Introduction
Protein acetylation is an essential and reversible post-translational modification (PTM) utilized throughout the cell to control many cellular processes(reviewed in 1). Acetyl modifications are added to proteins through an enzymatic process carried out by lysine acetyltransferases and removed via lysine deacetylases. While acetylation of histones, which leads to epigenetic regulation of gene expression is well-studied, non-histone protein acetylation is also an important PTM that may be disrupted in neurological, metabolic, cancer, and cardiovascular diseases1. There is growing evidence that disrupted acetylation mechanisms may lead to the progression of type 2 diabetes (T2D), and a recent review highlights several PTMs including histone acetylation as a key regulatory mechanism(reviewed in 2, 3). Additionally, there is emerging evidence that non-histone acetylation is also important in the setting of T2D and this newsletter highlights key findings and recent examples.

HDAC6 Interacts with Leptin to Control Obesity and T2D
T2D is a metabolic disease characterized by a heterogeneous loss of islet b cell insulin secretion and chronic insulin resistance (IR) (reviewed in[4]). Interestingly, T2D accounts for 96% of all diabetes cases and is often associated with obesity, metabolic dysfunction, and dyslipidemia. Due to T2Ds strong association with obesity it is frequently seen that pathways and processes that regulate obesity also impact T2D. One example is the protein Leptin, an adipocytokine that signals the brain to suppress appetite and maintain blood glucose (reviewed in [5]), whose dysregulation has been associated with severe diabetes pathologies[6, 7]. In 2018, CRISPR-mediated deletion of Leptin receptor specifically in agouti-related peptide (AGRP) neurons was shown to be sufficient to cause severe obesity and diabetes further linking this signaling mechanism to T2D [8]. More recently, Cakir et al. showed that histone deacetylase 6 (HDAC6), a lysine deacetylase, can affect leptin-regulated obesity[9]. Specifically, the group showed that HDAC6 inhibitors sensitize diet-induced obese mice to leptin signaling as a mechanism to improve systemic glucose homeostasis and reduce obesity. A new study shows that HDAC6 can interact with the Leptin receptor to suppress Leptin signaling[10], and depletion of HDAC6 in AGRP neurons or treatment with blood-brain-barrier impermeable HDAC6 inhibitors showed no anti-obesity effect further supporting Leptin signaling as a specific mechanism utilized by HDAC6 to affect obesity and T2D. In a different study, Rosenblum et al. showed that Leptin signaling can activate STAT1 pathways[11], which is particularly interesting as STAT1 was recently implicated in Type I diabetes[12]. Emerging evidence by Leslie et al. shows that in human pancreatic b cells, STAT1 signaling can be regulated by HDAC6 and may be important for diabetes progression. Collectively, these findings point to multiple mechanisms by which HDAC6 may regulate T2D.
Sirtuin 1 is a Critical Deacetylase in T2D
Sirtuin-1 (SIRT1) is an NAD+ lysine deacetylase that has been linked to fat mobilization13 and glucose metabolism14; thus, implicating it as a critical regulator of obesity and a potential target for the treatment of T2D. Interestingly, the SIRT1 targeting drug, resveratrol, was shown to increase insulin sensitivity, increase mitochondrial number, and enhance life-span in multiple model organisms15, 16. To pinpoint the role of SIRT1 in metabolic disease, a mouse model with moderate overexpression of SIRT1 was developed, and this mouse was resistant to high-fat diet-induced metabolic-related pathologies17. Unfortunately, clinical trials with resveratrol were not successful in part due to low bioavailability and negative side effects; however, a third-generation drug, SRT2104, which is 1000 times more effective at activating SIRT1, is now being tested in numerous clinical trials for an array of diseases including in diabetes(reviewed in 18). Interest in the role of SIRT1 in T2D remains high and has focused on downstream targets of SIRT1 in disease-related settings. For example, a study by Velagapudi et al. identified Toll-like receptor 4 as a direct target of SIRT1 deacetylation in β-cells in the setting of cardiovascular disease19, which is a disease-complication for many patients with T2D. Another group showed that tuberous sclerosis complex 2 (TSC2) is also a target for SIRT1 deacetylation, and they showed that in a pancreatic β-cell model, deacetylation of TSC2 with resveratrol treatment altered mitochondrial turnover which may be important in metabolic disease settings like T2D20. Several other studies have also looked at downstream effectors of SIRT1 in other secondary diseases such as diabetic nephropathy, diabetic cardiomyopathy, and others.
