+3
Loading...
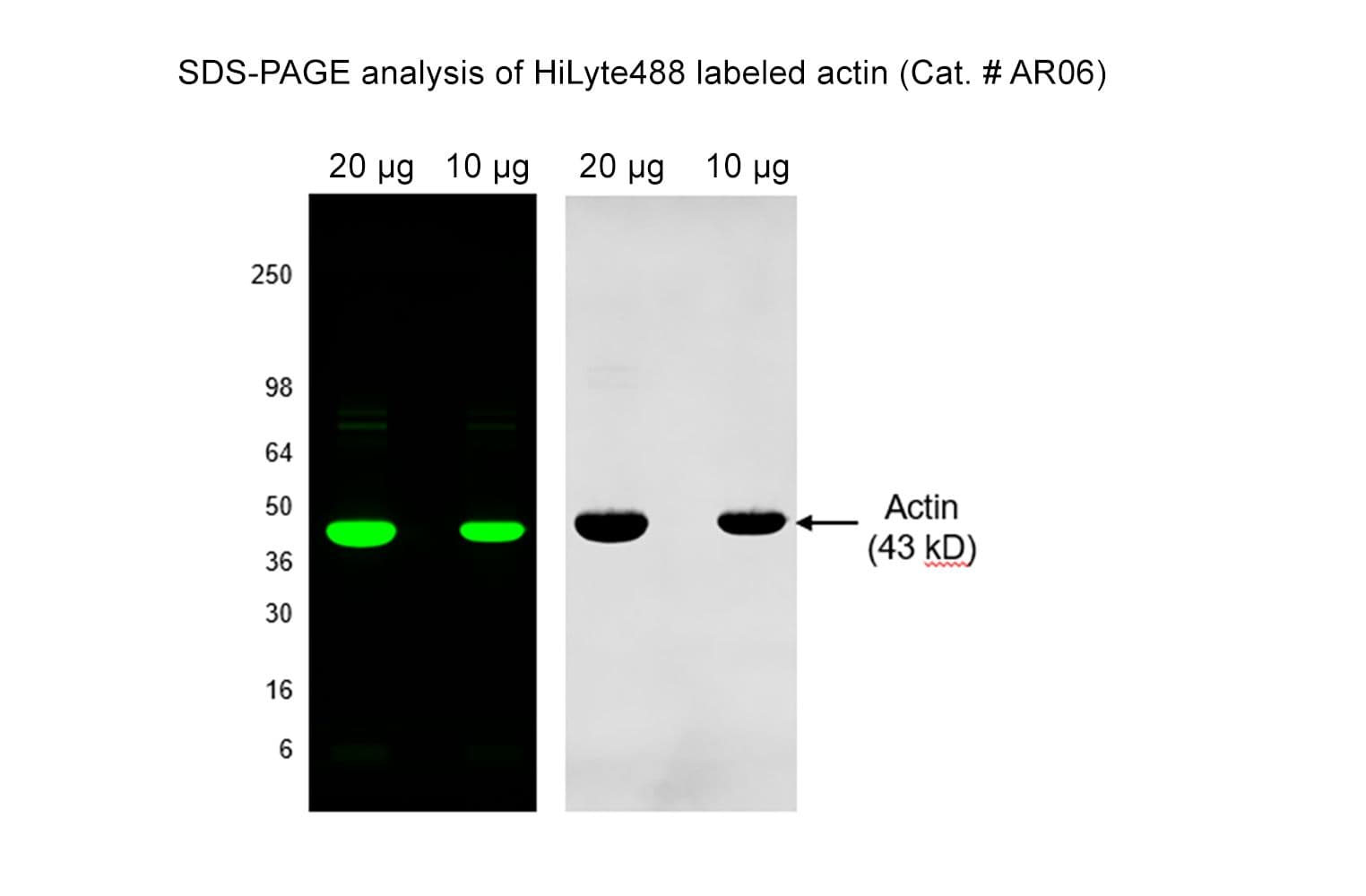
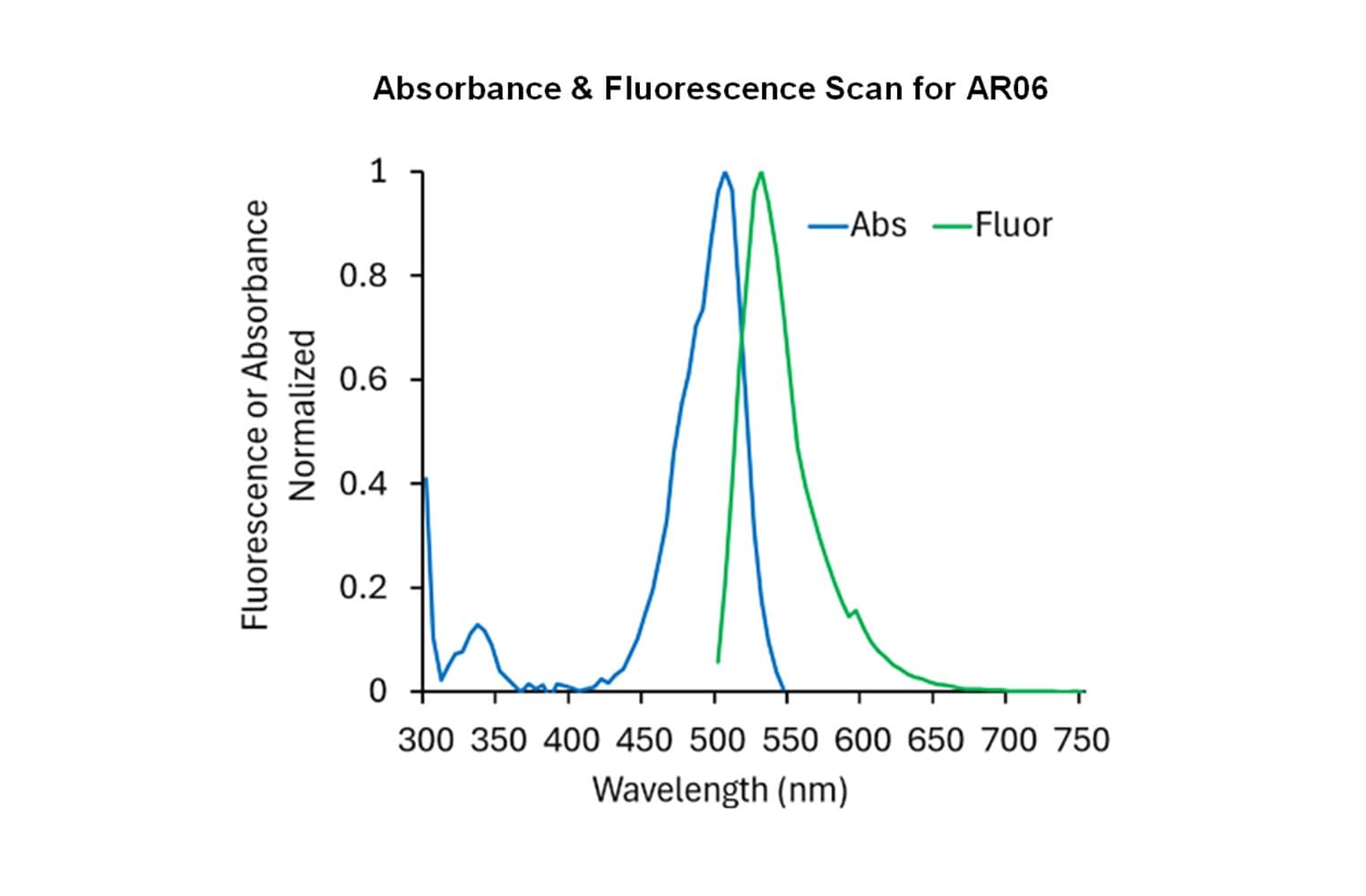
Rabbit skeletal muscle actin AKL99 is randomly labeled on surface lysines with an activated ester of HiLyte™488 fluorescent dye:
Protein purity is assessed by scanning densitometry of Coomassie Blue-stained protein on a12% polyacrylamide gel. Purity was determined to be >99% pure.
