Introduction
Glucose stimulates insulin release from pancreatic β cells in a biphasic manner. The initial rapid and short-lived release – facilitated by Large Dense Core Vesicles (LDCVs) at the plasma membrane – is followed by a sustained release of insulin that depends on the mobilization of insulin-secreting granules (ISGs) to the plasma membrane for exocytosis1. Actin dynamics are critical for both phases of insulin release2,3. In the first phase, depolymerization of the filamentous subcortical actin barrier (F-actin) is required to rapidly release LDCVs2. In the second phase, ISGs are transported along microtubules and then transferred to F-actin to transport ISGs from microtubules to the plasma membrane for exocytosis2. Thus, rapid depolymerization for phase I is followed by a requirement for actin polymerization to facilitate sustained ISG release in phase II.
Glucagon-like peptide-1(GLP-1) is a 30 amino acid-long hormone secreted by enteroendocrine L cells that mediates glucose homeostasis4. GLP-1 and analogs have been used to treat Type II diabetes mellitus5. GLP-1 maintains glucose levels by stimulating pancreatic β cells to produce insulin, slowing gastric emptying, and decreasing glucagon release4. The role of GLP-1 in insulin secretion is actin-dependent, and the relationship between actin polymerization and GLP-1 is bidirectional. GLP-1 modulates actin polymerization, influencing insulin secretion. In turn, appropriate F-actin remodeling is required for GLP-1 exocytosis. This newsletter reviews the research that has uncovered this bidirectional relationship and actin's role in glucose homeostasis with regard to endogenous GLP-1 and therapeutic GLP-1 receptor agonists.

GLP-1 modulates actin polymerization, improving insulin sensitivity and glucose-stimulated insulin secretion
Chronic hyperglycemia, or glucotoxicity, causes pancreatic β cell dysfunction, diminished glucose-stimulated insulin secretion, and the production of long actin stress fibers6,7. Nevins and Thurmond demonstrated that β cells formed filamentous F-actin associated with the t-SNARE proteins syntaxin 1A and SNAP-25 in response to glucose, slowing insulin release8, a finding later confirmed by Quinault et al. in another β cell line7. They also demonstrated that glucotoxicity resulted in β cells that were less responsive to glucose, blunting glucose-induced actin dynamics and impeding ISG fusion7. F-actin, organized into a dense actin barrier at the plasma membrane, impedes rapid exocytosis of insulin-containing LDCVs3. Depolymerization of F-actin restores β cell glucose-stimulated insulin secretion after glucotoxicity6.
GLP-1 promotes the disassembly of F-actin, improving β cell glucose sensitivity and insulin secretion following sustained glucotoxicity7. Several studies have reported mechanisms by which GLP-1 leads to this disassembly. One such study found that GLP-1 elevates intracellular cAMP, subsequently activating protein kinase A (PKA), which in turn inhibits RhoA/ROCK signaling, resulting in the disassembly of glucotoxicity-induced stress fibers6. Another study determined that GLP-1 could restore insulin secretion via modulation of actin polymerization, independent of a recently identified role for the mammalian target of rapamycin complex 2 (mTORC2) in actin modulation and insulin sensitivity9. In addition to depolymerizing stress fibers and dismantling the actin barrier, which impedes the rapid release of insulin during the first phase of insulin secretion, GLP-1 impacts the second prolonged phase, including ISG fusion7.
