Introduction
Airway inflammation and intermittent narrowing of airways are key characteristics of the chronic lung condition known as asthma, which, due to an increasing prevalence, is now estimated to affect 300 million people worldwide(reviewed in 1). Standard therapeutic interventions for asthma include corticosteroids for managing chronic disease changes and β2‐adrenoreceptor agonists for acute asthma attacks. While they provide many benefits, these interventions do not correct all pathological changes to the remodeling airways associated with asthma. One of the key cellular targets in asthma is airway smooth muscle cells (ASMCs), whose contraction is controlled via Ca2+-dependent and Ca2+-sensitization mechanisms(reviewed in 2).As discussed in the review by Chiba et al., one of the primary mechanisms regulating Ca2+-sensitization in ASMC contraction is the small GTPase, RhoA.Recently, several studies have expanded our understanding of RhoA’s role in regulating asthma through airway hyperresponsiveness (AHR) and ASMC contraction, as well as through other asthma pathological features such as β‐adrenergic desensitization and airway inflammation(reviewed in [3]). This newsletter will discuss recent findings about the role of RhoA in the pathophysiology of asthma.
RhoA regulates ASMC hyperresponsiveness, contraction, and remodeling in asthma
The small GTPase RhoA functions as a molecular switch oscillating between an active state when bound to GTP and an inactive state when bound to GDP.Canonical RhoA signaling involves the recruitment and activation of the serine/threonine kinase, ROCK, which leads to downstream activation of myosin light chain, increased F-actin formation, and contraction.Early studies showed that antigen-induced AHR, an exaggerated and excessive narrowing of the airways in response to stimuli, resulted in elevated RhoA levels in mice and rats4,5.Importantly, treatment with a RhoA inhibitor, Clostridium botulinum C3 exoenzyme, blocked acetylcholine-induced AHR. A recent study found that the RhoGEF, Arhgef12, was highly expressed in patients with asthma, treatment of tracheal rings with RhoGEF inhibitors showed decreased contractility, and in an asthma model involving house dust mite allergic sensitization, Arhgef12-KO mice had significantly decreased AHR6. This study supports the role of RhoA signaling in AHR and identifies novel targets for potential intervention.
RhoA signaling has also been shown to affect other features of asthma, such as airway remodeling(reviewed in 3). This signaling pathway contributes to enhanced airway narrowing in asthma through a multitude of ways(reviewed in 3), but critically, through enhanced Ca2+-sensitization-induced contraction of ASMCs. Several signaling molecules have been shown to regulate RhoA-mediated ASMC contraction; for example, work by the Sakai lab recently identified the lncRNA, MALAT1, as a positive regulator of RhoA protein expression in ASMCs7. Mechanistically, they found that MALAT1 regulates RhoA by inhibiting miR-133a-3p in ASMCs to augment hypercontractility. Cytokine-driven activation of RhoA has also been shown to drive remodeling in asthma, and a recent study linked cytokines IL-13 and IL-17A to β1 integrin activation, which led to enhanced force transmission in ASMCs in a RhoA-dependent fashion8. Other force transmission and cytoskeletal fluidization mechanisms also contribute to ASMC contraction in asthma(reviewed in 9). In addition to augmenting ASMC contraction, RhoA also regulates airway remodeling phenotypes such as ASMC proliferation and migration in response to an array of stimulants, such as VEGF10 and kisspeptin11.
