TECH GUIDE
+3
Loading...
Cat. #BK005

Kit contents (300 assays)
Equipment & materials required
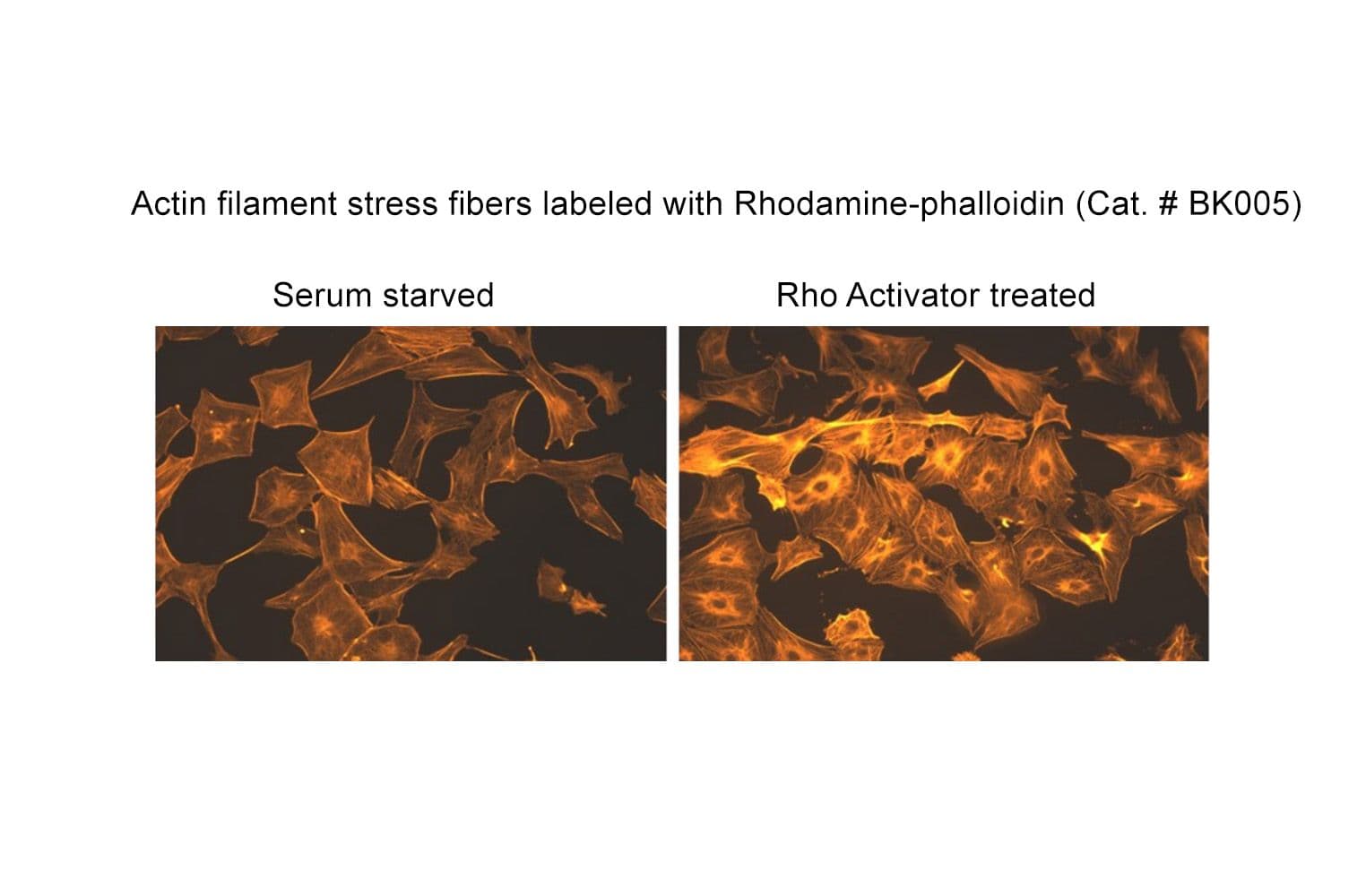
This fluorescent phalloidin-based actin staining kit enables precise visualization of F-actin in fixed cells. It includes all necessary fixation buffers for consistent, high-specificity results.
Key characteristics