+3
Loading...
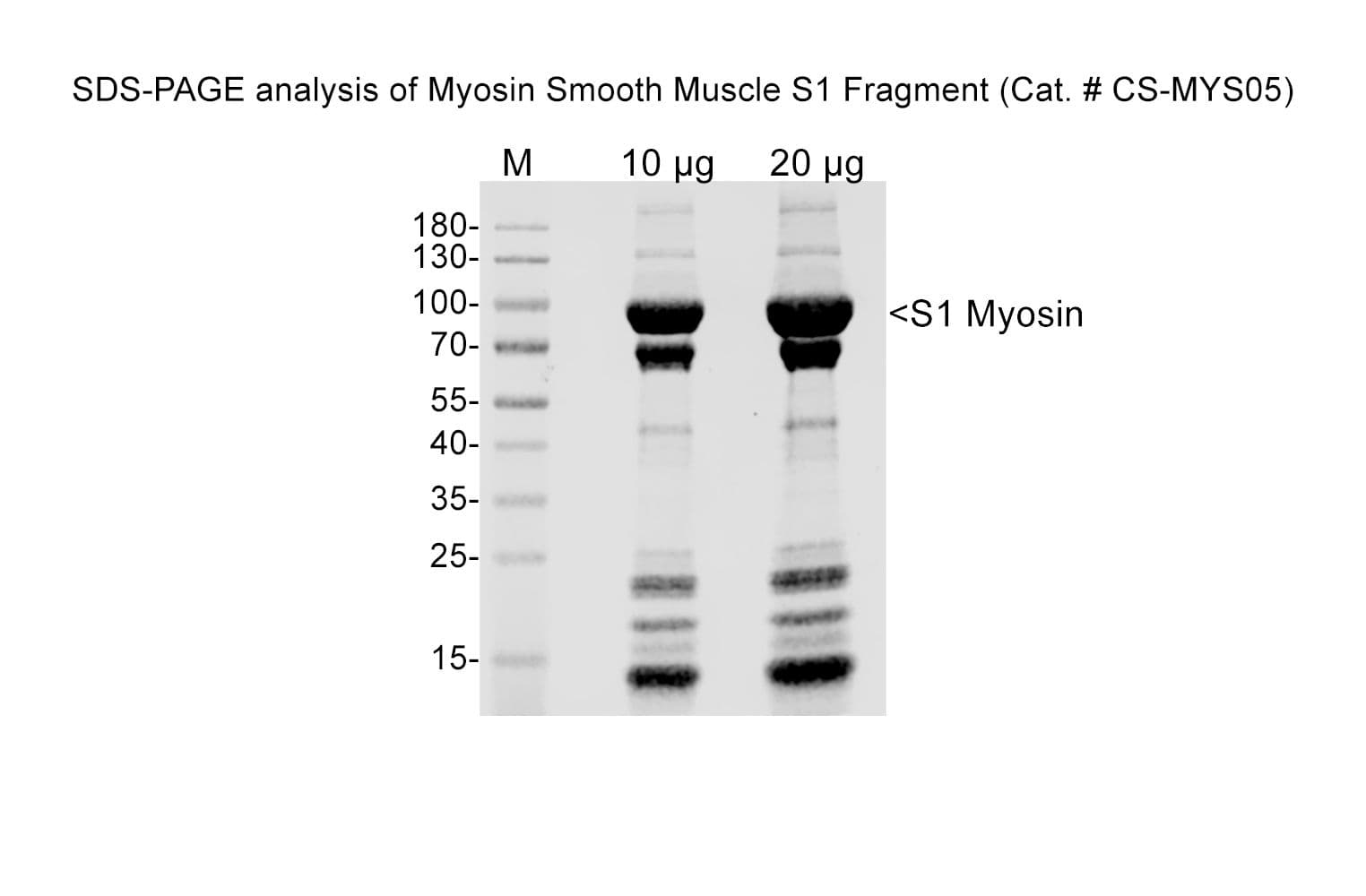
Smooth muscle myosin S1 fragment is prepared by digesting full-length chicken gizzard myosin with -chymotrypsin in the presence of EDTA to liberate the soluble S1 fragment.
Smooth muscle myosin S1 fragment is produced from chicken gizzard myosin that is ≥90% pure
The biological activity of chicken gizzard S1 myosin can be determined from its rate of F-actin-activated ATP hydrolysis. The assay is constructed by first polymerizing actin to form F-actin. Myosin is then added in sub-stoichiometric amounts, and the reaction is initiated with ATP.
Cat. #CS-MYS05