probe simplifies the methodology by using standard fluorescence lifetime measurements (see Flipper-TR FAQ for practical details). Here we describe the background of Flipper-TR in more detail.
Lipid membranes are dynamic, fluid structures (~4 nm thick) which is a biological necessity as they must change shape and tension for a cell to engage in basic cellular and subcellular physiological functions such as migration, cell spreading, phagocytosis, cell division, endocytosis, mechano-transduction, and metabolism, to name but a few. Consequently, membrane tension is under constant regulation due to its required dynamicity, and in turn, membrane tension regulates cell growth, development, motility, endocytosis, and metabolism. As the membrane is remodeled during these cellular processes, bending, tearing, and stretching of the membrane is common. These changes in membrane shape and tension occur over time and in different locations around and inside the cell and are important parameters to measure in order to understand how membrane tension is regulated and how it regulates these various basic, essential cellular processes. Understanding how membrane tension regulates cellular physiology is relevant in the study of healthy and diseased cells.
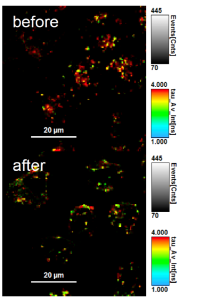
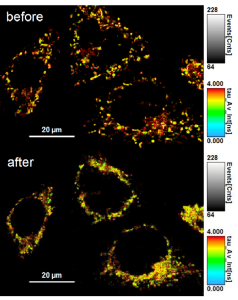
Membrane tension measurements usually relied on low resolution and slow physical methods to determine forces and tension within the plasma membrane. For example, a standard technique for measuring membrane tension involves pulling on membrane tubes from the plasma membrane with a bead trapped in an optical tweezer – a technique fraught with several methodological and technical limitations. For these reasons, novel, sensitive, reliable, and non-invasive research tools capable of rapidly measuring in vivo changes in membrane tension in real-time are in great demand. The fluorescent membrane tension probe Flipper-TR® (Spirochrome, Ltd.) answers these challenges as it has achieved unparalleled membrane tension sensitivity and temporal resolution through the use of FLIM (fluorescence lifetime imaging microscopy) to visualize Flipper-TR® staining of membranes in living cells. Flipper-TR® is a live cell fluorescent membrane tension probe and the first fluorescent membrane tension reporter developed for the field of mechanobiology. The fluorescence lifetime of Flipper-TR® is strongly dependent on the membrane tension. Using FLIM, the precise measurement of the spatio-temporal distribution of tension in membranes is now possible. Flipper-TR® opens up a whole new field by providing a sensitive, reliable, and non-invasive means by which rapid, real-time membrane tension in live cells is measured (Figure 1).
Figure 1 - Flipper-TR sensing membrane tension in cell undergoing hyperosmotic shock.
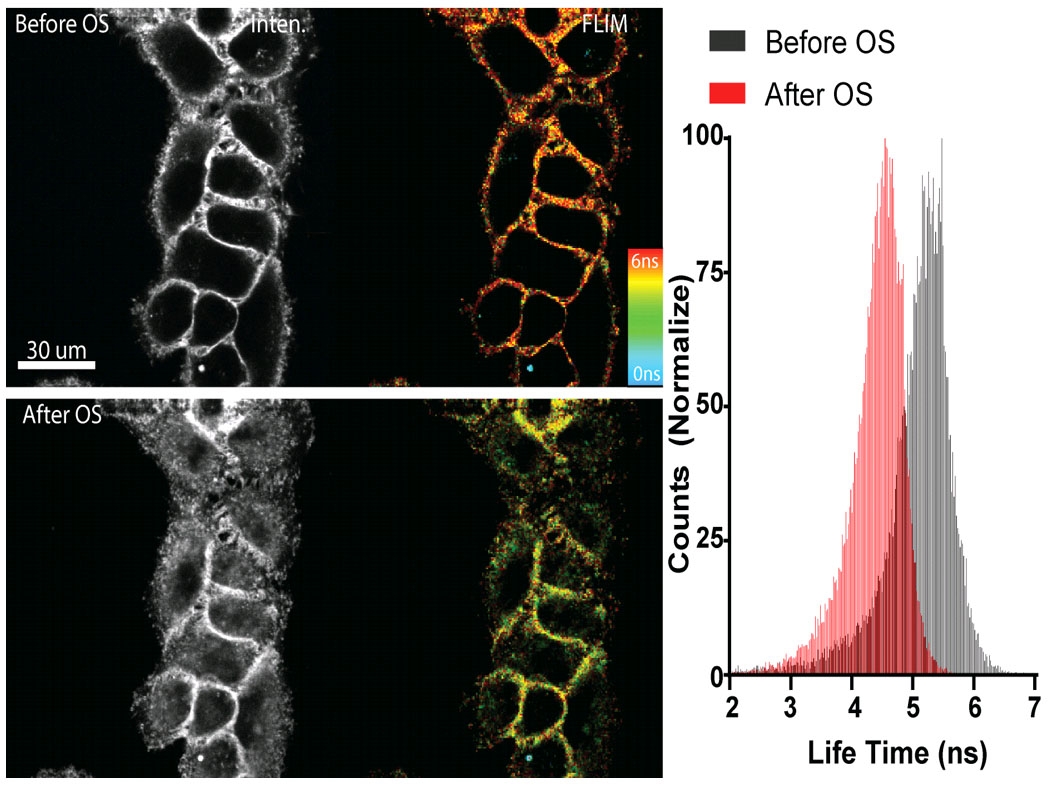
