Human KIF2C (MCAK) is a microtubule depolymerizing motor protein essential for regulating spindle dynamics and chromosome alignment during mitosis. By controlling microtubule length at kinetochores, KIF2C ensures accurate chromosome segregation and genomic stability.
The wild-type human motor domain of KIF5B is produced in a bacterial expression system.
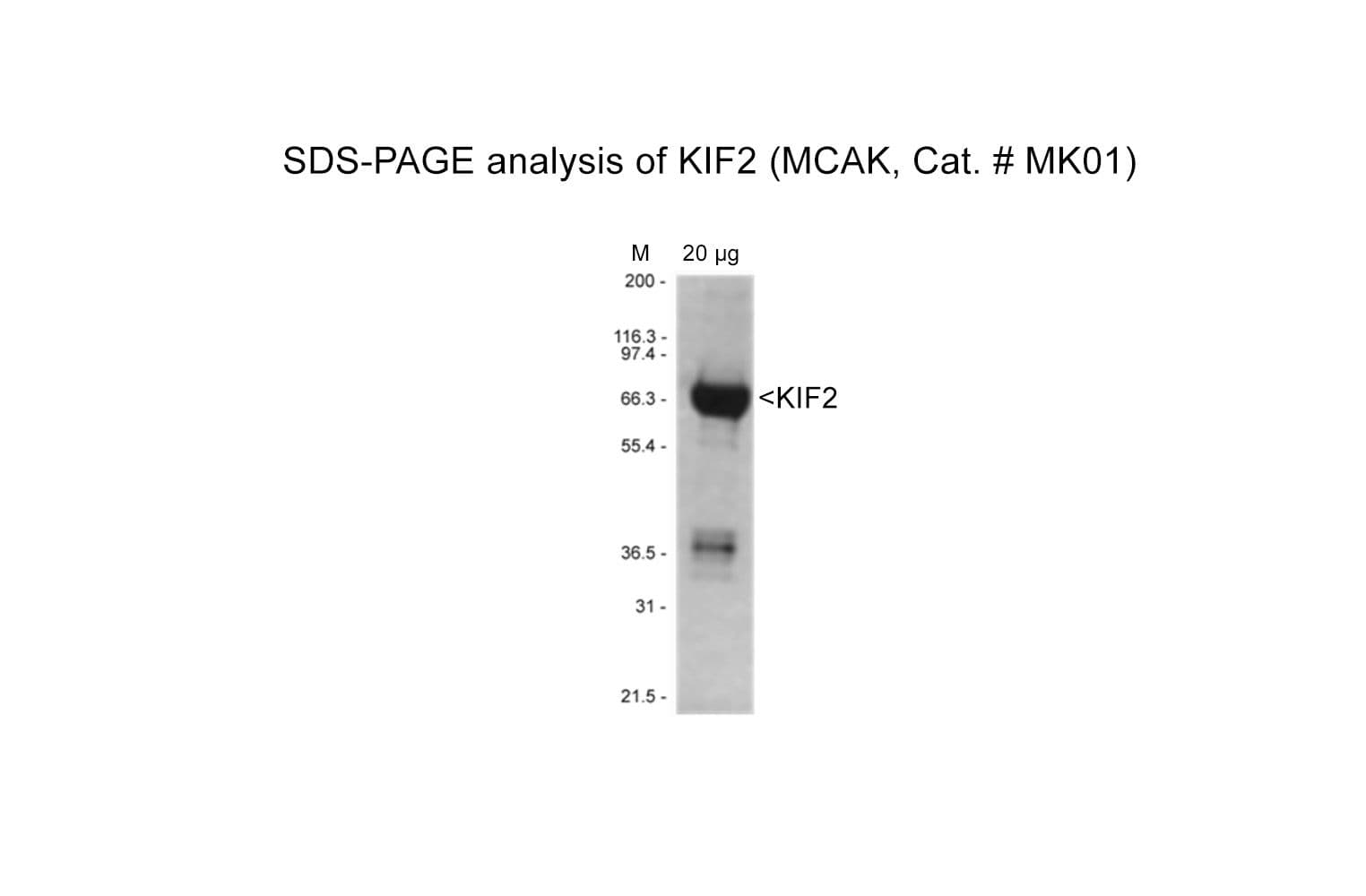
Protein purity is assessed by scanning densitometry of Coomassie Blue-stained protein on a4-20% polyacrylamide gel. Purity is determined to be >80% pure.
