Key takeaways
1. SUMOylation has critical functions in the formation of stress granules, nucleoli, and promyelocytic leukemia protein nuclear bodies.
2. Condensate formation incorporates SUMOylated proteins as structural elements.
What are SUMOylation and condensates?
Post-translational modifications (PTMs) such as ubiquitination, acetylation, phosphorylation, and SUMOylation are covalent modifications, which are crucial regulatory elements that can become dysregulated in many diseases. Biomolecular condensates, which often form through liquid-liquid phase separation (LLPS), are compartments without an encapsulating membrane that provide cellular organization and enrich key target proteins or complexes to drive specific biological processes(reviewed in 1).
What connection is there between SUMOylation and condensates?
There is a growing body of evidence describing meaningful interplay between condensate formation and PTM regulation of target proteins(reviewed in 2). The PTM, SUMOylation, is particularly interesting as it has been shown to have critical functions in biomolecular condensates like stress granules, Cajal bodies, nucleoli, Promyelocytic leukemia protein (PML) nuclear bodies, and others(reviewed in 3). Interestingly, there are potentially thousands of proteins targeted for SUMOylation, yet the number of regulatory SUMO ligases and proteases is relatively limited; therefore, investigators have sought to identify other cellular mechanisms that may assist with directing SUMOylation events. Biomolecular condensates may be a key mechanism that spatially and temporally regulates SUMOylation, and this newsletter discusses recent findings dissecting the interplay between SUMOylation and biomolecular condensates.

What were the initial observations that connected SUMOylation and LLPS condensates?
Proteins involved in LLPS formation often contain intrinsically disordered regions (IDRs) that utilize weak multivalent interactions for enrichment and formation of protein complexes(reviewed in 1). Due to the heterogeneity of LLPS condensates, there is still much to learn about the components and processes involved in their biogenesis. In 2016, Banani et al. identified SUMO proteins as an important player in LLPS4. The group showed that mixing a pair of artificial proteins – one composed of SUMO3 repeats and the other composed of SIM repeats from PIASx – was sufficient to form phase-separated droplets in vitro, while an ectopically expressed GFP-SUMO localized to PML nuclear bodies in cells4.
Interestingly, a bioinformatics study analyzed the composition of PML nuclear bodies and determined that of the 205 proteins identified in PML bodies, approximately 44% contain SUMO binding sites14.Additionally, they reported that SUMO-related proteins like UBC9 (SUMO E2 enzyme) and SUMO1 were key scaffold proteins in PML nuclear bodies. This is in line with previous work that showed PML nuclear bodies were enriched with UBC9, SUMO isoforms, and SUMO E3 ligases(reviewed in 3).
What structural elements are involved?
Structural / activity studies determined that lysines residing in disordered regions are preferentially targeted by SUMO5, further linking SUMOylation to condensate mechanics. Schmit and colleagues utilized the polySUMO/polySIM model to further understand how SUMOylation contributes to organization within the condensate, and found that the polySMO-polySIM interaction formed a zipper-like filament structure, which had defects in unsatisfied bonds that could interact with “client” proteins6. These findings provided compelling evidence that SUMOylation events can produce structural states within LLPS condensates that are critical for driving 3D network formation.
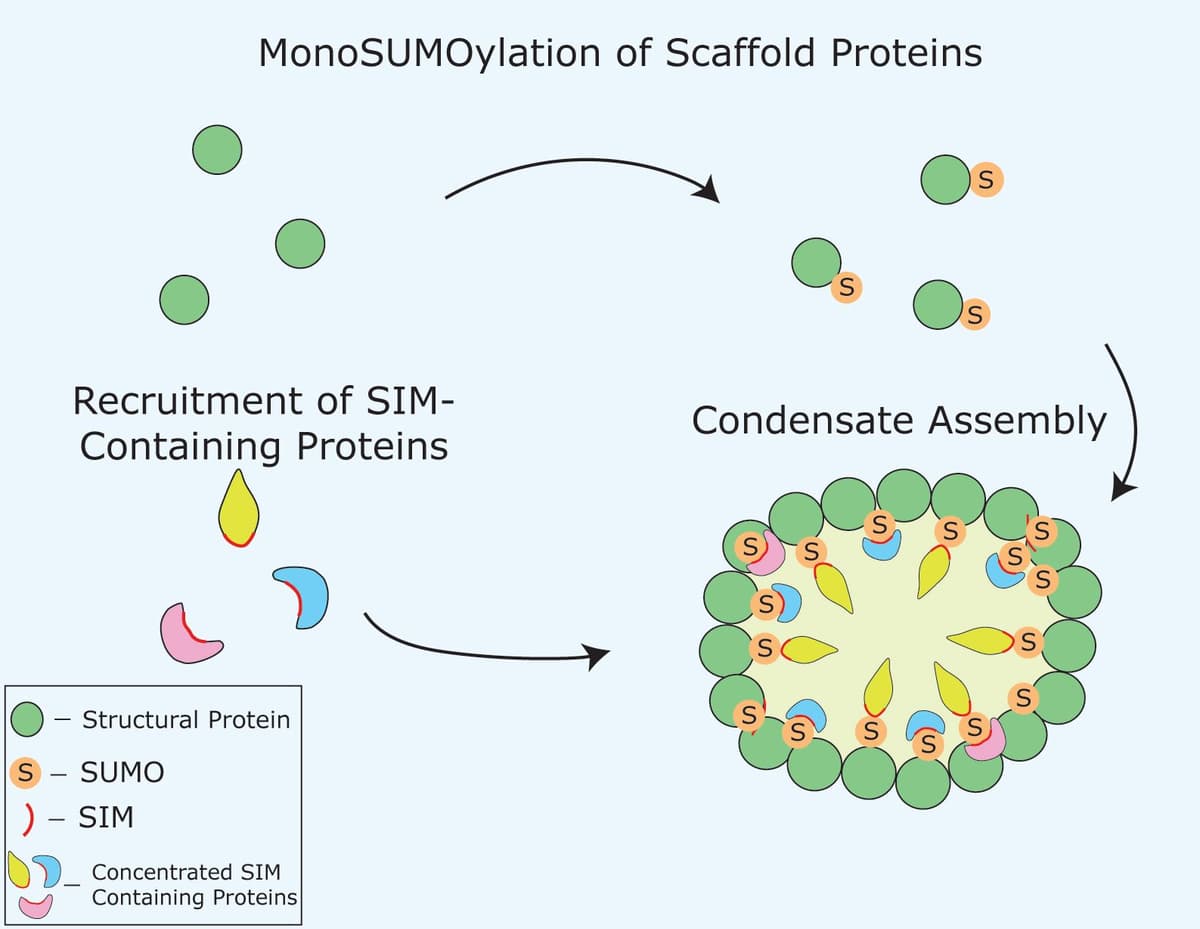
Is SUMOylation a pre-requisite for condensation?