The NAT2 N-Acetyltransferase Regulates Insulin Sensitivity
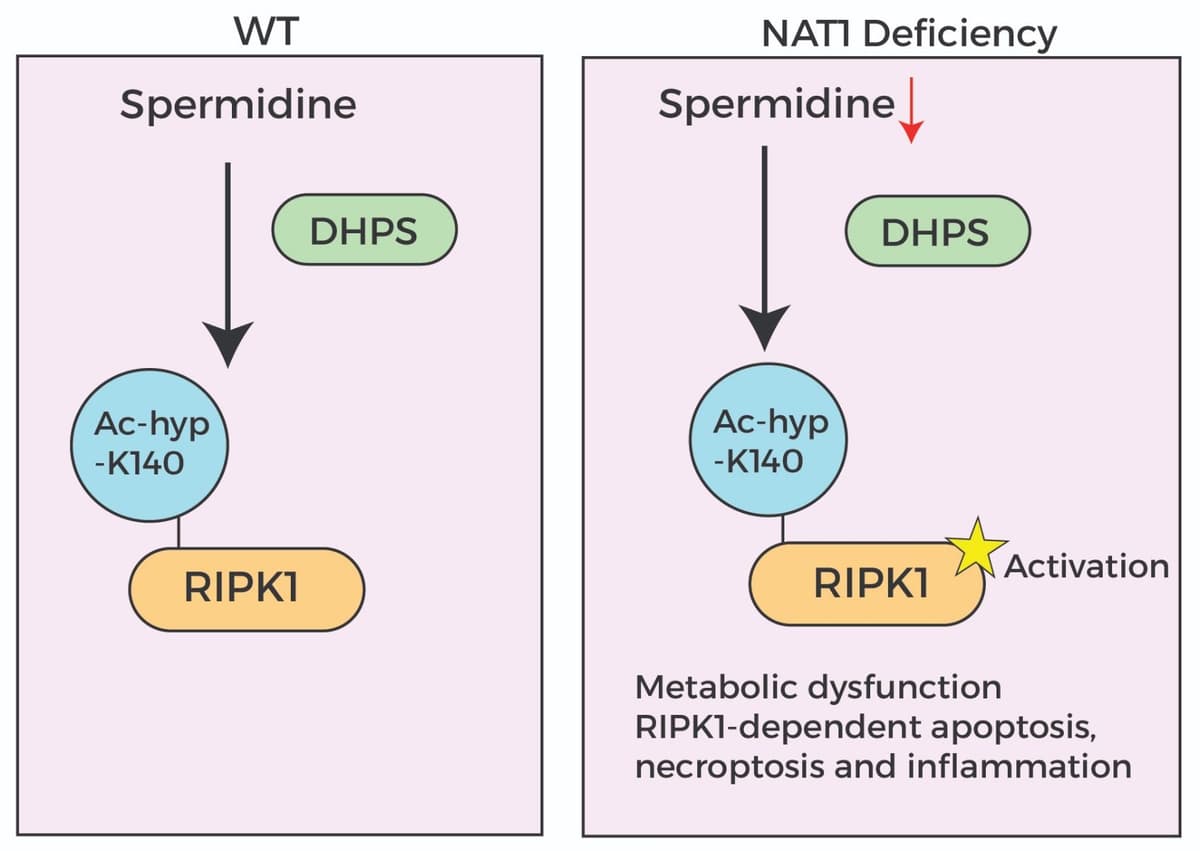
While it is clear that IR is a necessary component for the progression of T2D, the precise genetic factors that may control IR are not fully understood. Many known genetic factors regulate insulin secretion, processing, and synthesis, but not insulin sensitivity directly. In 2015 a genome-wide association study was performed, and a nonsynonymous variant of N-acetyltransferase 2 (NAT2) was shown to correlate with decreased insulin sensitivity21. The group performed in vivo studies with the mouse homolog NAT1 (mNAT1) and found that it altered insulin sensitivity in mice21. A follow-up study using a mNAT1 knockout mouse model further confirmed that it is important for muscle, liver, and adipose tissue insulin sensitivity, but also promotes reduced mitochondrial activity and increased ectopic lipid deposition22. NAT2 is an arylamine N-acetyltransferase that promotes the turnover of specific xenobiotics through acetylation. A recent study identified spermidine, an aliphatic polyamine found naturally in cells, as an endogenous target of NAT223. Spermidine is an intriguing molecule as its levels have been linked to age-related pathologies24. Zhang et al. recently identified a mechanism where NAT2 acetylates spermidine, leading to a novel acetylhypusination PTM on receptor-interacting serine/threonine-protein kinase 1 (RIPK1) (see figure 1)25. Interestingly, the group found decreased spermidine and enhanced RIPK1 activation in vascular tissue of human patients with diabetes. Using in vivo mouse models, the group showed that diabetic phenotypes induced by mNAT1 deficiency could be reversed with spermidine supplementation or RIPK1 inhibition25. Surprisingly, a study has found potential negative crosstalk between NAT2 and SIRT1, whereby, silencing of SIRT1 increased N-acetylation capacity26. NAT2 function appears to play an integral role in T2D, and it will be interesting to further understand the pathways and processes involved.
Conclusion and Future Insights
