G-LISA Technical Tips
Click here for kit-specific technical notes
G-LISA® Activation Assays: General Technical Notes
Growth and Treatment of Cell Lines
The health and responsiveness of your cell line is the single most important parameter for the success and reproducibility of the small G-protein (GTPase) activation assays. The next most important parameters are time course of activation and titration of the activating factor (or time course of inhibition and titration of inhibiting factor). Time should be taken to read this section and to carefully maintain cell lines in accordance with the guidelines given below.
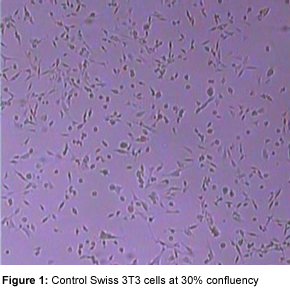
The optimal confluency of adherent cells will depend on which G-LISA activation assay you are using and which cell lines are being utilized. Optimal conditions will need to be determined empirically and based on reports in the literature. For example, adherent Swiss 3T3 cells are seeded at 5 x 105 per ml and grown for 3 days. Non-adherent cells should be ready at approximately 3 x 105 cells per ml. In our experiences with fibroblast cells (Swiss 3T3), 30-50% confluency is optimal for serum starvation or other treatments (see Figure 1). With human umbilical vein endothelial cells (HUVECs), the cells should be used when fully confluent.
When possible, the untreated samples should have cellular levels of GTPase activity in a “controlled state”. For example, when looking for GTPase activation, the “controlled state” cells could be serum-starved. Serum starvation will inactivate cellular GTPases and lead to a much greater response to a given activator. For inhibitor studies, you can omit the serum starvation step unless you are first activating the cells.
Cells should also be checked for their responsiveness (“responsive state”) to a known stimulus. In many cases, poor culturing technique can result in essentially non-responsive cells. An example of poor culturing technique includes the sub-culture of cells that have previously been allowed to become overgrown. In general, cells grown to >70-80% confluency should not be used for GTPase activation studies.
To confirm the “controlled state” and “responsive state” of your cells, it is a good idea to include a small coverslip in your experimental tissue culture vessels and analyze the “controlled state” cells vs. the “responsive state” cells by staining of actin filaments with fluorescently-labeled phalloidin. See Figures 2 through 4 for changes in actin filaments in control and activated cells stained with Acti-stain 488 phalloidin (Cat. # PHDG1).
Serum-starved and Rho-activated HeLa Cells.
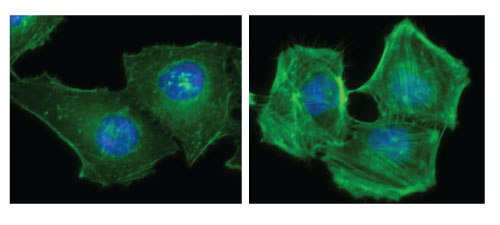
Figure 2: HeLa cells grown in DMEM plus 10% fetal bovine serum were serum-starved (left panel) for 24 h prior to treatment with a Rho activator (right panel) for either 10 min (Cat. # CN01) or 120 min (Cat. # CN03). Similar results were obtained with each activator. Cells were fixed and stained with Acti-stain 488 and DAPI as described in the Acti-stain protocols. Observe the limited number of F-actin filaments (green) in the cytoplasm of serum-starved cells compared to activated cells.
Serum-starved and Rho-activated Vero Cells.
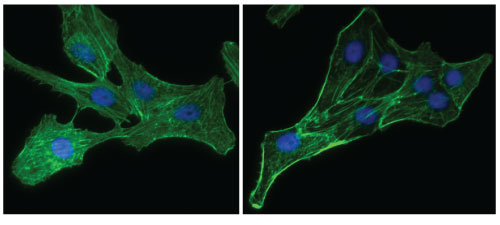
Figure 3: Vero cells grown in DMEM plus 10% fetal bovine serum were serum-starved (left panel) for 24 h prior to treatment with a Rho activator (right panel) for either 10 min (Cat. # CN01) or 120 min (Cat. # CN03). Similar results were obtained with each activator. Cells were fixed and stained with Acti-stain 488 and DAPI as described in the Acti-stain protocols. Observe the differential distribution of F-actin filaments (green) in the cytoplasm of serum-starved cells compared to activated cells.
