Rac1 protein: GST tagged: human wild type
Cat. #RCG01-C

Cat. #RCG01-C


Rac proteins are small GTP-binding proteins that regulate actin-based structures such as lamellipodia, membrane ruffles, and dorsal ruffles, enabling dynamic remodeling of the cytoskeleton. They are essential for processes including cell migration, adhesion, and growth factor–mediated signaling.
Wild-type human Rac1 protein is produced in a bacterial expression system.
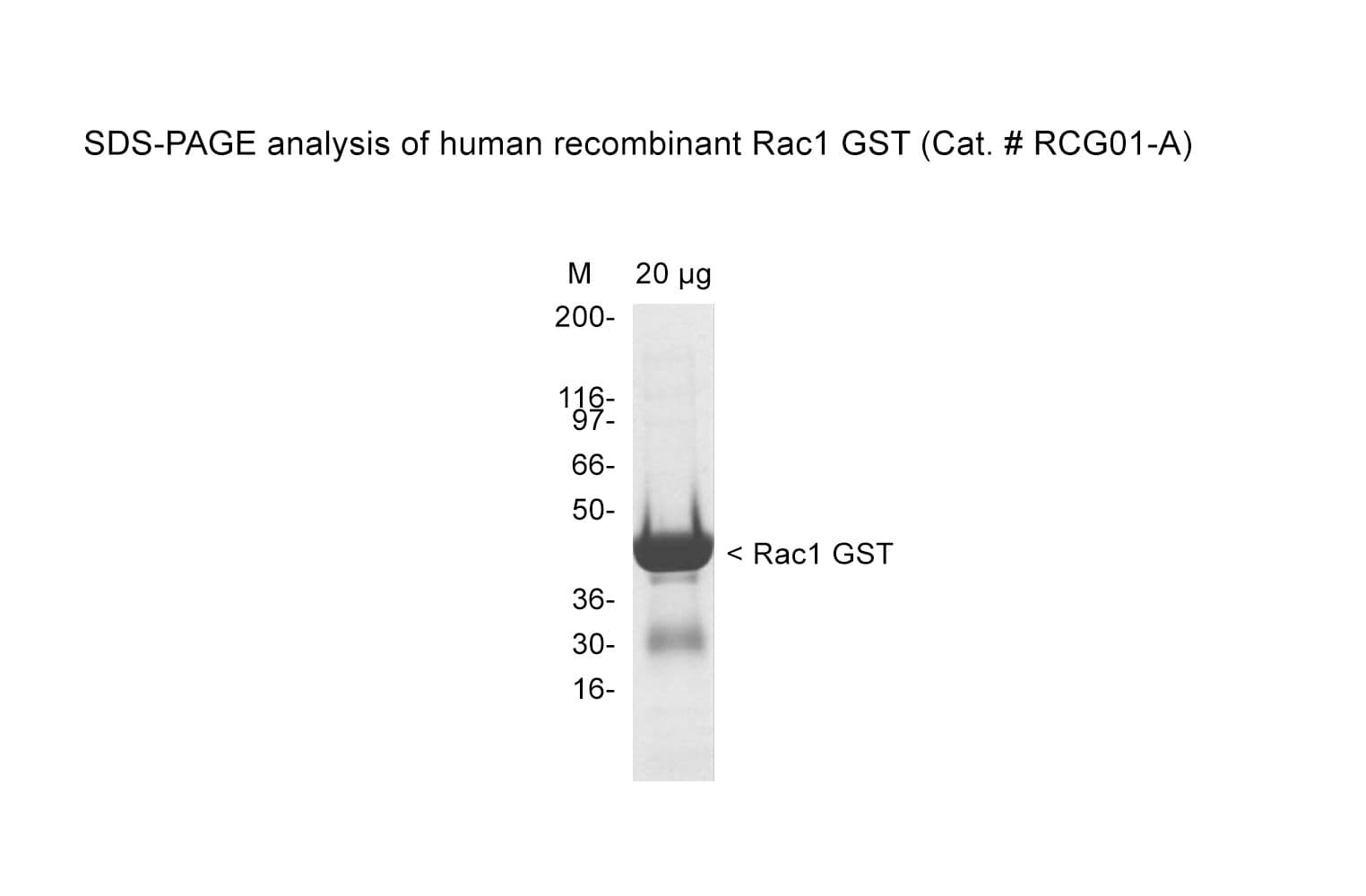
Protein purity is assessed by scanning densitometry of Coomassie Blue-stained protein on a 4-20% polyacrylamide gel. Purity was determined to be ≥90% pure.
The biological activity of RCG01 is evaluated by measuring its GTPase activity—its ability to hydrolyze GTP. Strict quality control ensures that GST-Rac1 exhibits at least a threefold increase in GTP hydrolysis rate in the presence of the Rac GAP, p50 Rho GAP.