Tubulin protein (97% pure): porcine brain
Product Uses Include
- Economical alternative to T240 in primary screens for tubulin ligand drugs
- High-throughput tubulin polymerization or depolymerization screens
- Production of microtubule substrates for HTS motor assays
Material
Tubulin protein has been isolated from porcine brain. The final product contains approximately 97% tubulin and 3% Microtubule Associated Proteins (MAPs). This version of porcine tubulin is an economical alternative to our highly purified tubulin (Cat. # T240) for anti-tubulin ligand drug discovery. It has been formulated to be compatible with 96 or 384-well format polymerization assays such as in Biochem™ kits BK004P or BK011P .
Purity
Protein purity is determined by scanning densitometry of Coomassie Blue stained protein on a 12% SDS-PAGE gel. HTS02 contains 97% tubulin (MW. 55 kDa) and 3% MAPs (MW 35-280 kDa).
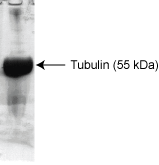
Figure 1: A 100 µg sample of HTS03 protein was separated by electrophoresis on a 12% SDS-PAGE gel and stained with Coomassie Blue. Protein quantitation was performed using the Precision Red Protein Assay Reagent (Cat.# ADV02).
Biological Activity
An ASSAY UNIT is defined as the amount of HTS03 protein needed to achieve a tubulin polymerization signal of OD340 of 0.1 - 0.15 in 30 minutes at 37°C in G-PEM buffer. The assay volume is 100 µl and assumes a spectrophotometer pathlength of 0.5 cm. The protein concentration for HTS02 in this assay is approximately 4 mg/ml. The biological activity of HTS03 is assessed by a tubulin polymerization assay. One ASSAY UNIT of tubulin is used for each polymeriztion assay. An OD340 of 0.10 - 0.15 is required to pass quality control. Tubulin polymerization must also be responsive to polymerization enhancers (paclitaxel) and inhibitors (nocodazole) at 5 µM drug concentration.
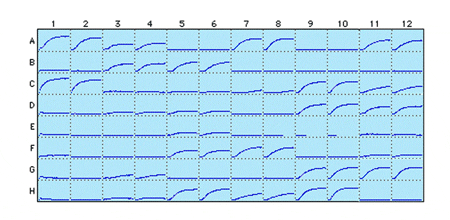
Figure 2: Tubulin polymerization in a 96-well format using HTS03. All samples are in duplicates. Each well has a mini-polymerization curve with 20 min on the x-axis and 0.30 OD 340 nm on the y-axis. The Vmax parameter is used to compare inhibitors, the CV is 13% in this format, so a 50% cut off is required for a 98% confidence.
For product Datasheets and MSDSs please click on the PDF links below. For additional information, click on the FAQs tab above or contact our Technical Support department at tservice@cytoskeleton.com
Question 1: Does tubulin polymerization using 97% pure porcine brain tubulin (Cat. # HTS03) require additional enhancers such as glycerol or taxol?
Answer 1: No additional enhancers are required when using 3 or 4 mg/ml of the MAP-enriched 97% pure tubulin. The recommended polymerization reaction using 97% pure tubulin contains 100 μl of 3 or 4 mg/ml tubulin in 80 mM PIPES pH 6.9, 0.5 mM EGTA, 2 mM MgCl2 and 1 mM GTP. Polymerization is started by incubation at 37°C and followed by absorption readings at 340 nm. Under these conditions, polymerization will reach a maximal OD340 between 0.15 – 0.25 within 30 minutes. In this experimental set up (100 μl volume in a spectrophotometer with a pathlength of 0.5 cm), an OD340 of 0.1 is approximately equal to 1 mg per ml of tubulin polymer mass. Under these conditions only 40% of the tubulin is polymerized, offering the flexibity to study how polymerization enhancers (e.g., taxol) or inhibitors (e.g., nocodazole) alter tubulin polymerization. For enhancers, we recommend using 3 mg/ml tubulin whereas for inhibitors, 4 mg/ml tubulin works better.
Question 2: Upon resuspension as directed, what are the components of the final buffer?
Answer 2: Upon reconstitution as directed, the tubulin will be in the following buffer: 80 mM PIPES, 1 mM EGTA, 1 mM MgCl2, pH 7.0, 1 mM GTP, 1% Ficoll, 5% sucrose and <1mM DTT.
If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com