Several studies have linked the SUMOylation process to biomolecular condensates, such as a recent study by Li et al. that utilized their DenseMAP photocrosslinking strategy, which identifies protein interactomes in condensates, and showed that SUMOylation has a critical role in controlling nucleolar proteome homeostasis7. This is in agreement with previous studies showing SUMOylation contributes to condensate formation of target molecules; for example, it was shown to be important for enhanced SOP-2 phase separation in vitro8.
In another instance, the insulin receptor tyrosine kinase substrate protein was able to regulate heterochromatin by infiltrating condensates and recruiting the E2 ligase Ubc9, which led to the SUMOylation of HP1α9 In a third example, SUMOylation was shown to regulate piwi-interacting RNA transcription in biomolecular condensates10. Finally, it was shown that cAMP triggers SUMOylation through the formation of Epac1 and SUMO-containing condensates11. Importantly, condensate formation not only brings SUMO together with its target substrates, but it also enhances the rate of SUMOylation. The Rosen group showed, using their polySUMO/polySIM model that SUMOylation rates can increase up to 36-fold in phase droplets compared to the surrounding bulk12.Collectively, these findings suggest that the interplay between SUMO and LLPS may be important both for condensate formation and for effective SUMOylation.
How are ligase coordinated with PML to create PML Nuclear Bodies?
PML nuclear bodies are nuclear-localized, membrane-less, and dynamic structures that are involved in many biological processes, including apoptosis, chromatin remodeling, DNA replication, anti-viral response, and many others(reviewed in 13). The PML protein is a key component of PML nuclear bodies that functions as a scaffold for other proteins in the condensate compartment. SUMOylation of PML and other proteins within PML nuclear bodies is critical for the proper formation and function of these condensates(reviewed in 3, 13).
How do SUMO-condensates affect viral infection?
SUMOylation is thought to be a critical cellular process that is activated to help cells respond to various forms of cell stress(reviewed in 23). A significant amount of cellular stress occurs during viral infection, and many studies have examined the positive and negative role that SUMOylation plays in viral infection and pathogenesis(reviewed in 24). Numerous viruses have been shown to hijack or circumvent the host SUMO machinery during infection, but recent studies have begun linking this mechanism of viral infection to biomolecular condensates as well. For example, the Polycomb Repressive Complex component CBX4 was shown to form nuclear bodies with LLPS on the HIV-1 long terminal repeat to maintain HIV-1 latency25.These CBX4 nuclear bodies recruit EZH2 and SUMOylate it using its E3 ligase activity as a mechanism to stimulate the H3K28 methytransferase activity of EZH2. In another example, the Toll-interacting protein (TOLLIP) was shown to promote IDR-dependent condensates that promoted SENP1 aggregation and the deSUMOylation of the mitochondrial antiviral signaling (MAVs) protein26. The group found that TOLLIP condensation was critical for regulating MAVs activity and viral infection. Viral cells can also control the SUMO machinery in condensates. Work by Lin et al. showed that the Tomato bushy stunt virus (TBSV) p33 protein co-opts the host SUMO machinery, and found this interaction to be critical for viral condensate formation and ultimately viral replication27. Another report described how SUMOylation of the SARS-CoV-2 nucleocapsid protein is important for its ability to undergo LLPS and evade innate antiviral response28. Interestingly, an interfering peptide of TRIM28, the E3 ligase that mediates SARS-CoV-2 NP SUMOylation, can significantly inhibit viral replication. Studies investigating the interplay between viral replication and SUMO-mediated condensates are just beginning to emerge, but the current findings align with previous SUMO studies that suggest its contribution to viral infection is complex.
Summary and Future Perspectives
The compilation of studies above identifies an undeniable interplay between SUMOylation and biomolecular condensates that are important both for condensate formation and SUMO regulation of target proteins. While there is still much to learn about the full extent to which biomolecular condensates impact SUMO signaling, it is clear that they do provide spatial and temporal regulatory environments for this PTM. The information on STUBL mechanisms and how biomolecular condensates like PML nuclear bodies provide favorable environments for this crosstalk between ubiquitin and SUMOylation is a compelling example. Additionally, there is a growing body of work showing that other PTMs like phosphorylation also crosstalk with SUMOylation in biomolecular condensates to control critical cellular processes29, 30 and it will be exciting to see how much PTM crosstalk occurs in these compartments.
References
23. Enserink, J.M., Sumo and the cellular stress response. Cell Div, 2015. 10: p. 4.