Serum-starved and Rac-activated HUVEC Cells.
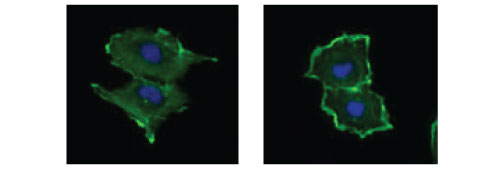
Figure 4: HUVEC cells grown in DMEM plus 15% fetal bovine serum were serum-starved (left panel) for 24 h prior to treatment with a Rac activator (right panel) for 2 min with Cat. # CN02. Cells were fixed and stained with Acti-stain 488 and DAPI as described in the Acti-stain protocols. Observe the F-actin filament staining (green) which translocates to the cell periphery (membrane ruffles) in activated cells.
If you are having difficulty determining a “controlled state” for your experiment then contact technical assistance at 303-322-2254 or e-mail tservice@cytoskeleton.com.
Timing and Titration of Activator
Upon stimulation, GTPases are generally activated very rapidly and transiently. Maximal activation ranges from 30 s to 30 min and declines thereafter to basal levels. For potent activators, the intensity of maximal GTPase activation over “control state” (serum-starved) cells is generally in the order of 1.5 to 3.0 fold. However, using a single time point you are more likely to miss this maximum activation peak. It is therefore critical to take timed samples for at least the first experiment with an unknown activating entity. Recommended time points are 0, 1.5, 3, 6, 10 and 30 min. (A time course is also recommended for GTPase inactivation studies). The concentration-response curve for activators or inhibitors should also be determined for the time point that provides optimal activation or inhibition, respectively.
In practical terms the timed experiment should be performed sequentially one time point at a time. This allows rapid processing of each single time point. Once one time point lysate is collected, it should be snap-frozen in “experiment sized” aliquots immediately and kept at -70°C. We recommend preparing and immediately snap-freezing duplicate samples per time-point or condition. For a list of commonly used GTPase activators please click here.
Assay Preparation For G-LISA®
It is critical to get the assay components ready before preparing cell lysates or thawing previously prepared lysates, as a defrosted activated lysate is unstable and should be assayed as soon as possible after preparation. Please see the specific kit instructions for additional details on reagent preparation.
Reagent | Preparation |
GTPase-GTP binding 96-well plate |
|
Lysis Buffer | Either pipette 10 ml of Lysis buffer into 15 ml Falcon tube and add 100 µl of protease inhibitors (cat# PIC02) into it, mix and place in ice, or,
|
Control Protein |
|
Rapid processing of cells
GTP bound (active) GTPases are labile entities as the bound GTP is susceptible to hydrolysis by GAPs during and after cell lysis, resulting in GTPase inactivation. Also, GEF and GAP activities in extracts contribute to a change in the actual state of activation prior to lysis. Rapid processing at 4°C is essential for accurate and reproducible results. Some experiments may benefit from not washing cells, e.g., serum free grown cells, because the cells are disturbed by the washing process; however for serum grown cells the washing is essential because the extracts are measured for protein content and equated for concentration prior to assay. Please note that PBS should not be used with the Cdc42 G-LISA because the protein is activated by the washing. The following guidelines are useful for rapid washing of cells.
Washing
-
Retrieve culture dish from incubator and immediately aspirate out all of the media.
-
Immediately rinse cells with an appropriate volume of ICE-COLD PBS (except for Cdc42 G-LISA) to remove serum proteins (see Table below for recommended wash volumes) and place the plate on ice.
-
Aspirate off all residual PBS buffer. This is essential so that the Lysis Buffer is not diluted. Correct aspiration requires that the culture dish be placed at an angle on ice for 10 to 30 s to allow excess PBS to collect in the vessel for complete removal. Do not leave on ice too long because the cells will respond to this temperature change.
-
In the case where no washing is performed, adding the appropriate amount of ice cold lysis buffer while placing the plate on ice ensures lysis and cold are applied together.
