Cofilin 1 protein: human recombinant
Cat. #CF01
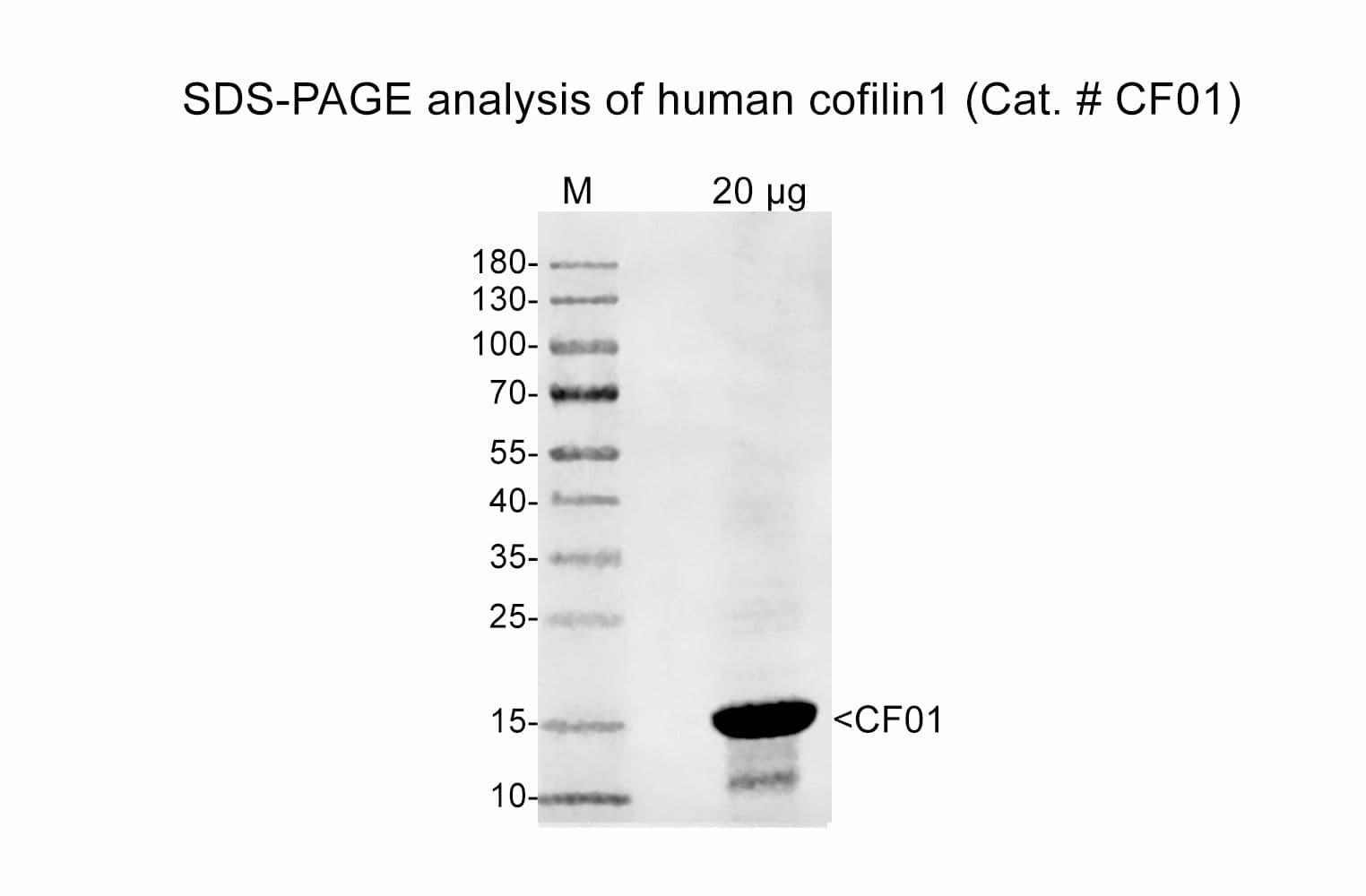

Cat. #CF01


Cofilin 1 is an actin-binding protein that regulates cytoskeletal dynamics by severing and depolymerizing actin filaments, playing a key role in cell motility, endocytosis, and signal transduction. It is essential for processes such as cell migration, neural development, and immune responses, and its dysregulation has been linked to cancer metastasis and neurodegenerative diseases.
The human cofilin protein (isotype 1) has been produced in a bacterial expression system.
Protein purity is determined by scanning densitometry of Coomassie Blue-stained protein on a 4-20% polyacrylamide gel. Purity is ≥95%
The biological activity of recombinant cofilin is assessed by its pH-dependent binding and severing of F-actin.
Quality control ensures these binding/severing patterns are consistently observed by SDS-PAGE analysis.