Rap1b protein: His tagged: human wild type
Product Uses Include
- Rap1b biochemistry
- Rap1b GTPase assays
- Rap1b nucleotide exchange assays
- Rap1b binding studies
Material
The human Rap1b protein has been produced in a bacterial expression system. The protein is supplied as a lyophilized powder. When it is reconstituted in distilled water to 1 mg/ml, the protein is in the following buffer: 2 mM Tris pH 7.6, 0.5 mM MgCl2, 0.5% sucrose, 0.1% dextran. Protein concentration is determined by the Precision Red Advanced Protein Assay Reagent, Cat. # ADV02.
The recombinant protein is approximately 26 kDa, consisting of the Rap1b protein plus a linker and a 6X His tag in the amino-terminus.
For many other purified small G-proteins, see our main small G-protein product page.
Purity
Purity is determined by scanning densitometry of proteins on SDS-PAGE gels. Rap1b samples are >90% pure (Figure 1).
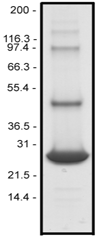
Figure 1: Rap1b His-tagged protein purity determination. A 10 µg sample of RR02 (molecular weight approx. 26 kDa) was separated by electrophoresis in a 12% SDS-PAGE system. The protein was stained with Coomassie Blue.
Biological Activity
The biological activity of Rap1b is determined from its ability to exchange GDP for GTP. EDTA is used to sequester magensium ions from the Rap1b protein, thereby stimulating nucleotide exchange activity. The RhoGEF exchange assay Biochem Kit™ (Cat. # BK100) is used to monitor the exchange (Figure 2).
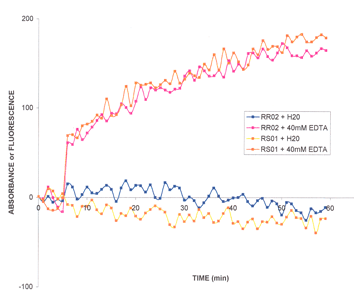
Figure 2: Rap1b GTP exchange assay using BK100. Rap1b protein (1 µM) was mixed with exchange buffer and aliquoted to four wells of a 96-well half area plate. After 5 cycles of reading in a fluorimeter, EDTA was added to 40 mM in the test samples and Milli-Q water to the control samples. Reactions were monitored for 30 min.
For product Datasheets and MSDSs please click on the PDF links below. For additional information, click on the FAQs tab above or contact our Technical Support department at tservice@cytoskeleton.com
Coming soon! If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com