Cell Lysis
To avoid making too dilute or too concentrated lysate samples, it is recommended to adjust the amount of Lysis Buffer depending on your cell type and plate type. The following table gives guidelines for suitable lysis volumes for 3T3 cells which tend to give low protein yields. The exact lysis volumes for any given cell line will have to be determined empirically.
Recommended Wash and Lysis Volumes for 3T3 Cell Cultures
| Culture Vessel | Vessel surface area (cm2) | Volume of PBS wash (ml) | Volume of Lysis Buffer (µl) |
35 mm dish | 8 | 2.0 | 100 |
60 mm dish | 21 | 3.0 | 250 |
100 mm dish | 56 | 10.0 | 600 |
150 mm dish | 148 | 15.0 | 2000 |
6-well cluster plate | 9.5 / well | 3.0 | 100 |
12-well cluster plate | 4 / well | 1.5 | 60 |
T-25 Flask | 25 | 4.0 | 250 |
T-75 Flask | 75 | 10.0 | 1200 |
T-150 Flask | 150 | 15.0 | 2000 |
Cell Lysis, continued
The time period between cell lysis and addition of lysates to the wells is critically important. Take the following precautions:
Work quickly.
Keep solutions and lysates embedded in ice so that the temperature is below 4°C. This helps to minimize changes in signal over time. The Assay Protocol gives very specific instructions regarding temperature and must be strictly adhered to for successful results.
We strongly recommend cell lysates should be immediately frozen after harvest and clarification. A sample of at least 20 µl should be kept on ice for protein concentration measurement. The lysates must be snap frozen in liquid nitrogen and stored at -70°C. Lysates can be stored at -70°C for up to 3 months.
Thawing of cell lysates prior to use in the G-LISA assay should be in a room temperature water bath until 30% ice is left, followed by rapid transfer to ice and immediate use in the assay. Do not leave the extracts in the water bath to thaw completely. The remaining frozen lysate will thaw quickly once transferred to ice.
Protein Concentration:
Equal protein concentration in all samples is a prerequisite for accurate comparison between samples in G-LISA activation assays. Cell extracts should be equalized with ice cold buffer plus protease inhibitor cocktail. The recommended protein concentration range depends on the GTPase being studied. The optimal concentration will depend on the relative amounts of the GTPase in the cells/tissues which will be need to be determined empirically by titrating lysate concentrations or by a quantitative Western blot with a known GTPase protein standard. See the Assay Protocol section of each kit's manual for a simple equation to calculate the appropriate dilution volume for each sample.
The Precision Red™ Advanced Protein Assay Reagent (Part # GL50; can be purchased separately as Cat. # ADV02) is included with the kit in order to measure protein concentration with a rapid one-step procedure. It is ideal for this analysis because it is detergent compatible and it is rapid and simple to perform. The protein assay can be performed in a 1 ml cuvette format as described in the Assay Protocol section. Alternatively, a 96-well format can be used where 10 μl of sample is pipetted into a well followed by the addition of 300 μl of Protein Assay Reagent. In the latter case, the absorbance reading at 600 nm is multiplied by 3.75 to obtain the protein concentration in mg/ml. See the Assay Protocol section of the kit's manual for more details.
Assay Linearity
Assay linearity depends on which GTPase activation assay kit is being used. A positive control protein is included with each kit and should be used as a guideline for determining if experimental OD readings are out of the linear range of the assay. In such cases you should reduce the amount of total cell protein per assay. The lower level of detection of the assay is approximately 20% above the background reading of Lysis Buffer only. Readings lower than this require increased cell protein per assay.
Use of a Multi-channel Pipettor
When processing more than 16 wells it is imperative to use a multi-channel or multi-dispensing pipettor with a range of 25 to 200 μl per dispense. Critical steps such as lysate addition, post-binding wash step and the Antigen Presenting Buffer step all have requirements for accurate and timely additions. Attempting to perform >16 assays with a single channel pipettor will also increase the likelihood of allowing wells to dry out before reagent addition can be completed, resulting in variable signals. Therefore, use a multi-channel or at least a multi-dispensing pipettor wherever possible. If neither of these pipettor options is available we highly recommend that you limit each experiment to a maximum of 16 wells.