RhoA role in airway inflammation in asthma
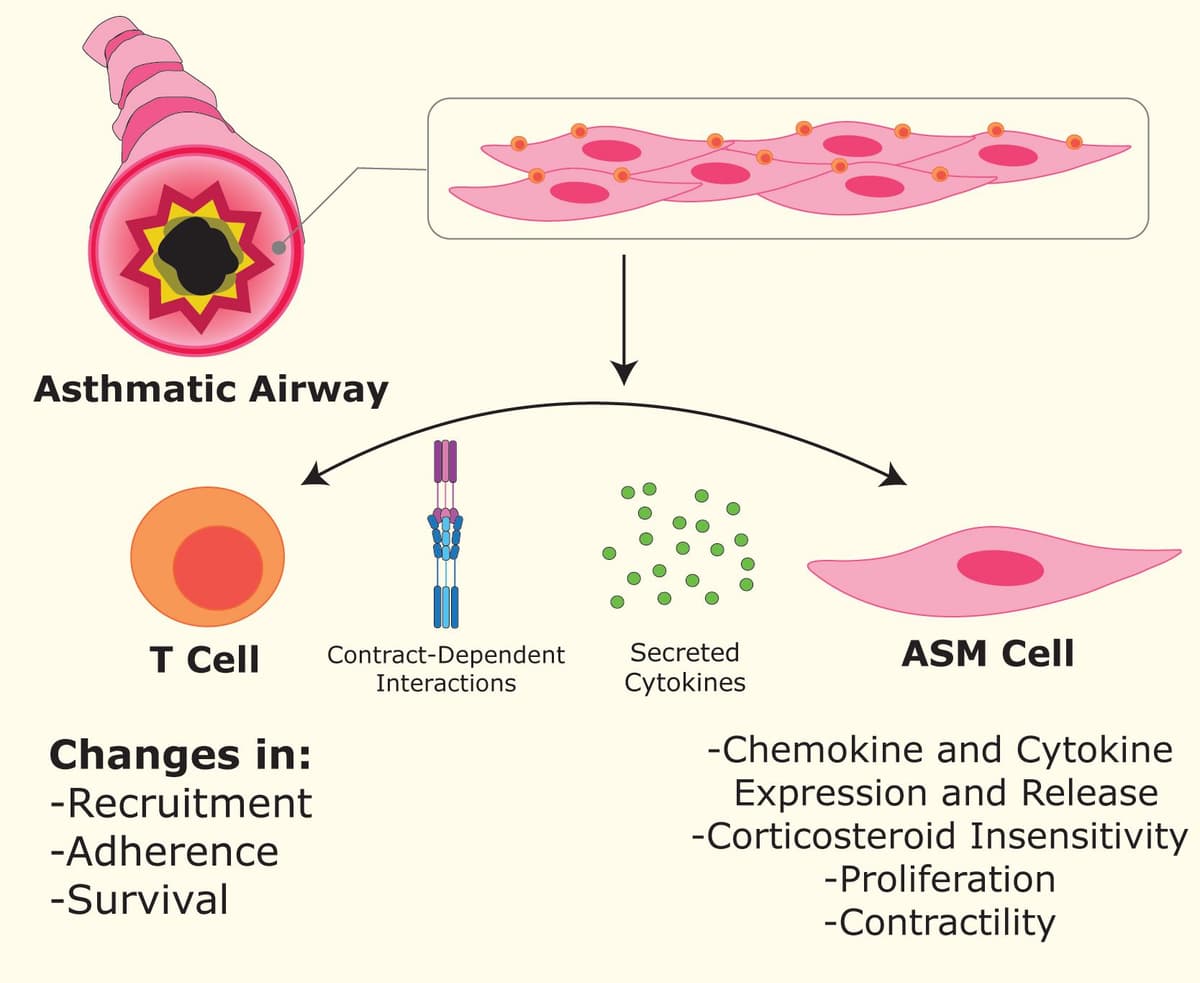
Airway inflammation involves the interplay among immune cells, epithelial cells, and ASMCs via various cytokines and mediators12; for instance, work by Ramos-Barbón et al. described how CD4+ T cells drive ASMC remodeling in experimental asthma13 (see figure 1). Another study also found significant crosstalk between CD4+ T cells and ASMCs, but in a pediatric obesity-related asthma model14. In addition to CD4+ T cells, eosinophils, mast cells, and other immune cells are augmented in asthmatic patients15. There is a body of work suggesting that the RhoA/ROCK signaling pathway can influence immune cell function in a multitude of disease settings(reviewed in 16). One recent example identified a paradoxical function for neutrophils, where they release anti-inflammatory extracellular vesicles, which are formed by RhoA-dependent budding17. Importantly, that was not in the setting of airway inflammation, but there are several specific examples where the RhoA/ROCK signaling does regulate immune cells to promote airway inflammation3.
