Arp2/3 protein complex: porcine brain
Cat. #RP01P

Cat. #RP01P



The Arp2/3 complex (Actin-Related Proteins) is a highly conserved seven-subunit protein assembly that initiates the growth of branched actin filaments, playing a critical role in cell motility, endocytosis, and intracellular trafficking. Activated by nucleation-promoting factors, it binds to existing actin filaments and nucleates new branches, driving dynamic rearrangements of the actin cytoskeleton.
The Arp2/3 complex has been purified from porcine brain.
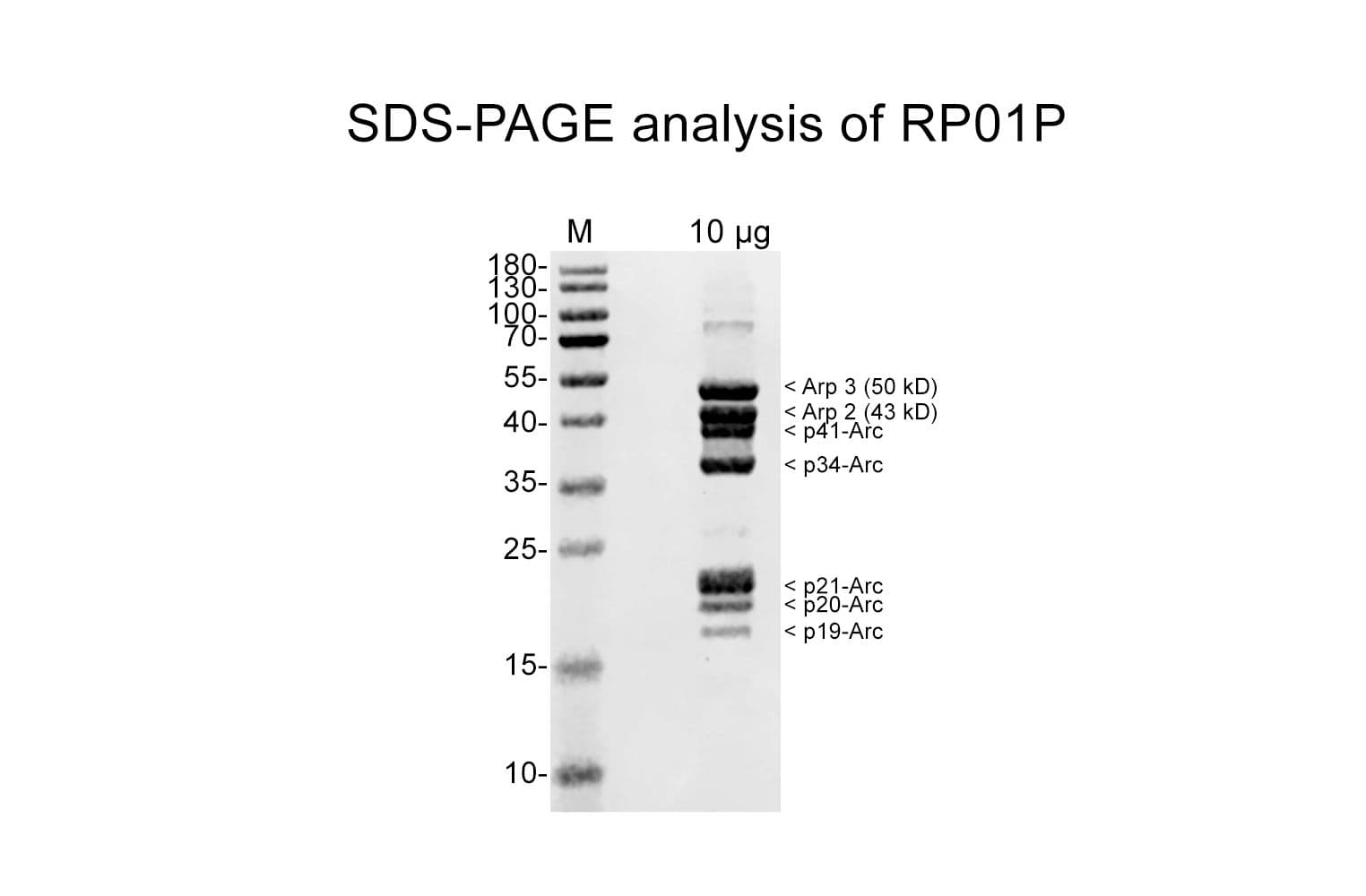