Removal of Solutions from Wells
Removal of solutions from the wells is accomplished by turning the plate upside down and flicking out the well contents into a waste bin. This is followed by patting the plate several times on a paper towel to get rid of residual solution. It has been found that the complete removal of solutions from the well requires a vigorous flick of the plate and a vigorous series of pats onto paper towels (5-7 hard pats). The complete removal of solution from wells between steps of the G-LISA assay is very important as it avoids high background readings in the buffer only wells. The buffer only wells should read between 0.10 – 0.35 at an absorbance of 490 nm. If background readings are higher than 0.10 – 0.35 then a more vigorous removal of solutions from the well should be practiced.
Plate Shaker Recommendations
It is essential to use an orbital plate shaker at 400 rpm. Rocking or tilting plate shakers will not be sufficient for this assay. As a back-up you can use a 200 rpm orbital shaking culture incubator or a normal orbital rotating platform. Signals will be lower with the 200 rpm option. Here are a few suppliers with suitable orbital plate shakers:
Model # 4625 Titer Plate Shaker, Lab-Line Instruments, Barnstead Intl.
(average price)
Model # RF7854 Digital Microplate Shaker, ML Market Lab, researchml.com
(economical price)
Model # RF7855 Incubating Microplate Shaker, ML Market Lab, researchml.com
(deluxe model)
Plate Reader Settings
| Parameters | Character | Comments |
Wavelength | 490 nm | Bandwidth 20 nm or less |
Shake | Orbital, normal rate | 5 s |
Temperature | Room temperature | 24°C is also fine. |
Protocol Type | End point |
|
Vortex of Samples after Binding Buffer Addition
Binding buffer is a viscous solution and requires thorough mixing after addition to cell lysates. The best way to do this is a brief 3-5 second vortex on high. This step will greatly reduce variability between duplicate samples and will increase overall assay accuracy.
Please inquire to Technical assistance for help in setting up G-LISA assays (call 303-322-2254 for immediate help, or e-mail tservice@cytoskeleton.com for assistance within 24 h).
G-LISA Activation Assays: Kit-Specific Technical Notes
Click on the product below to see kit-specific technical notes:
RhoA G-LISA (Cat. # BK121)
RhoA G-LISA (Cat. # BK124)
Rac1,2,3 G-LISA (Cat. # BK125)
Rac1 G-LISA (Cat. # BK126)
Cdc42 G-LISA (Cat. # BK127)
Rac1 G-LISA (Cat. # BK128)
RalA G-LISA (Cat. # BK129)
Growth & Treatment of Cell Lines | Adherent cells should be ready for serum starvation or treatment at 30-50% confluency or for non-adherent cells at approximately 3 x 105 cells per ml. Briefly, adherent cells are seeded at 5 x 104 per ml and grown for 3 days. It has been found that cells cultured for several days (3 days) prior to treatment are significantly more responsive than cells that have been cultured for a shorter period of time. |
Activators/Inhibitors: Timing and Titration | Typical Rho activators are calpeptin (Cat. # CN01) and LPA. Upon stimulation, Rho proteins are generally activated very rapidly and transiently with maximal activation ranging from 30 s to 30 min and declining thereafter to basal levels. For potent activators such as calpeptin or LPA, the intensity of maximal Rho activation over “control state” (serum-starved) cells is generally in the order of 2-5 fold. However, you are more likely to miss this maximal activation using a single time point and the signal will likely be 2 fold activation. It is therefore critical to take timed samples for at least the first experiment with an unknown activating entity. Recommended time points are 0, 1, 3, 6, 12 and 30 min (the time course is also recommended for Rho inactivation studies). In addition, activators (and inhibitors) should be titrated for maximal effects at the optimal time point. Rho activation causes the formation of characteristic stress fibers. For a list of commonly used GTPase activators please click here. |
Protein Concentration Range | This kit uses 25 µl of lysate (0.4-2 mg/ml lysate protein concentration) per assay. We recommend 0.5 mg/ml total protein as a good starting point. However, as different cell lines or tissues will have different amounts of active Rho protein, we highly recommend titrating lysates. |
Assay Linearity | The assay is linear to 1 ng of bound activated RhoA. The positive control protein is at 1 ng; therefore, any assay readings that are greater than the positive control will be out of the linear range of the assay. In such cases, you should reduce the amount of total cell protein per assay. The lower level of detection of the assay is approximately 20% above the background reading of Lysis Buffer only. |