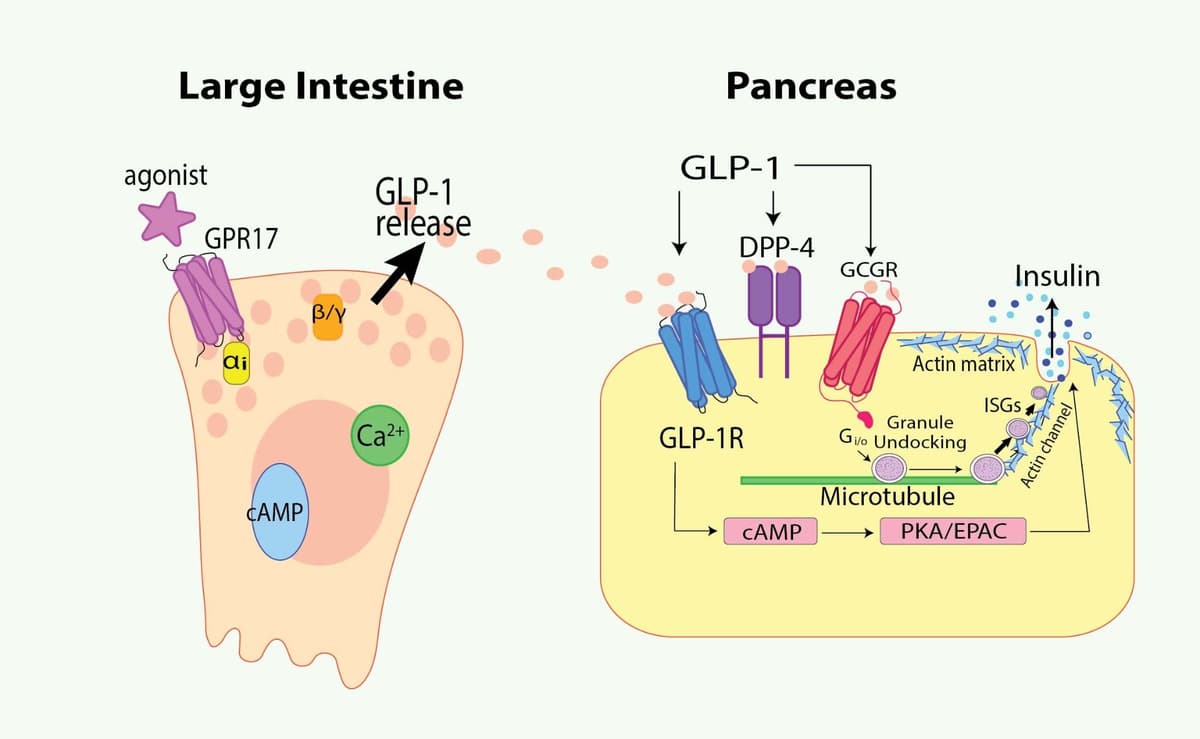
Actin cytoskeletal dynamics required for GLP-1 exocytosis
GLP-1 is secreted by enteroendocrine L cells in response to food intake in a calcium-dependent manner10,11. In response to glucose and membrane depolarization, voltage-dependent Ca2+ channels open. The resulting influx of Ca2+ causes vesicular exocytosis of GLP-1-containing vesicles. Just as actin dynamics are critical for the biphasic secretion of insulin and the exocytosis of insulin-containing granules from β cells, they are equally essential for biphasic GLP-1 exocytosis from L cells12. In this way, GLP-1 and actin polymerization play bidirectional regulatory roles. A 2023 study determined that the docking dynamics of GLP-1 granules are regulated by F-actin polymerization12. Actin is required for transport to the plasma membrane and fusion of GLP-1 secretory granules.
Summary
Together, these findings demonstrate that elevated glucose results in actin polymerization that impedes both phases of biphasic insulin secretion, and that GLP-1 can restore glucose sensitivity and promote insulin release by correcting errors in actin dynamics at both phases. At the same time, actin dynamics are essential for exocytosis of GLP-1 granules, creating a regulatory feedback loop between the actin cytoskeleton and GLP-1. GLP-1 receptor agonists mimic GLP-1, lowering blood glucose by stimulating insulin secretion, slowing gastric emptying, and decreasing glucagon release13. GLP-1-based therapeutics have also been shown to lower the risk of neurodegenerative diseases and improve cardiovascular health, in addition to weight management and diabetes treatment10. The roles of GLP-1 with regard to actin dynamics in cardiovascular and neurodegenerative disease processes have not yet been well established.