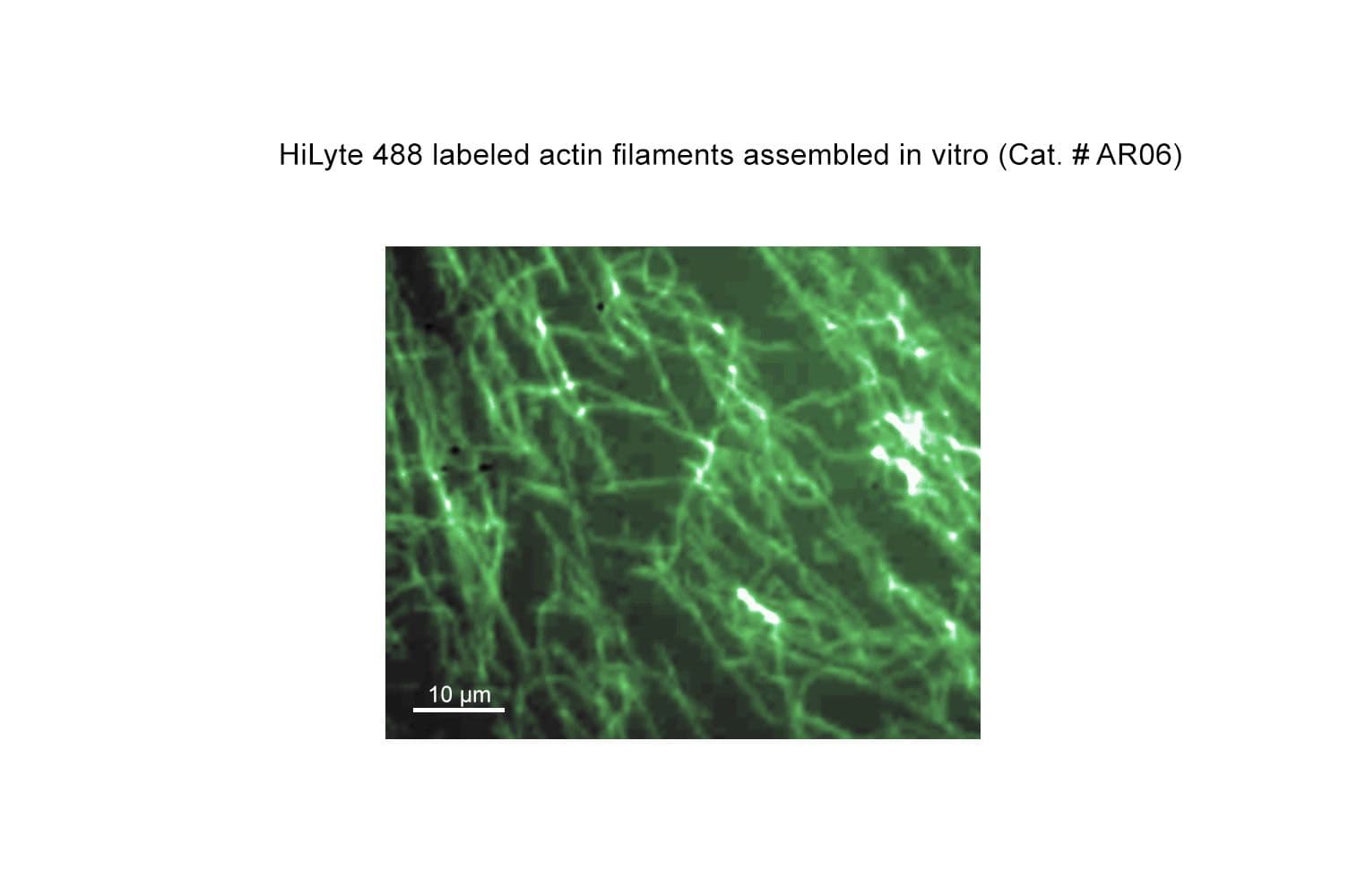
The biological activity of HiLyte™488 actin can be determined by its ability to polymerize into filaments in vitro efficiently and separate from unpolymerized components in a spin-down assay. Stringent quality control ensures that ≥90% of the labeled actin can polymerize.
Cat. #AR06