Rapid Processing of Cells | GTP bound (active) Rho is a labile entity and the bound GTP is susceptible to hydrolysis during and after cell lysis, resulting in Rho inactivation. Rapid processing at 4°C is essential for accurate and reproducible results. We highly recommend that the final concentration of equalized lysates lies between 0.4–2 mg/ml. However, due to the difference of Rho-GTP levels in different cell lines, we highly recommend you titrate your lysate. |
Luminometer Settings | Luminometers vary widely in their sensitivity and absolute readings. It is therefore recommended to run a G-LISA assay with blank and positive control to confirm that you are in the linear range of the assay. When in the linear range of the assay the positive control should read 3-5 fold higher than the blank wells. |
Growth & Treatment of Cell Lines | Adherent cells should be ready for serum starvation or treatment at 30-50% confluency or for non-adherent cells at approximately 3 x 105 cells per ml. Briefly, adherent cells are seeded at 5 x 104 cells per ml and grown for 3 days. It has been found that cells cultured for several days (3 days) prior to treatment are significantly more responsive than cells that have been cultured for a shorter period of time. |
Activators/Inhibitors: Timing and Titration | Typical Rho activators are calpeptin (Cat. # CN01) and LPA. Upon stimulation, Rho proteins are generally activated very rapidly and transiently with maximal activation ranging from 30 s to 30 min and declining thereafter to basal levels. For potent activators such as calpeptin or LPA, the intensity of maximal Rho activation over “control state” (serum-starved) cells is generally in the order of 2-5 fold. However, you are more likely to miss this maximal activation using a single time point and the signal will likely be 2 fold activation. It is therefore critical to take timed samples for at least the first experiment with an unknown activating entity. Recommended time points are 0, 1, 3, 6, 12 and 30 min (the time course is also recommended for Rho inactivation studies). In addition, activators (and inhibitors) should be titrated for maximal effects at the optimal time point. Rho activation causes the formation of characteristic stress fibers. For a list of commonly used GTPase activators please click here. |
Protein Concentration Range | This kit uses 25 µl of lysate (0.4-2 mg/ml lysate protein concentration) per assay. We recommend 0.5 mg/ml total protein as a good starting point. However, as different cell lines or tissues will have different amounts of active Rho protein, we highly recommend titrating lysates. |
Assay Linearity | The assay is linear between 0.05 - 2 ng of bound activated RhoA. The positive control protein is at 1 ng; therefore, any assay readings that are two fold or more above the positive control will be out of the linear range of the assay. In such cases, you should reduce the amount of total cell protein per assay. The lower level of detection of the assay is approximately 20% above the background reading of Lysis Buffer only. Readings lower than this require increased cell protein per assay. |
Rapid Processing of Cells | GTP bound (active) Rho is a labile entity and the bound GTP is susceptible to hydrolysis during and after cell lysis, resulting in Rho inactivation. Rapid processing at 4°C is essential for accurate and reproducible results. We highly recommend that the final concentration of equalized lysates lies between 0.4–2 mg/ml. However, due to the difference of Rho-GTP levels in different cell lines, we highly recommend you titrate your lysate. |
Luminometer Settings | Not applicable |
Growth & Treatment of Cell Lines | Adherent cells should be ready for serum starvation or treatment at 30-50% confluency or for non-adherent cells at approximately 3 x 105 cells per ml. Briefly, adherent cells are seeded at 5 x 104 per ml and grown for 3 days. It has been found that cells cultured for several days (3 days) prior to treatment are significantly more responsive than cells that have been cultured for a shorter period of time. |
