Rac3, a Rho GTPase and regulator of actin-based cytoskeletal dynamics
Ras-related C3 botulinum toxin substrate 3 (Rac3) is a 21 kDa GTPase encoded by 192 amino acids that acts as a molecular switch in a variety of cellular events that include cell motility, development, signaling, transport, cytoskeletal organization, cell cycle, reactive oxygen species production, and vesicle trafficking. Along with the Rho isoforms and Cdc42, it is a member of the Rho sub-family of Ras super-family GTPases and is one of 3 Rac isoforms (Rac1 and Rac2 being the others). At the nucleotide level, human Rac3 has 77% identity with Rac1, 83% identity with Rac2, and 69% identity with RhoG. At the amino acid level, Rac3 and Rac1 are 92% identical and Rac3 and Rac2 are 89% identical. Like all Ras super-family GTPases, Rac3 is bound to GTP in its active form, whereas it is inactive in its GDP-bound form. Rac3 is activated by guanidine activating proteins (GEFs) that exchange bound GDP for GTP and in-activated by GTPase-activating proteins (GAPs) that promote GTP hydrolysis. Because of the hydrophobic isoprenyl moiety at the C-terminus, Rac3 is associated with endo-membranes and cell membranes. Nuclear localization has also been reported. In the cytoplasm it associates with the chaperone Rho GDP-dissociation inhibitor (RhoGDI).
Rac3 mRNA is present in a variety of human cancer cell lines and tumor tissue samples including brain, liver, kidney, pancreas, breast, and prostate. Active Rac3 has been detected in MDA-435, T47D, and MCF7 breast cancer cell lines and treatment of cells with siRNA against Rac3 inhibits SNB19 glioblastoma and BT549 breast cancer cell line invasion in an in vitro assay. Conversely, expression of a constitutively active Rac3 isoform in MDA-MB-435 breast cancer cells resulted in increased invasion and motility in vitro.
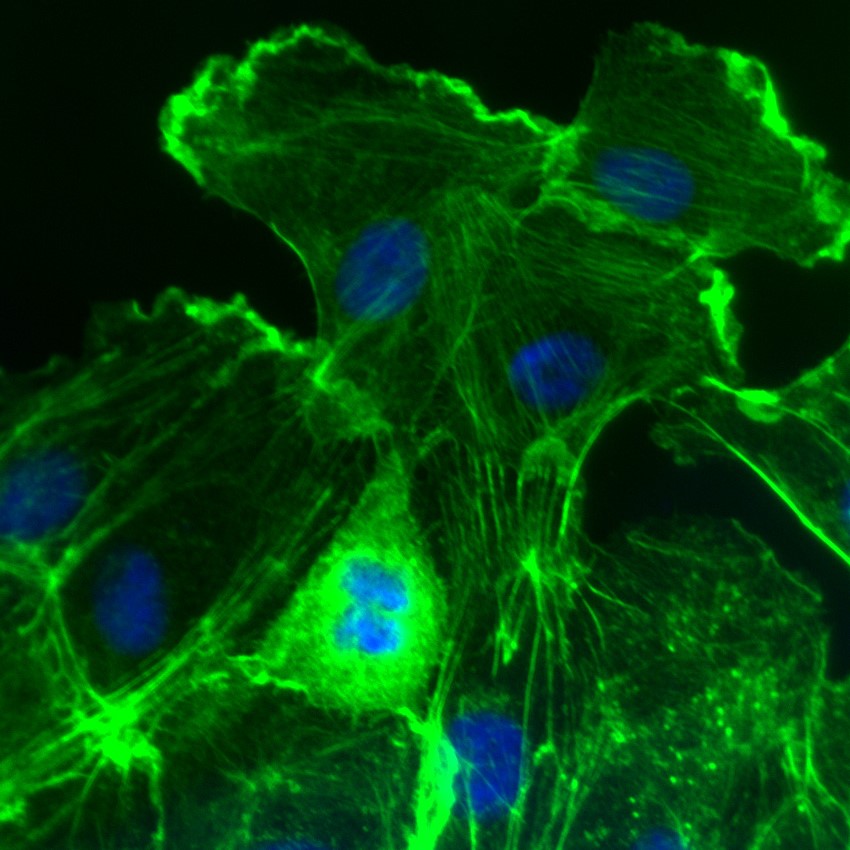
Rac activation in Swiss 3T3 cells. F-actin is visualized with fluorescent green phalloidin staining (Cat.# PHDG1) and nuclear blue DNA staining with Dapi. Cells were activated with Cat. # CN04 (right).
For more information on Rac3, please see here:
Hwang S.L. et al. 2005. Expression of Rac3 in human brain tumors. J. Clin. Neurosci. 12, 571-574.
Related Products
Kits
Rac1 G-LISA Activation Assay Kit (Colorimetric Based) 96 assays (Cat. # BK128)
Rac1 G-LISA Activation Assay (Luminescence format) - 96 assays (Cat. # BK126)
Proteins
Rac1 protein: His tagged: human wild type (Cat. # RC01)
Rac2 protein: His tagged: human wild type (Cat. # RC02)
Tiam1 MBP-DHPH domain: human recombinant (1040– 1406) (Cat. # CS-TIAM1)
Antibodies
Anti-Rac1: mouse Mab (Cat. # ARC03)
Live Cell Imaging Probes
Spirochrome SiR-Actin Kit (Cat. # CY-SC001)
Acti-Stain Phalloidins
Rhodamine Phalloidin (Cat. # PHDR1)
Acti-stain 488 phalloidin (Cat. # PHDG1)
Acti-stain 555 phalloidin (Cat. # PHDH1)
Acti-stain 670 phalloidin (Cat. # PHDN1)
G-Switch Activators and Inhibitors
Rac/Cdc42 Activator II (Cat. # CN02)