Legend Figure 1: Flipper-TR® staining of cells. Left side: Image of cells stained with Flipper-TR®, greyscale represents fluorescence intensity, and color codes represent fluorescence lifetime. On the right is a histogram of fluorescence emission lifetime versus photon count for both per-osmotic shock (grey) and post-osmotic shock (red). Images courtesy of A. Colom from the Roux group UNIGEM (Ref. 1). Flipper-TR is a registered trademark of UNIGEM, Switzerland.
The fluorescent Flipper-TR® probe works by specifically targeting the plasma membrane of cells and reports membrane tension changes through its fluorescence lifetime changes. It is the most advanced member of the Flipper probes family2,3, which sense changes in the organization of lipid bilayer membranes through changes of the twist angle and polarization between the two twisted dithienothiophenes of the mechanophore. Flipper-TR® spontaneously inserts into the plasma membrane of cells and is only fluorescent when inserted into a lipid membrane. It has a broad absorption and emission spectrum; excitation can be commonly performed with a 488 nm laser, while emission is collected between 575 and 625 nm (Figure 2).
Figure 2 - Absorbance and emission spectra of Flipper-TR

Legend Figure 2 - A; Schematic diagram of the mechanism of Flipper-TR and its interaction with membranes with different tension. On the left, basic molecular structure of Flipper-TR. On the right, low tension green twisted Flipper-TR and on the right high tension planar structure. B;. Flipper-TR solution in ethylacetate was subject to absorbance and emission scans and the results plotted on the same graph. Absorbance is dotted orange line, and emission is a solid orange line.
Click Here to learn more about Flipper-TR® fluorescent cell membrane tension probes.
FAQs of the revolutionary Flipper-TR® probe
Q1. What is FLIM microscopy and how does it work for Flipper-TR?
A1. FLIM microscopy stands for Fluorescent Lifetime Imaging Microscopy. The importance for membrane tension studies is that prior to Flipper-TR membrane tension measurements were very labor and equipment intensive, but now relatively straightforward adaptation of your current microscope will enable highly sensitive tension measurements. Nowadays it is a very standard technique with equipment available from many suppliers, it is based on recording the time that emission takes after excitation of the fluorophore, which is usually very rapid, on the order of 1-10 nano-seconds. FLIM can also be combined with other high resolution microscopic techniques such as Total Internal Reflection Fluorescence (TIRF) or Stimulated Emission Depletion (STED) microscopy for high spatial resolution. FLIM microscopy requires time resolved light detectors which many scientific microscope vendors have available, for example PicoQuant's upgrade kit (https://www.picoquant.com/news/item/picoquants-flim-fcs-upgrade-kit-now-supports-zeiss-lsm780-and-leica-sp2). Reference 1 describes more details about the experimental setup for FLIM microscopy. (Flipper-TR is a registered trademark of UNIGEM, Switzerland)
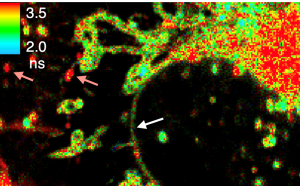
Figure 1 - Diagram of FLIM set-up and time resolved color coded cell image.