Activators/Inhibitors: Timing and Titration | Typical Rac activators are EGF (Cat. # CN02) or PDGF. Upon stimulation, Rac proteins are generally activated very rapidly and transiently. Maximal activation ranges from 30 s to 30 min and declines thereafter to basal levels. For potent activators such as EGF or PDGF, the intensity of maximal Rac activation over “control state” (serum-starved) cells is generally in the order of 2-5 fold. However, using a single time point, you are more likely to miss this maximum activation peak. It is therefore critical to take timed samples for at least the first experiment with an unknown activating entity. Recommended time points are 0, 1, 3, 6, 12 and 30 min (the time course is also recommended for Rac inactivation studies). In addition, activators (and inhibitors) should be titrated for maximal effects at the optimal time point. Rac activation causes the formation of characteristic membrane ruffles. For a list of commonly used GTPase activators please click here. |
Protein Concentration Range | The G-LISA kit uses 25 ul of lysate (0.2 – 0.5 mg/ml lysate protein concentration) per assay. However, as different cell lines or tissues will have different amounts of active Rac protein, we highly recommend titrating lysates. |
Assay Linearity | The assay is linear from 10 pg to 1 ng of bound activated Rac. The positive control protein is at 0.4 ng; therefore, any assay readings that are more than 2.5 fold higher than the positive control will be out of the linear range of the assay. In such cases you should reduce the amount of total cell protein per assay. The lower level of detection of the assay is approximately 20% above the background reading of Lysis Buffer only. Readings lower than this require increased cell protein per assay. |
Rapid Processing of Cells | GTP bound (active) Rac1 is a labile entity and the bound GTP is susceptible to hydrolysis by Rac-GAPs during and after cell lysis resulting in Rac1 inactivation. It is reported that the Rac1 hydrolysis rate is faster than that of RhoA. We highly recommend that the final concentration of equalized lysates is between 0.2-0.5 mg/ml. However, due to different Rac-GTP levels across cell lines, we highly recommend you titrate your lysate. |
Luminometer Settings | Not applicable |
Growth & Treatment of Cell Lines | Adherent cells should be ready for serum starvation or treatment at 30-50% confluency or for non-adherent cells at approximately 3 x 105 cells per ml. Briefly, adherent cells are seeded at 5 x 104 per ml and grown for 3 days. It has been found that cells cultured for several days (3 days) prior to treatment are significantly more responsive than cells that have been cultured for a shorter period of time. |
Activators/Inhibitors: Timing and Titration | Typical Rac activators are EGF or PDGF. Upon stimulation, Rac proteins are generally activated very rapidly and transiently. Maximal activation ranges from 30 s to 30 min and declines thereafter to basal levels. For potent activators such as EGF or PDGF, the intensity of maximal Rac activation over “control state” (serum-starved) cells is generally in the order of 2-5 fold. However, using a single time point, you are more likely to miss this maximum activation peak. It is therefore critical to take timed samples for at least the first experiment with an unknown activating entity. Recommended time points are 0, 1, 3, 6, 12 and 30 min (the time course is also recommended for Rac inactivation studies). In addition, activators (and inhibitors) should be titrated for maximal effects at the optimal time point. Rac activation causes the formation of characteristic membrane ruffles. For a list of commonly used GTPase activators please click here. |
Protein Concentration Range | The G-LISA kit uses 25 ul of lysate (0.25-2 mg/ml lysate protein concentration ) per assay. However, as different cell lines or tissues will have different amounts of active Rac protein, we highly recommend titrating lysates. |
Assay Linearity | The assay is linear from 10 pg to 2 ng of bound activated Rac1. The positive control protein is at 2 ng, therefore any assay readings that are more than the positive control will be out of the linear range of the assay. In such cases you should reduce the amount of total cell protein per assay. The lower level of detection of the assay is approximately 20% above the background reading of Lysis Buffer only. Readings lower than this require increased cell protein per assay. |
Rapid Processing of Cells | GTP bound (active) Rac1 is a labile entity and the bound GTP is susceptible to hydrolysis by Rac-GAPs during and after cell lysis resulting in Rac1 inactivation. It is reported that the Rac1 hydrolysis rate is faster than that of RhoA. We highly recommend that the final concentration of equalized lysates is between 0.2-0.5 mg/ml. However, due to different Rac1-GTP levels across cell lines, we highly recommend you titrate your lysate. |
