Tubulin protein (biotin): porcine brain
Product Uses Include
- Immobilizing tubulin onto a solid surface via streptavidin.
- Creating tubulin or microtubule affinity matrices with streptavidin coated sepharose beads
- High through-put screening using a proximity assay.
- Microinjection into cells followed by electron microscopy of streptavidin conjugated gold particles to determine the cellular localization of the tubulin.
- Nanotechnology
Material
Porcine brain tubulin (>99% pure, see Cat. # T240) has been modified so that random surface lysines contain a covalently linked, long-chain biotin derivative. A long-chain biotin derivative was selected for this procedure because it allows the biotin molecules to be spaced far enough away from the tubulin protein so as not to interfere with its activity, e.g., ligand binding to SPA beads or other streptavidin based reagents. Biotin labeled tubulin is supplied as a lyophilized powder.
Cytoskeleton, Inc. also offers biotinylated cancer cell tubulin (Cat. # H003 )
Purity
The protein purity of the tubulin used for labeling is determined by scanning densitometry of Coomassie Blue stained protein on a 4-20% polyacrylamide gel. The protein used for T333P is >99% pure tubulin. Labeled protein is run on an SDS gel, transfered to a nitrocellulose membrane and detected with streptavidin alkaline phosphatase (Fig 1). 10 ng of T333P is readily detectable. No free biotin is detectable in the final product.
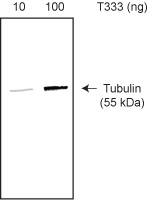
Figure 1: Purity determination of biotin tubulin. 10 and 100 ng of T333P was run on a 4-20% SDS-PAGE gel, transferred to a nitrocellulose membrane and biotin labeled material was detected with streptavidin alkaline phosphatase.
Biological Activity
The biological activity of T333P is assessed by a tubulin polymerization assay. To pass quality control, a 5 mg/ml solution of fluorescein labeled tubulin in G-PEM plus 5% glycerol must polymerize to >85%. This is comparable to unlabeled tubulin under identical conditions.
For product Datasheets and MSDSs please click on the PDF links below. For additional information, click on the FAQs tab above or contact our Technical Support department at tservice@cytoskeleton.com
Question 1: What is the proper way to store the tubulin to insure maximum stability and activity?
Answer 1: The recommended storage condition for the lyophilized tubulin product is 4°C with desiccant to maintain humidity at <10% humidity. Under these conditions the protein is stable for 6 months. Lyophilized protein can also be stored desiccated at -70°C where it will be stable for 6 months. However, at -70°C the rubber seal in the lid of the tube could crack and allow in moisture. Therefore we recommend storing at 4°C. If stored at -70°C, it is imperative to include desiccant with the lyophilized protein if this storage condition is utilized. After reconstituting the protein as directed, the concentrated protein in G-PEM buffer should be aliquoted, snap frozen in liquid nitrogen and stored at -70°C (stable for 6 months). NOTE: It is very important to snap freeze the tubulin in liquid nitrogen as other methods of freezing will result in significantly reduced activity. Defrost rapidly by placing in a room temperature water bath for 1 min. Avoid repeated freeze/thaw cycles.
Question 2: Does the biotinylated tubulin polymerize as well as unlabeled tubulin?
Answer 2: Yes, the biotinylated tubulin (Cat. # T333P) polymerizes as well as unlabeled tubulin (Cat. # 240). The biological activity of biotinylated tubulin is determined from its ability to efficiently polymerize into microtubules in vitro and separate from unpolymerized protein in a spin-down assay. Stringent quality control ensures that >85% of the biotinylated tubulin can polymerize in this assay. This is comparable to the polymerization capacity of unmodified tubulin (Cat. # T240).
Question 3: Why does Cytoskeleton recommend the use of general tubulin buffer and GTP for resuspending tubulin?
Answer 3: We recommend resuspending tubulin in general tubulin buffer + GTP to maintain tubulin monomer protein stability and conformation and to provide the necessary components for polymerization. For resuspension, we recommend using a general tubulin buffer (Cat. # BST01-001) which consists of 80 mM PIPES, 2 mM MgCl2, 1 mM EGTA, pH 7.0, supplemented with 1 mM GTP (Cat. # BST06-001). Tubulin requires GTP and magnesium ions for proper stability and conformation, even in its monomeric state. GTP is also required for the polymerization process as its hydrolysis during tubulin polymerization is necessary for polymerization to occur. EGTA is a chelator of calcium which is a potent inhibitor of tubulin polymerization. Glycerol is often added to a final concentration of 5 - 10% to enhance polymerization; however, glycerol is not necessary for the maintenance of biologically active tubulin and does not need to be included when reconstituting and storing tubulin. When aliquoting reconstituted tubulin for storage, it is essential to aliquot and snap-freeze tubulin in liquid nitrogen at a concentration of >6 mg/ml to preserve tubulin’s biological activity. Then the aliquots should be stored at -70°C. When thawing the aliquots, thaw rapidly in a room temperature water bath and place on ice until right before experimental use.
If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com.











