Rho Activator I
Cat. #CN01
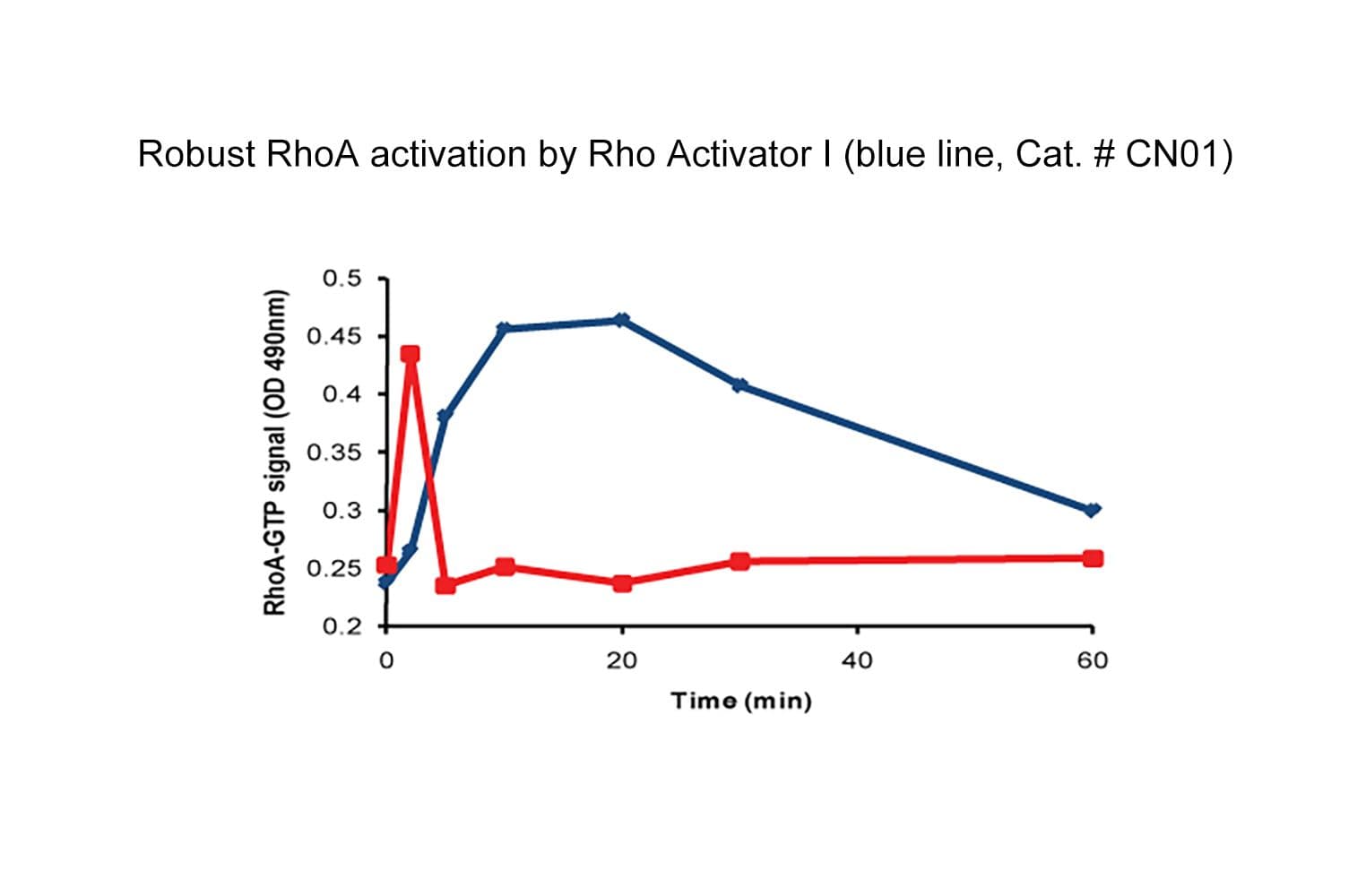

Part of the G-Switch™ line, CN01 (calpeptin) activates RhoA, RhoB, and RhoC in a variety of cell types through indirect activation. The mechanism involves inhibition of Shp-2 phosphatase, which results in constitutive activation of Rho GEFs.
In addition to its Rho-related effects, CN01 inhibits calpain-1 through a separate, unrelated mechanism.
Preparation & Use:
≥95% by HPLC
Biological activity of CN01 is demonstrated by its ability to activate RhoA in live Swiss 3T3 cells. Treatment at 1 unit/ml produces optimal activation, approximately twofold, within 10-20 minutes as measured by a RhoA G-LISA assay BK124. Rho activation by CN01 is typically more sustained than growth factor activations, which typically peak in 1-2 minutes and then rapidly decline. Responses to CN01 may vary depending on the cell line used.