Luminometer Settings | Luminometers vary widely in their sensitivity and absolute readings. It is therefore recommended to run a G-LISA assay with blank and positive control to confirm that you are in the linear range of the assay. When in the linear range of the assay the positive control should read 3-5 fold higher than the blank wells. |
Growth & Treatment of Cell Lines | Adherent cells should be ready for serum starvation or treatment when they are 30-50% confluent. Non-adherent cells should be ready at approximately 3 x 105 cells per ml. Briefly, adherent cells are seeded at 5 x 104 per ml and grown for 3 days. It has been found that cells cultured for several days (3 days) prior to treatment are significantly more responsive than cells that have been cultured for a shorter period of time. |
Activators/Inhibitors: Timing and Titration | Typical Cdc42 activators are EGF, TNF or bradykinin. Upon stimulation, Cdc42 proteins are generally activated very rapidly and transiently. Maximal activation ranges from 30 s to 30 min and declines thereafter to basal levels. For potent activators such as EGF or Bradykinin, the intensity of maximal Cdc42 activation over “control state” (serum-starved) cells is generally in the order of 1.5 to 3.0 fold. However, using a single time point, you are more likely to miss this maximum activation peak. It is therefore critical to take timed samples for at least the first experiment with an unknown activating entity. Recommended time points are 0, 1.5, 3, 6, 10 and 30 min (the time course is also recommended for Cdc42 inactivation studies). In addition, activators (and inhibitors) should be titrated for maximal effects at the optimal time point. Cdc42 activation causes the formation of characteristic filopodia and microspikes. For a list of commonly used GTPase activators please click here. |
Protein Concentration Range | This kit uses 50 µl of lysate (0.15-1.00 mg/ml lysate protein concentration) per assay. We recommend 0.25 mg/ml total protein as a good starting point. However, as different cell lines or tissues will have different amounts of active Cdc42 protein, we highly recommend titrating lysates. |
Assay Linearity | The assay is linear from 0.5 ng to 10 ng of bound activated Cdc42. The positive control protein is at 2 ng; therefore, if any OD readings are more than 5 times this sample, they will be out of the linear range of the assay. In such cases, you should reduce the amount of total cell protein per assay. The lower level of detection of the assay is approximately 20% above the background reading of Lysis Buffer only. Readings lower than this require increased cell protein per assay. |
Rapid Processing of Cells | GTP bound (active) Cdc42 is a labile entity and the bound GTP is susceptible to hydrolysis by Cdc42-GAPs during and after cell lysis resulting in Cdc42 inactivation. It is reported that the Cdc42 hydrolysis rate is faster than that of RhoA which indicates that extract processing time and conditions for Cdc42 activity are even more critical than for Rac and Rho. Total protein concentration should be 0.15-1.0 mg/ml for Cdc42 G-LISA activation assays. We recommend 0.25 mg/ml total protein as a good starting point. However, due to different Cdc42-GTP levels across cell lines, we highly recommend you titrate your lysate. |
Luminometer Settings | Not applicable |
Growth & Treatment of Cell Lines | Adherent cells should be ready for serum starvation or treatment when they are 30-50% confluent. Non-adherent cells should be ready at approximately 3 x 105 cells per ml. Briefly, adherent cells are seeded at 5 x 104 per ml and grown for 3 days. It has been found that cells cultured for several days (3 days) prior to treatment are significantly more responsive than cells that have been cultured for a shorter period of time. |
Activators/Inhibitors: Timing and Titration | Typical Rac activators are EGF or PDGF. Upon stimulation, Rac proteins are generally activated very rapidly and transiently. Maximal activation ranges from 30 s to 30 min and declines thereafter to basal levels. For potent activators such as EGF or PDGF, the intensity of maximal Rac activation over “control state” (serum-starved) cells is generally in the order of 2-5 fold. However, using a single time point, you are more likely to miss this maximum activation peak. It is therefore critical to take timed samples for at least the first experiment with an unknown activating entity. Recommended time points are 0, 1, 3, 6, 12 and 30 min (the time course is also recommended for Rac inactivation studies). In addition, activators (and inhibitors) should be titrated for maximal effects at the optimal time point. Rac activation causes the formation of characteristic membrane ruffles. For a list of commonly used GTPase activators please click here. |