This newsletter highlights several non-histone, acetylation mechanisms that have profound effects on T2D progression. Whether any of these pathways will result in viable therapies is still unknown, but having a better understanding of specific downstream targets and cell types impacted by these mechanisms will provide the ability to develop more selective therapies. Additionally, as diabetes leads to many secondary health complications, which often lead to morbidity and mortality, it will be critical to understand how acetylation contributes to those diseases in the setting of T2D. Examples of this include acetylation in viral infection in diabetes27, neuronal injury in diabetes28, diabetic cerebral ischemia-reperfusion injury29, among others.
References
- Jiang, N., et al., Acetylation in pathogenesis: Revealing emerging mechanisms and therapeutic prospects. Biomed Pharmacother, 2023. 167: p. 115519.
- Jazieh, C., et al., Unraveling the epigenetic fabric of type 2 diabetes mellitus: pathogenic mechanisms and therapeutic implications. Front Endocrinol (Lausanne), 2024. 15: p. 1295967.
- Yang, C., et al., Regulation of insulin secretion by the post-translational modifications. Front Cell Dev Biol, 2023. 11: p. 1217189.
- Lu, X., et al., Type 2 diabetes mellitus in adults: pathogenesis, prevention and therapy. Signal Transduct Target Ther, 2024. 9(1): p. 262.
- Friedman, J., 20 years of leptin: leptin at 20: an overview. J Endocrinol, 2014. 223(1): p. T1-8.
- Zhang, Y., et al., Positional cloning of the mouse obese gene and its human homologue. Nature, 1994. 372(6505): p. 425-32.
- Chen, H., et al., Evidence that the diabetes gene encodes the leptin receptor: identification of a mutation in the leptin receptor gene in db/db mice. Cell, 1996. 84(3): p. 491-5.
- Xu, J., et al., Genetic identification of leptin neural circuits in energy and glucose homeostases. Nature, 2018. 556(7702): p. 505-509.
- Cakir, I., et al., Histone deacetylase 6 inhibition restores leptin sensitivity and reduces obesity. Nat Metab, 2022. 4(1): p. 44-59.