For example, ROCK-depleted mice challenged with ovalbumin to produce an inflammatory response similar to asthma, displayed decreased IL-13 and IL-5 cytokine levels, lower eosinophil counts, and reduced AHR compared to wild-type mice18. In another study, RhoA levels were shown to be elevated in eosinophils of children with allergic asthma, and RhoA inactivation attenuated allergen-induced eosinophil infiltration into allergic airway-inflamed mouse lungs19.Work by the Zhang lab sought to deduce how obesity increases the severity of AHR in asthma, and identified the G-protein coupled receptor 40 (GPCR40) as a critical protein that responded to long-chain fatty acids to exacerbate several features of asthma20.Furthermore, inhibition of GPR40 with a small-molecule antagonist, DC260126, reduced TH2 cytokine levels, decreased inflammatory cell infiltration, and reduced AHR; all of these measurements also correlated with a downregulation in RhoA and ROCK in their model20.Collectively, these studies suggest that RhoA/ROCK signaling in immune cells is also important for controlling airway inflammation in asthma.
RhoA as a potential asthma therapeutic
While corticosteroids provide relief of chronic airway inflammation, they do not reverse all pathological effects associated with asthma.Targeting the RhoA/ROCK pathway in pre-clinical studies showed tremendous promise towards reversing acetylcholine or ovalbumin-AHR, and while the ROCK inhibitor Y-27632 was not as effective as a β2‐adrenoreceptor agonist for acute asthma treatment, it may be a viable additive therapy as it works by a completely different mechanism21. Several other ROCK inhibitor studies have shown positive results in attenuating risk factors of asthma, like airway mucus hypersecretion and inflammation, through the modulation of key cytokines22,23. A recent study corroborated these findings in another allergic asthma animal model, and found that the ROCK inhibitor hydroxyfasudil reduced the concentration of Th2 cytokine IL-13 while reducing inflammatory cell levels to provide improved airway remodeling24. In 2007, lovastatin was shown to reduce hyperresponsiveness in a rat model of asthma, and the benefit seen by lovastatin appeared to be RhoA-dependent25. Recently, the transgelin-2 targeting molecule, TSG1180, was shown to have beneficial effects for treating asthma, and in part occurred through altering the phosphorylation states of ROCK and RhoA26. One last example identified that the vitamin E isoform, β-tocotrienol molecule, was capable of inhibiting PDGF-BB-induced proliferation and migration of ASMCs, which it achieved through the inhibition of RhoA activation and ROS production27.
Summary and Future Insights
RhoA appears to play an integral role in several key pathologies associated with asthma and undoubtedly is a critical regulator of ASMCs and immune cells in the setting of airway inflammation, airway narrowing, and AHR. Remarkably, new data suggest that RhoA may even affect other cells involved in asthma, such as mesenchymal stem cells28. Several promising studies using ROCK inhibitors have shown a critical role for RhoA/ROCK signaling in asthma progression in pre-clinical studies, and it will be fascinating to see if viable RhoA/ROCK-targeting therapeutics eventually emerge for the treatment of asthma.
References
1. Dharmage, S.C., J.L. Perret, and A. Custovic, Epidemiology of Asthma in Children and Adults. Front Pediatr, 2019. 7: p. 246.
2. Chiba, Y., K. Matsusue, and M. Misawa, RhoA, a possible target for treatment of airway hyperresponsiveness in bronchial asthma. J Pharmacol Sci, 2010. 114(3): p. 239-47.
3. Zhang, Y., et al., RhoA/Rho-kinases in asthma: from pathogenesis to therapeutic targets. Clin Transl Immunology, 2020. 9(5): p. e01134.
4. Chiba, Y., et al., Augmented acetylcholine-induced, Rho-mediated Ca2+ sensitization of bronchial smooth muscle contraction in antigen-induced airway hyperresponsive rats. Br J Pharmacol, 1999. 127(3): p. 597-600.
5. Chiba, Y., et al., Involvement of RhoA-mediated Ca2+ sensitization in antigen-induced bronchial smooth muscle hyperresponsiveness in mice. Respir Res, 2005. 6(1): p. 4.
6. Fong, V., et al., Arhgef12 drives IL17A-induced airway contractility and airway hyperresponsiveness in mice. JCI Insight, 2018. 3(21).
7. Chiba, Y., et al., A lncRNA MALAT1 is a positive regulator of RhoA protein expression in bronchial smooth muscle cells. Life Sci, 2023. 313: p. 121289.
8. Ngo, U., et al., IL-13 and IL-17A activate beta1 integrin through an NF-kB/Rho kinase/PIP5K1gamma pathway to enhance force transmission in airway smooth muscle. Proc Natl Acad Sci U S A, 2024. 121(34): p. e2401251121.