Protein Concentration Range | This kit uses 50 µl of lysate (0.25-1.0 mg/ml lysate protein concentration) per assay. However, as different cell lines or tissues will have different amounts of active Rac protein, we highly recommend titrating lysates. |
Assay Linearity | The assay is linear from 1 ng to 8 ng of bound activated Rac1. The positive control protein is at 2 ng; therefore, any assay readings that are 4 fold greater than the positive control will be out of the linear range of the assay. In such cases, you should reduce the amount of total cell protein per assay. The lower level of detection of the assay is approximately 20% above the background reading of Lysis Buffer only. Readings lower than this will require increased cell protein per assay. |
Rapid Processing of Cells | GTP bound (active) Rac1 is a labile entity and the bound GTP is susceptible to hydrolysis by Rac-GAPs during and after cell lysis, resulting in Rac1 inactivation. It is reported that the Rac1 hydrolysis rate is faster than that of RhoA. We highly recommend that the final concentration of equalized lysates is between 0.25–1 mg/ml. However, due to different Rac1-GTP levels across cell lines, we highly recommend you titrate your lysate. |
Luminometer Settings | Not applicable |
Growth & Treatment of Cell Lines | Adherent cells should be ready for serum starvation or treatment when they are 30-50% confluent. Non-adherent cells should be ready at approximately 3 x 105 cells per ml. Briefly, adherent cells are seeded at 5 x 104 per ml and grown for 3 days. It has been found that cells cultured for several days (3 days) prior to treatment are significantly more responsive than cells that have been cultured for a shorter period of time. |
Activators/Inhibitors: Timing and Titration | Typical Ral activators are EGF, LPA or ionomycin. Upon stimulation, Ral proteins are generally activated very rapidly and transiently. Maximal activation ranges from 30 s to 30 min and declines thereafter to basal levels. For potent activators such as EGF, LPA or ionomycin, the intensity of maximal Ral activation over “control state” (serum-starved) cells is generally in the order of 2-5 fold. However, using a single time point, you are more likely to miss this maximum activation peak. It is therefore critical to take timed samples for at least the first experiment with an unknown activating entity. Recommended time points are 0, 1, 3, 6, 12 and 30 min (the time course is also recommended for Ral inactivation studies). In addition, activators (and inhibitors) should be titrated for maximal effects at the optimal time point. |
Protein Concentration Range | This kit uses 25 µl of lysate (0.25-1 mg/ml lysate protein concentration) per assay. We recommend 0.5 mg/ml total protein as a good starting point. However, as different cell lines or tissues will have different amounts of active Ral protein, we highly recommend titrating lysates. |
Assay Linearity | The assay is linear from 0.5 ng to 5 ng of bound activated RalA. The positive control protein is at 2.5 ng; therefore, if any OD readings are more than 2 times this sample, they will be out of the linear range of the assay. In such cases, you should reduce the amount of total cell protein per assay. The lower level of detection of the assay is approximately 20% above the background reading of Lysis Buffer only. Readings lower than this will require increased cell protein per assay. |
Rapid Processing of Cells | The GTP bound (active) Ral is a labile entity and the bound GTP is susceptible to hydrolysis by Ral-GAPs during and after cell lysis resulting in Ral inactivation. Also GEF and GAP activities in extracts contribute to a change in the actual state of activation prior to lysis. Rapid processing at 4°C is essential for accurate and reproducible results. We recommend 0.5 mg/ml total protein as a good starting point. However, due to the difference of Ral-GTP levels in different cell lines, we highly recommend you titrate your lysate. |
Luminometer Settings | Not applicable |
G-LISA is a registered trademark of Cytoskeleton, Inc (CO). All rights reserved.
