* Limited stock available. If stock is not available, Cytoskeleton will produce a new batch upon request. Minimum order will apply. Inquire for more information.
Product Uses
- Measurement of Microtubule-activated ATPase assays
- Identification/characterization of proteins or small molecules that affect motor ATPase activity
- Identification/characterization of proteins or small molecules that affect motor/microtubule interactions.
Material
The conserved motor domain of human MKLP2 (KIF20A) was expressed in a prokaryotic system. The recombinant protein contains a His-Tag at the amino terminal end and has a combined molecular weight of 54 kDa. The protein has been determined to be biologically active in a microtubule-activated ATPase activity test (see below). The protein is supplied as a lyophilized powder.
| MP05 Size | Minimum amt. per tube | Actual amt. per tube | Vol. of buffer for 5 mg/ml resuspension | Minimum* ATPase (Vmax) (nmol/min/mg) |
| CS-MP05-A | 50 ug | ~65 ug | 10 ul | 600 |
Storage and Reconstitution
The lyophilized protein is stable for at least 1 year when stored at 4°C with a desiccant (humidity <10%). Alternatively, the lyophilized protein can be stored at -70°C and is stable at least 1 year. The protein should be reconstituted to 5 mg/ml with ice cold Milli-Q water. See above table for resuspension volumes. The protein can be aliquoted into experiment sized tubes and snap frozen in liquid nitrogen. When reconstituted and stored at -70°C, the protein will be stable for at least 4 months. NOTE: Kinesins do not respond well to repeated freeze /thaws and for storage at -70°C the protein concentration should not be less than 5 mg / ml. Kinesin diluted below 5 mg/ml should not be re-frozen as it will lose activity.
Purity
Protein purity is estimated by scanning densitometry of a coomassie-stained SDS-PAGE gradient gel. Figure 1 shows 5 μg of CS-MP05 protein and purity was determined to be >70%. The total protein in each tube will therefore be approximately 30% greater than the amount shown on the tube. The major contaminants are at approximately 51 & 70 kD. The microtubule-activated ATPase activity of the MKLP2 motor is not affected by these contaminants.
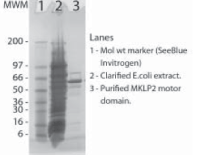
Figure 1. MKLP2 Motor Domain protein gel. A 5 μg sample of recombinant MKLP2 Motor Domain protein was separated on a 4-20% SDS-PAGE gradient gel, along with See Blue molecular weight markers (Invitrogen). The fusion protein runs at 54 kD on the polyacrylamide gradient gel. Protein quantitation was determined using Advanced Protein assay (cat.# ADV02).
Reagents
1) Kinesin ELIPA Assay Kit (cat.# BK060)
Equipment
1) Monochromatic spectrophotometer (set to 360 nm) or a filter based spectrophotometer with a 360 nm filter and bandwidth of <10 nm.
MICROTUBULE ACTIVATED ATPase ASSAY
MKLP2 ATPase activity was measured by monitoring real time free phosphate generation using the Kinesin ELIPA Assay Kit (cat.# BK060). The assay is based upon an absorbance shift (330 nm - 360 nm) that occurs when 2-amino-6-mercapto-7-methylpurine ribonucleoside (MESG) is catalytically converted to 2-amino-6-mercapto-7-methylpurine in the presence of inorganic phosphate (Pi). One molecule of Pi will yield one molecule of 2-amino-6-mercapto- 7-methylpurine in an essentially irreversible reaction. Hence, the absorbance at 360 nm is directly proportional to the amount of Pi generated in the kinesin ATPase reaction. Under the conditions outlined below, the Vmax for MKLP2 microtubule-activated ATPase activity for this Lot was 1330 nmoles ATP generated per minute per mg of CS-MP05 (Figure 2). This value is above the guaranteed minimum.
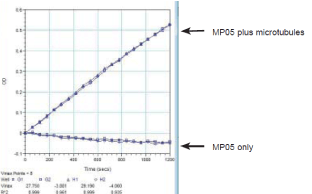
Figure 2. CS-MP05 microtubule-activated ATPase activity using Kinesin ELIPA Assay Kit (cat.# BK060).
Method (ELIPA ATPase assay)
The reactions were conducted in 1/2 area 96 well plates (100 ul reaction volumes). Each reaction contains 0.24 μg of MKLP2 protein (CS-MP05), 5 μM taxol stabilized Microtubules (cat# MT002), 0.2 mM MESG, 0.3U PNP, 20 μM taxol, 12 mM PIPES pH 7, 8 mM MgCl2, 1 mM ATP. Control reactions were carried out in the absence of MTs. These reactions gave readings of <0.1. Reactions were measured in a SpectraMax M2 (Molecular Devices) set in kinetic mode and 360nm absorbance wavelength. Readings were taken at room temperature once every 30 seconds for a total reaction time of 20 minutes. The nmoles of ATP generated in a given time was determined by the use of a phosphate standard curve (not shown).
For Reagents and Equipment see above.
References
1. Hackney, D and Jiang, W. (2001) Methods in Molecular Biology (Humana Press) 164:65-71
2. Kuriyama et al. 1994. J. Cell Science 107:3485-3499.
3. Nislow et al. 1992. Nature 359:543-547.
For product Datasheets and MSDSs please click on the PDF links below.
If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com
Coming soon! If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com