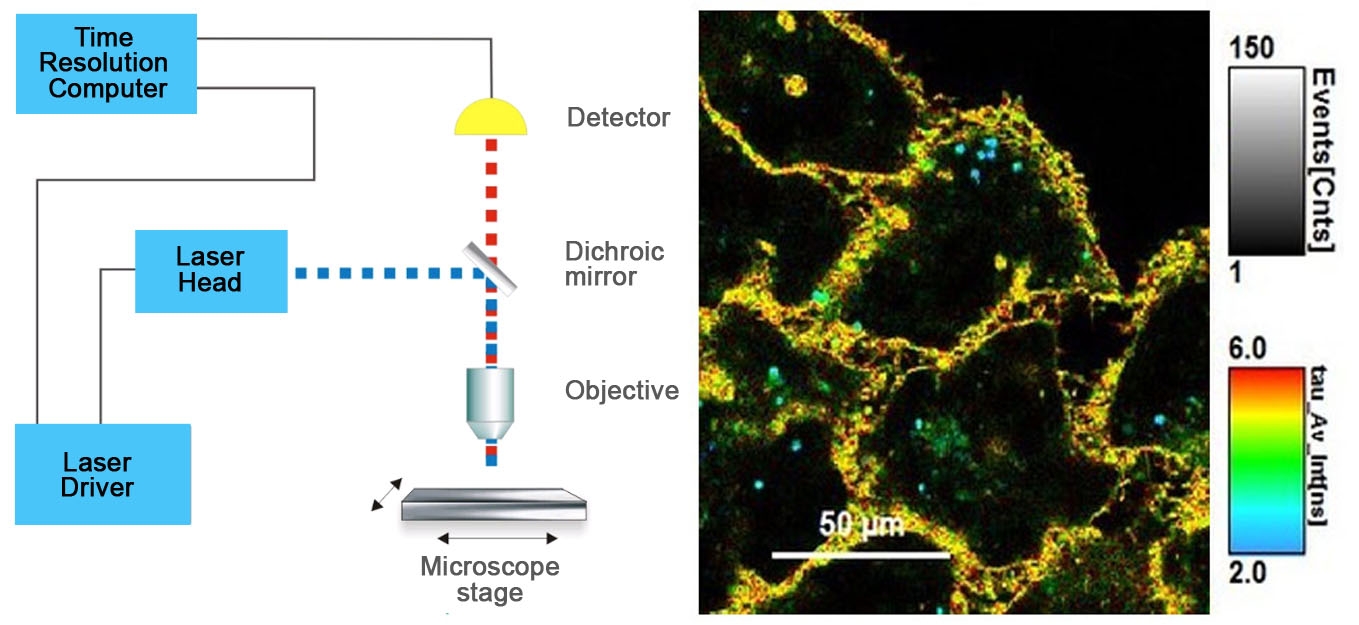
Figure 1. On the left, standard FLIM microscope layout arranged in simple units that are readily available from microscope vendors. On the right, image of cells stained with Flipper-TR®, greyscale represents fluorescence intensity, and color codes represent fluorescence lifetime. Images courtesy of Colom et al. 2018 (Ref. 1). Flipper-TR is a registered trademark of Spirochrome SA, Switzerland.
Q2. Does the Flipper-TR probe work to change fluorescence lifetime?
A2. The fluorescent Flipper-TR® probe works by specifically targeting the plasma membrane of cells and reports membrane tension changes through its fluorescence lifetime changes. It is the most advanced member of the Flipper probes family (Ref. 2,3,4,5). Flipper-TR® spontaneously inserts into the plasma membrane of cells and is only fluorescent when inserted into a lipid membrane. The probe senses changes in the organization of lipid bilayer membranes through the twist angle and polarization between two twisted dithienothiophenes of the mechanophore (see Fig. 2). The emission lifetime is short (2-4 ns) when in the tense state (dithienothiophenes aligned), and longer in the relaxed state (4.1-8.0 ns, dithienothiophenes twisted). Variance (cv) is in the order of 0.3 ns (cv = 4-15%) which allows high resolution of subtle changes in membrane tension. Typically the shorter lifetimes are color coded green, medium lifetimes coded yellow, and longer lifetimes are orange and red (see Fig. 2).
