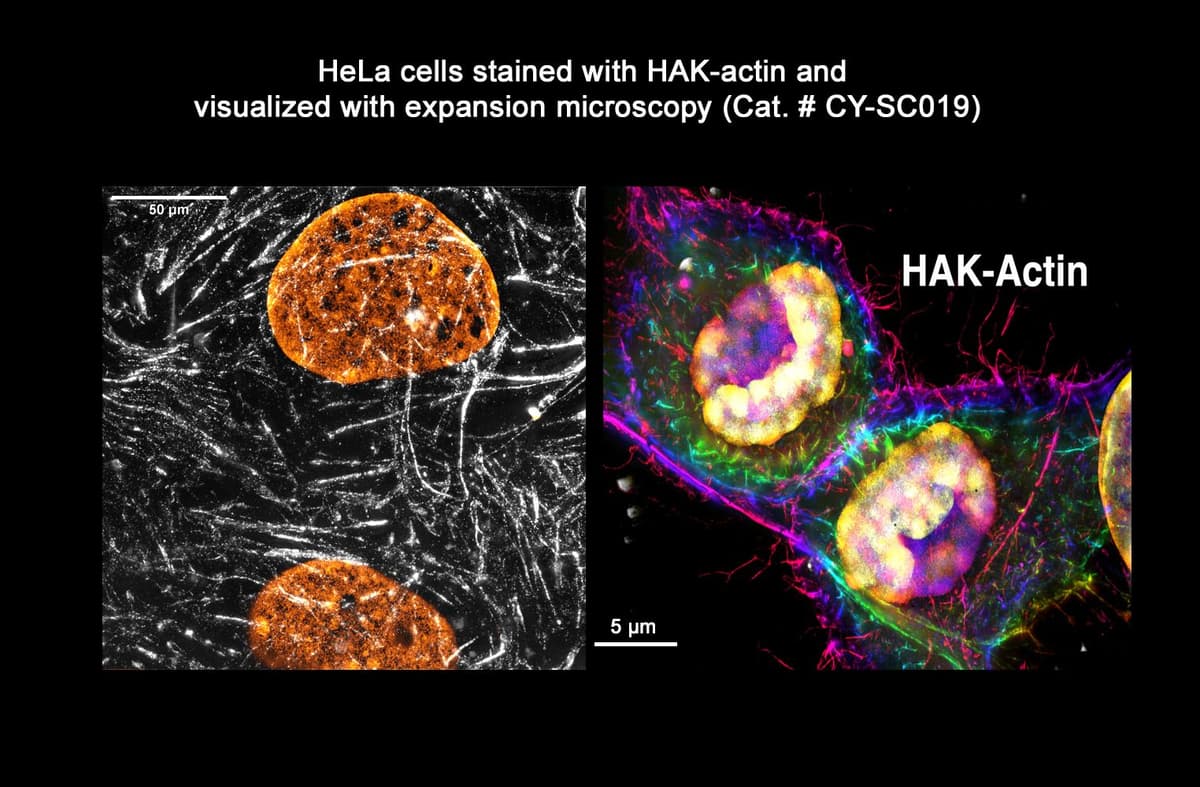
How does HAK-actin™ work?
The HAK-actin™ probe contains 3 key elements:
1. A pan-species, highly specific, high affinity F-actin ligand for a robust binding to polymerized actin (F-actin) only.
2. An anchoring moiety that specifically reacts with acrylamide and covalently links HAK-actin™ to the expansion gel.
3. The HA-tag epitope sequence for a strong and specific recognition by anti-HA antibodies, even after expansion.
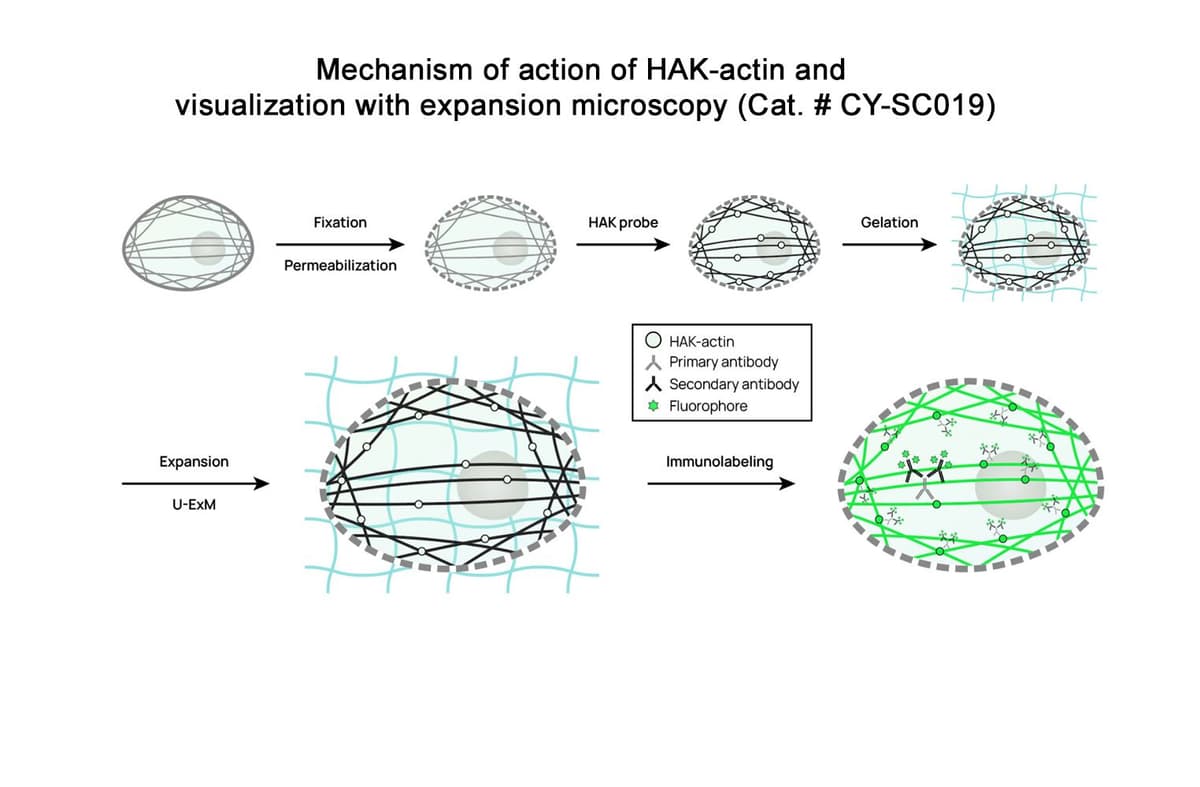
Figure 1. After fixation (chemical or cryo) and permeabilization, the sample to be expanded is treated with HAK-actin™. The probe’s actin ligand uniformly labels F-actin within the cells. The sample is then subjected to U-ExM (or iU-ExM) protocol. During the protocol, HAK-actin™ is covalently bound within the expansion gel and expands faithfully with the sample. The final immunolabeling step using a primary anti-HA and a fluorescently labeled secondary antibody boosts the fluorescent signal and reveals the expanded actin structure with high contrast.