Protein purity is determined by scanning densitometry of Coomassie Blue-stained protein on a 4-20% polyacrylamide gel. Purity is >90%
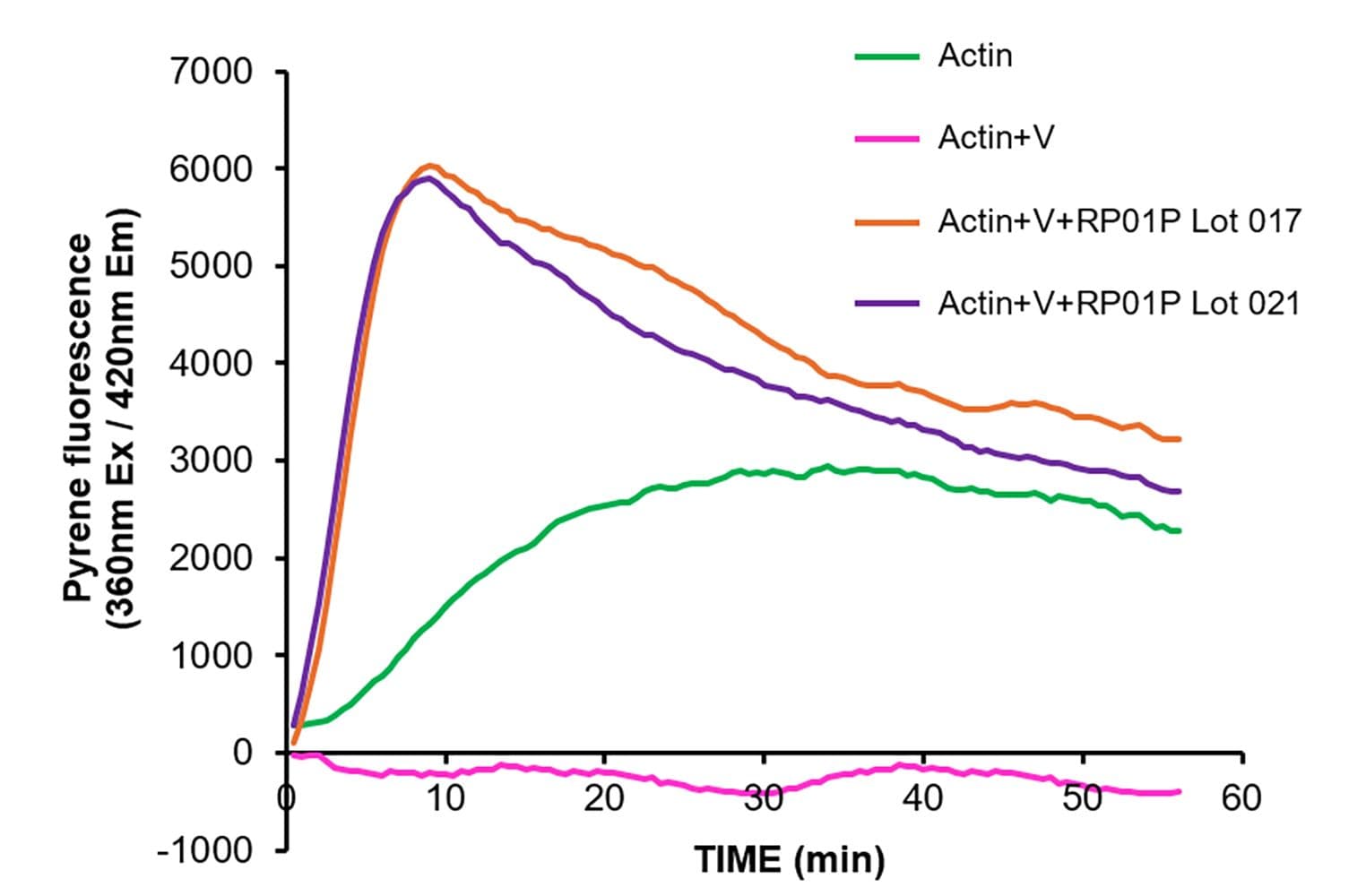
The biological activity of RP01P is determined by its ability to induce the branched polymerization of actin filaments in vitro at a molar ratio of 1:200 (Arp2/3:actin) in the presence of the VCA domain of N-WASP. Under the experimental conditions (see datasheet for details), the Arp2/3 complex can enhance actin polymerization 3-5 fold. Actin polymerization is assessed by kinetic fluorescence enhancement of pyrene actin at Ab: 360 nm, Em: 405 nm.