- Guan, D., et al., Central inhibition of HDAC6 re-sensitizes leptin signaling during obesity to induce profound weight loss. Cell Metab, 2024. 36(4): p. 857-876.e10.
- Rosenblum, C.I., et al., Functional STAT 1 and 3 signaling by the leptin receptor (OB-R); reduced expression of the rat fatty leptin receptor in transfected cells. Endocrinology, 1996. 137(11): p. 5178-81.
- Chaimowitz, N.S., et al., STAT1 Gain of Function, Type 1 Diabetes, and Reversal with JAK Inhibition. N Engl J Med, 2020. 383(15): p. 1494-1496.
- Picard, F., et al., Sirt1 promotes fat mobilization in white adipocytes by repressing PPAR-gamma. Nature, 2004. 429(6993): p. 771-6.
- Rodgers, J.T., et al., Nutrient control of glucose homeostasis through a complex of PGC-1alpha and SIRT1. Nature, 2005. 434(7029): p. 113-8.
- Cohen, H.Y., et al., Calorie restriction promotes mammalian cell survival by inducing the SIRT1 deacetylase. Science, 2004. 305(5682): p. 390-2.
- Baur, J.A., et al., Resveratrol improves health and survival of mice on a high-calorie diet. Nature, 2006. 444(7117): p. 337-42.
- Pfluger, P.T., et al., Sirt1 protects against high-fat diet-induced metabolic damage. Proc Natl Acad Sci U S A, 2008. 105(28): p. 9793-8.
- Chang, N., et al., Emerging roles of SIRT1 activator, SRT2104, in disease treatment. Sci Rep, 2024. 14(1): p. 5521.
- Velagapudi, S., et al., Inhibition of de novo ceramide synthesis by sirtuin-1 improves beta-cell function and glucose metabolism in type 2 diabetes. Cardiovasc Res, 2024. 120(11): p. 1265-1278.
- Marques, P., et al., Regulation of TSC2 lysosome translocation and mitochondrial turnover by TSC2 acetylation status. Sci Rep, 2024. 14(1): p. 12521.
- Knowles, J.W., et al., Identification and validation of N-acetyltransferase 2 as an insulin sensitivity gene. J Clin Invest, 2015. 125(4): p. 1739-51.
- Camporez, J.P., et al., Mechanism by which arylamine N-acetyltransferase 1 ablation causes insulin resistance in mice. Proc Natl Acad Sci U S A, 2017. 114(52): p. E11285-E11292.
- Conway, L.P., et al., Unexpected Acetylation of Endogenous Aliphatic Amines by Arylamine N-Acetyltransferase NAT2. Angew Chem Int Ed Engl, 2020. 59(34): p. 14342-14346.
- Soda, K., Overview of Polyamines as Nutrients for Human Healthy Long Life and Effect of Increased Polyamine Intake on DNA Methylation. Cells, 2022. 11(1).
- Zhang, T., et al., Spermidine mediates acetylhypusination of RIPK1 to suppress diabetes onset and progression. Nat Cell Biol, 2024.
- Salazar-Gonzalez, R.A., et al., Expression and genotype-dependent catalytic activity of N-acetyltransferase 2 (NAT2) in human peripheral blood mononuclear cells and its modulation by Sirtuin 1. Biochem Pharmacol, 2018. 156: p. 340-347.
- Nobs, S.P., et al., Lung dendritic-cell metabolism underlies susceptibility to viral infection in diabetes. Nature, 2023. 624(7992): p. 645-652.
- Yao, Y., et al., Pyruvate dehydrogenase kinase 1 protects against neuronal injury and memory loss in mouse models of diabetes. Cell Death Dis, 2023. 14(11): p. 722.
- Zhou, H.J., et al., Interfering TRIB3 protects the blood brain barrier through PI3K/Akt pathway to alleviate cerebral ischemia-reperfusion injury in diabetes mellitus mice. Chem Biol Interact, 2024. 387: p. 110807.