9. McCullough, M., et al., Targeting cytoskeletal biomechanics to modulate airway smooth muscle contraction in asthma. J Biol Chem, 2025. 301(1): p. 108028.
10. Lv, C., et al., Vascular endothelial growth factor induces the migration of human airway smooth muscle cells by activating the RhoA/ROCK pathway. BMC Pulm Med, 2023. 23(1): p. 505.
11. Balraj, P., et al., Kisspeptin/KISS1R Signaling Modulates Human Airway Smooth Muscle Cell Migration. Am J Respir Cell Mol Biol, 2024. 70(6): p. 507-518.
12. Kume, H., Role of Airway Smooth Muscle in Inflammation Related to Asthma and COPD. Adv Exp Med Biol, 2021. 1303: p. 139-172.
13. Ramos-Barbon, D., et al., Antigen-specific CD4+ T cells drive airway smooth muscle remodeling in experimental asthma. J Clin Invest, 2005. 115(6): p. 1580-9.
14. Yon, C., et al., Crosstalk between CD4(+) T Cells and Airway Smooth Muscle in Pediatric Obesity-related Asthma. Am J Respir Crit Care Med, 2023. 207(4): p. 461-474.
15. Ying, S., et al., Expression of IL-4 and IL-5 mRNA and protein product by CD4+ and CD8+ T cells, eosinophils, and mast cells in bronchial biopsies obtained from atopic and nonatopic (intrinsic) asthmatics. J Immunol, 1997. 158(7): p. 3539-44.
16. Bros, M., et al., RhoA as a Key Regulator of Innate and Adaptive Immunity. Cells, 2019. 8(7).
17. Hsu, A.Y., et al., Neutrophil-derived vesicles control complement activation to facilitate inflammation resolution. Cell, 2025. 188(6): p. 1623-1641 e26.
18. Zhu, M., et al., Role of Rho kinase isoforms in murine allergic airway responses. Eur Respir J, 2011. 38(4): p. 841-50.
19. Xu, C., et al., Protein tyrosine phosphatase 11 acts through RhoA/ROCK to regulate eosinophil accumulation in the allergic airway. FASEB J, 2019. 33(11): p. 11706-11720.
20. Lin, X., et al., Targeting of G-protein coupled receptor 40 alleviates airway hyperresponsiveness through RhoA/ROCK1 signaling pathway in obese asthmatic mice. Respir Res, 2023. 24(1): p. 56.
21. Iizuka, K., et al., Evaluation of Y-27632, a rho-kinase inhibitor, as a bronchodilator in guinea pigs. Eur J Pharmacol, 2000. 406(2): p. 273-9.
22. Xie, T., et al., Rho-kinase inhibitor fasudil reduces allergic airway inflammation and mucus hypersecretion by regulating STAT6 and NFkappaB. Clin Exp Allergy, 2015. 45(12): p. 1812-22.
23. Zhang, W., X. Li, and Y. Zhang, Rho-kinase inhibitor attenuates airway mucus hypersecretion and inflammation partly by downregulation of IL-13 and the JNK1/2-AP1 signaling pathway. Biochem Biophys Res Commun, 2019. 516(2): p. 571-577.
24. Franova, S., et al., The anti-asthmatic potential of Rho-kinase inhibitor hydroxyfasudil in the model of experimentally induced allergic airway inflammation. Eur J Pharmacol, 2023. 938: p. 175450.
25. Chiba, Y., et al., Increase in the expression of matrix metalloproteinase-12 in the airways of rats with allergic bronchial asthma. Biol Pharm Bull, 2007. 30(2): p. 318-23.
26. Yuan, H.K., et al., Discovery of zolinium TSG1180 as a novel agonist of transgelin-2 for treating asthma. Biomed Pharmacother, 2023. 167: p. 115556.
27. Listyoko, A.S., et al., beta-Tocotrienol Decreases PDGF-BB-Induced Proliferation and Migration of Human Airway Smooth Muscle Cells by Inhibiting RhoA and Reducing ROS Production. Pharmaceuticals (Basel), 2024. 17(6).
28. Ke, X., et al., Ras homolog family member A/Rho-associated protein kinase 1 signaling modulates lineage commitment of mesenchymal stem cells in asthmatic patients through lymphoid enhancer-binding factor 1. J Allergy Clin Immunol, 2019. 143(4): p. 1560-1574 e6.