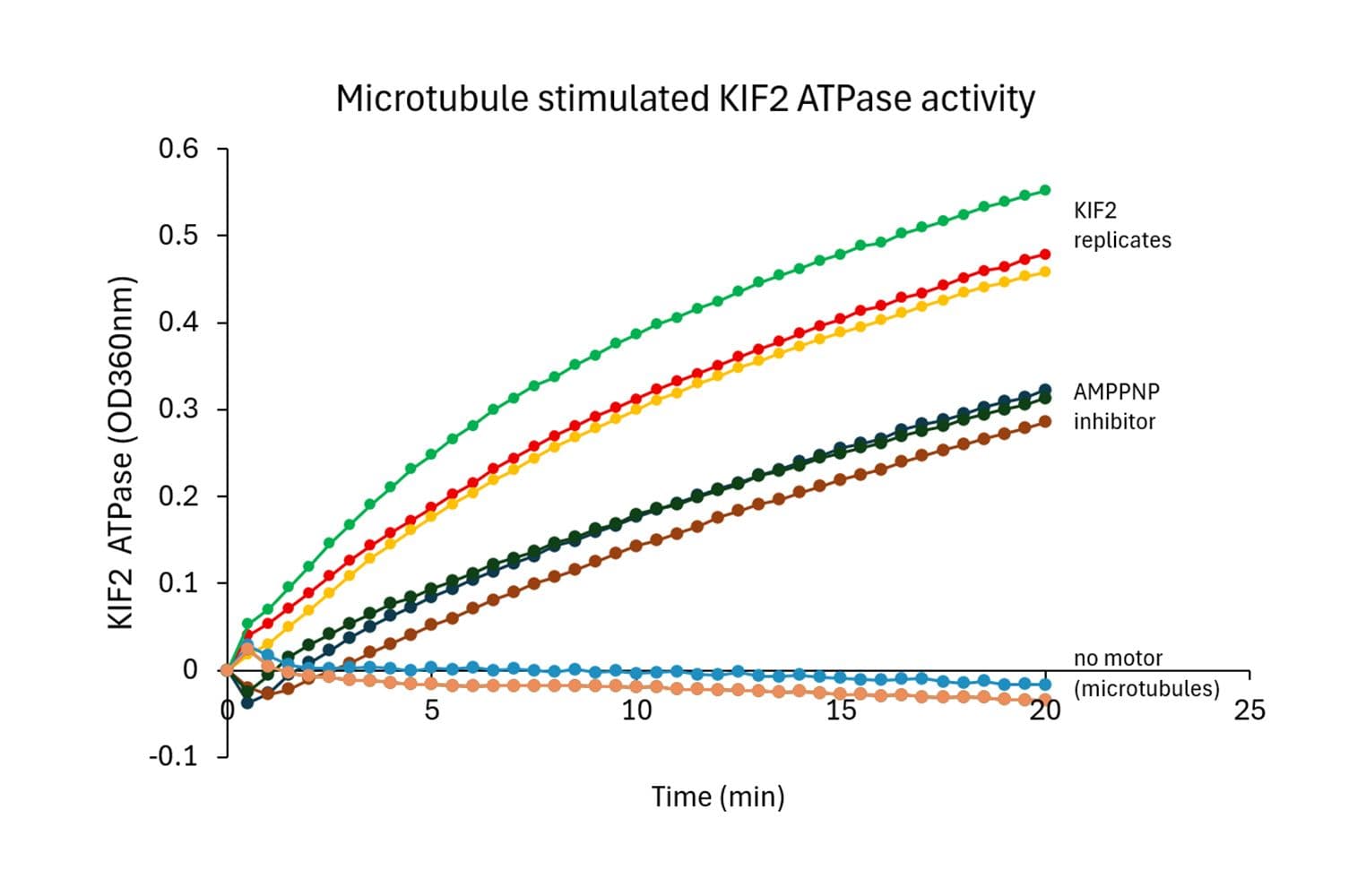
Biological activity of KIF2C is measured using a microtubule-activated ATPase assay. Under the conditions stated (see datasheet), KIF2C exhibits a microtubule-stimulated ATPase activity with a Vmax of ≥ 160 nmol of ATP produced per minute per mg of KIF2C.
Cat. #MK01