* Limited stock available. If stock is not available, Cytoskeleton will produce a new batch upon request. Minimum order will apply. Inquire for more information.
Product Uses
- Measurement of calcium activated myosin ATPase activity when bound to thin filaments.
- Identification/characterization of proteins or small molecules that affect the TT complex regulation and myosin ATPase activity
- Identification/characterization of proteins or small molecules that affect myosin / F- actin interaction
Materials
Cardiac myosin protein has been purified from bovine heart tissue(1, 2). The full length myosin protein was purified with its essential light chains (ELC) and regulatory light chains (RLC), see Figure 1and 2. Myosin was then digested with alpha-chymotrypsin to liberate the soluble subfragment-1 (S1) domain, which was isolated by centrifugation (3). The purified myosin S1 fragment has been determined to be biologically active in an F-actin activated ATPase assay (see biological activity assay). Bovine cardiac myosin S1 fragment protein is supplied as a white lyophilized powder.
Figure 1. Diagrammatic representation of the myosin protein and its subfragments
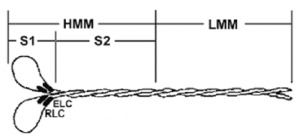
Legend: Myosin is a hexameric protein consisting of two heavy chains and two light chains. Myosin can be proteolytically cleaved into heavy meromyosin (HMM) and light meromyosin (LMM) by α-chymotrypsin in the presence of magnesium. In the presence of EDTA, however, α-chymotrypsin produces the soluble myosin S1 fragment (3).
Storage and Resonstitution
Briefly centrifuge to collect the product at the bottom of the tube. Reconstituting a 1 mg tube of MYS03 with 300 ul of Milli-Q water (3 ml for the 10 mg bottles) will generate a 3.3 mg/ml stock of cardiac S1 myosin in the following buffer: 20 mM PIPES pH 7.0,5% (w/v) sucrose and 1% (w/v) dextran. The protein should not be exposed to repeated freeze-thaw cycles. The lyophilized protein is stable at 4°C desiccated (<10% humidity) for 1 year.
Purity
Protein purity is determined by scanning densitometry of Coomassie Blue stained protein on a 4-20% gradient polyacrylamide gel. The myosin and its light chains used to produce the myosin S1 fragment was determined to be 90% pure (see Figure2).
Figure 2. Full length and S1 myosin

A 20 μg sample of full length bovine cardiac myosin protein (lane A) and the corresponding S1 myosin (lane B) were separated by electrophoresis using a 4-20% SDS-PAGE gel and stained with Coomassie Blue.The arrow indicates the myosin heavy chain (approx. 200 kDa), arrow heads indicate the RLC(approx. 20 kDa) and two ELC isoforms (approx. 25 and 21 kDa).Protein quantitation was performed using the Precision Red™ Protein Assay Reagent (Cat.# ADV02). Mark12 molecular weight markers are from Invitrogen.
Biological Activity Assay
The biological activity of bovine cardiac myosin S1 fragment can be determined from its rate of F-actin activated ATP hydrolysis.The assay is constructed by first polymerizing actin to form Factin,then the cardiac Tropomyosin/Troponin complex (TT complex) is mixed with the actin filaments in a stoichiometric amount. This creates coated filaments which are analogous to the thin filaments of muscle fibers. Myosin is added in substoichiometric amounts and the reaction initiated with ATP and calcium. Stringentquality control ensures that in the absence of calcium the TT complex completely inhibits myosin ATPase. On addition of 10 uMcalcium myosin ATPase will be restored. Calcium binds to Troponin C which dissociates from F-actin allowing myosin to bind.
Reagents
1. Cardiac TT complex (1 x 1 mg, # TT05)
2. Cardiac Myosin S1 (0.25 mg), # MYS03)
3. Cardiac Actin (1 mg, Cat. # AD99-A)
4. ATPase Assay Biochem Kit (Cat. # BK051)
5. 100 mM ATP in 50 mM Tris-HCl pH 7.5 (100ul)
6. 1 M Dithiothreitol in water (100 ul).
7. PM12 Reaction buffer (12 mM Pipes-KOH, pH 7.0, 2 mM MgCl2).8. 500 mM EGTA-Na pH 8.0.
Equipment
1. Spectrophotometer capable of measuring absorbance at 360 nm (+/- 5 nm bandwidth). We recommend a Spectra-Max M2 (Molecular Devices), filter based machines are not suitable.
2. Half area 96 well microtiter plate (Corning Cat.# 3696 or 3697)
3. Multi-channel pipette
Method
The following major steps are recognized:
Step 1. Assemble required reagents and compounds. (30min).
Step 2. Prepare F-actin polymer stock. (1h).
Step 3. Prepare Thin Filament stock (1.5h)
Step 4. Prepare Motor Mix and plate reader. (15min).
Step 5. Pipette Motor Mix into wells and start reaction/plate reader. (10min).
F-actin polymer stock
1. Resuspend AD99 with 2.5ml of Buffer to 0.4mg/ml (measure protein concentration for better reproducibility), in buffer 5mM Pipes-KOH pH 7.0, 500uM ATP, 500uM DTT.
2. Place at RT for 10min to solubilize the actin.
3. Then add 2.0 mM MgCl2 and 2.0 mM EGTA and incubate at RT for 20 min to polymerize. (shelf life 1h at RT).
Thin Filament stock
1. Resuspend 1 x 1mg TT05 on ice with ice cold water to 5 mg/ml. (200ul per vial for 1mg vial).
2. Mix the following to make 1.2 ml of actin/TT05 (TF):
1000 μl of F-actin stock
200 μl of TT05
3. Incubate at RT for 20min.
4. Centrifuge at 100K xg 4°C for 1h.
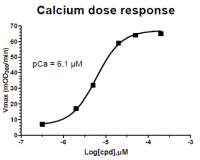
Figure 3 legend: The sarcomere assay was set up as described in the protocol above. Calcium was titrated between 2 and 200 μM and the results plotted on this dose response graph. pCa = 6.1 μM is similar to published pCa values for reconstituted cardiac sarcomeres (Holroyde et al. 1980, Fig.6).
References
1. Pollard, T.D., . 1982. Methods in Cell Biol. 24:333
2. Margossian, S.S., and Lowey, S. 1982. Methods in Enzymology.85:55-71.
3. Weeds, A.G., and Taylor, R.S. 1975. Nature (London) 257:54.
4. M.J. Holroyde et al. 1980. The calcium and magnesium binding sites on cardiac troponin their role in the regulation of myofibrillar adenosine triphosphatase.
For product Datasheets and MSDSs please click on the PDF links below.
Coming soon! For the most recent publications citing this product, please contact our Technical Service department at tservice@cytoskeleton.com
Coming soon! If you have any questions concerning this product, please contact our Technical Service department at tservice@cytoskeleton.com
