Product Uses Include
- Screening for fungal tubulin specific drugs
- Examining interactions of proteins with fungal tubulin
Material
Tubulin protein has been isolated from the mushroom, Agaricus bisporus, and purified by anion exchange chromatography. Tubulin consists of one α and one β subunit. Each subunit is 55 kDa in size, as demonstrated by SDS-PAGE analysis. Typically, the molar equivalent of tubulin is defined as the heterodimer, which has a molecular weight of 110 kDa. Tubulin is supplied as a white lyophilized powder in the heterodimeric form.
Purity
Protein purity is determined by scanning densitometry of Coomassie Blue-stained protein from 4-20% SDS-PAGE. F001 is determined to be >90% fungal tubulin

Figure 1: Fungal tubulin purity determination. A 30 µg sample of F001 was run on a 4-20% SDS-PAGE gel and stained with 0.1% coomassie blue.
Biological Activity
The biological activity of F001 is assessed by a tubulin polymerization assay. The polymerization of tubulin into microtubules is detected by measuring the optical density at 340 nm (see Figure 2). Under the experimental conditions defined below, a 100 µl solution of F001 at 1.0 mg/ml in a 1/2 area 96-well plate results in a change in optical density of 0.28-0.30 within 1 h at 30°C.
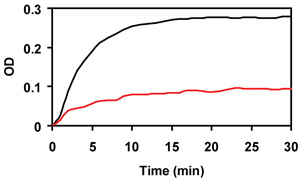
Figure 2: Inhibition of fungal tubulin polymerization with rhizoxin. Fungal tubulin was allowed to polymerize (as measured by an increase in optical density) in the absence (black) or presence (red) of the microtubule destabilizing drug rhizoxin.
For product Datasheets and MSDSs please click on the PDF links below. For additional information, click on the FAQs tab above or contact our Technical Support department at tservice@cytoskeleton.com
Coming soon! For the most recent publications citing this product, please contact our Technical Service department at tservice@cytoskeleton.com
Coming soon! If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com