Figure 2 - Schematic diagram of the structure and mechanism of tension reporting by Flipper-TR

Legend Figure 2. Schematic diagram of the mechanism of Flipper-TR and its interaction with membranes with different tension. On the left, basic molecular structure of Flipper-TR. On the right, low tension green twisted Flipper-TR and on the right high tension planar structure.
Q3. What are the filter sets for these probes?
A3. The Flipper-TR probe is visualized with a long separation filter set because its excitation peak is more than 100 nm shorter than the emission peak. Thus the ideal filter set is an excitation of 488 +/- 20 nm and an emission of 575 to 675 +/- 40 nm (Fig. 3). The time resolved measurement method allows very low bacground, which is additive with its low fluorescence in aqueous envirnments, see Q4 below.
Figure 3 - Absortion and emission spectra of Flipper-TR
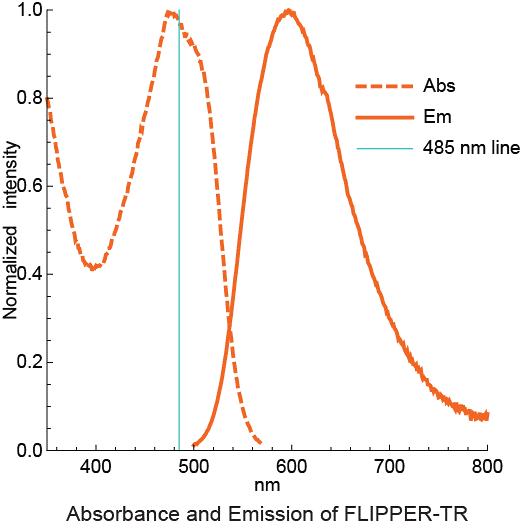
Legend Figure 3 - Flipper-TR solution in ethylacetate was subject to absornace and emission scans and the results plotted on the same graph. Absorbance is dotted orange line, and emission is a solid orange line.
Q4. Why does the Flipper-TR probe have a low background compared to other plasma-membrane probes?
A4. The Flipper-TR probes has very low background in aquence enviroments e.g. tissue culture media or fixative buffer, because it is a fully twisted state and tends to form micelles which quenches itself (Ref.3). After insertion into the membrane it becomes less twisted and starts to emit with high fluorescence (see Fig. 2).
Q5: Is the Flipper-TR probe stable at room temperature?
A5: Yes, the probe is stable in the powder form at room temperature for a few days. After reconstitution in anhydrous DMSO (do not use old-pre-opened bottles of DMSO, but do use ampoules of dry DMSO from Sigma or Spectrum Chemicals. It is stable to freezing and thawing at –20°C, but it is not recommended to divide into small aliquots for storage because it will degrade under these conditions.
Q6: Is the Flipper-TR toxic to cells?
A6: No, under the conditions given in the datasheet the probe is not toxic. Cells will be viable and fluorescent for 2-4 days depending on cell type and culture condition.
Q7: Which organisms and tissues are stained by the Flipper-TR probe?
A7: Currently all known organism have been stained with Flipper-TR, these include tissue culture cells, tissue section (fresh), mammalian cells, insect cells, plant cells, yeast and bacteria.
Q8. Does the Flipper-TR probe work in 3D cell cultures?
A8: Yes, the probe is able to stain cells in a 3D growth environment.
Q9: What is the quantum yield and extinction coefficient in the membrane?
A9: Quantum yield = 0.30 in ethylacetate.
References
1. FLIM microscopy: Lakowicz JR et al. 1994. Emerging biomedical and advanced applications of time-resolved fluorescence spectroscopy. J Fluoresc. 4(1):117-36. doi: 10.1007/BF01876666.
2. Riggi M et al. 2018. Decrease in plasma membrane tension triggers PtdIns(4,5)P2 phase separation to inactivate TORC2. Nat. Cell Biol. 20, 1043–1051.
3. Colom A et al. 2018. A fluorescent membrane tension probe. Nat. Chem. 10, 1118–1125.
4. Dal Molin M. et al. 2015. Fluorescent flippers for mechanosensitive membrane probes. J. Am. Chem. Soc. 137, 568-571.
5. Soleimanpour S. et al. 2016. Headgroup engineering in mechanosensitive membrane probes. Chem. Commun. (Camb). 52, 14450-14453.