References
1. Ashcroft FM, Rorsman P. Diabetes mellitus and the beta cell: the last ten years. Cell. 2012;148(6):1160-71. Epub 2012/03/20. doi: 10.1016/j.cell.2012.02.010. PubMed PMID: 22424227; PMCID: PMC5890906.
2. Li W, Li A, Yu B, Zhang X, Liu X, White KL, Stevens RC, Baumeister W, Sali A, Jasnin M, Sun L. In situ structure of actin remodeling during glucose-stimulated insulin secretion using cryo-electron tomography. Nat Commun. 2024;15(1):1311. Epub 2024/02/13. doi: 10.1038/s41467-024-45648-7. PubMed PMID: 38346988; PMCID: PMC10861521.
3. Wang Z, Thurmond DC. Mechanisms of biphasic insulin-granule exocytosis - roles of the cytoskeleton, small GTPases and SNARE proteins. J Cell Sci. 2009;122(Pt 7):893-903. Epub 2009/03/20. doi: 10.1242/jcs.034355. PubMed PMID: 19295123; PMCID: PMC2720925.
4. Nadkarni P, Chepurny OG, Holz GG. Regulation of glucose homeostasis by GLP-1. Prog Mol Biol Transl Sci. 2014;121:23-65. Epub 2014/01/01. doi: 10.1016/B978-0-12-800101-1.00002-8. PubMed PMID: 24373234; PMCID: PMC4159612.
5. Ahren B. GLP-1 for type 2 diabetes. Exp Cell Res. 2011;317(9):1239-45. Epub 2011/01/18. doi: 10.1016/j.yexcr.2011.01.010. PubMed PMID: 21237153.
6. Kong X, Yan D, Sun J, Wu X, Mulder H, Hua X, Ma X. Glucagon-like peptide 1 stimulates insulin secretion via inhibiting RhoA/ROCK signaling and disassembling glucotoxicity-induced stress fibers. Endocrinology. 2014;155(12):4676-85. Epub 2014/09/23. doi: 10.1210/en.2014-1314. PubMed PMID: 25243854.
7. Quinault A, Gausseres B, Bailbe D, Chebbah N, Portha B, Movassat J, Tourrel-Cuzin C. Disrupted dynamics of F-actin and insulin granule fusion in INS-1 832/13 beta-cells exposed to glucotoxicity: partial restoration by glucagon-like peptide 1. Biochim Biophys Acta. 2016;1862(8):1401-11. Epub 2016/04/23. doi: 10.1016/j.bbadis.2016.04.007. PubMed PMID: 27101990.
8. Nevins AK, Thurmond DC. Glucose regulates the cortical actin network through modulation of Cdc42 cycling to stimulate insulin secretion. Am J Physiol Cell Physiol. 2003;285(3):C698-710. Epub 2003/05/23. doi: 10.1152/ajpcell.00093.2003. PubMed PMID: 12760905.
9. Blandino-Rosano M, Scheys JO, Werneck-de-Castro JP, Louzada RA, Almaca J, Leibowitz G, Ruegg MA, Hall MN, Bernal-Mizrachi E. Novel roles of mTORC2 in regulation of insulin secretion by actin filament remodeling. Am J Physiol Endocrinol Metab. 2022;323(2):E133-E44. Epub 2022/06/21. doi: 10.1152/ajpendo.00076.2022. PubMed PMID: 35723227; PMCID: PMC9291412.
10. Kuhre RE, Deacon CF, Holst JJ, Petersen N. What Is an L-Cell and How Do We Study the Secretory Mechanisms of the L-Cell? Front Endocrinol (Lausanne). 2021;12:694284. Epub 2021/06/26. doi: 10.3389/fendo.2021.694284. PubMed PMID: 34168620; PMCID: PMC8218725.
11. Muller TD, Finan B, Bloom SR, D'Alessio D, Drucker DJ, Flatt PR, Fritsche A, Gribble F, Grill HJ, Habener JF, Holst JJ, Langhans W, Meier JJ, Nauck MA, Perez-Tilve D, Pocai A, Reimann F, Sandoval DA, Schwartz TW, Seeley RJ, Stemmer K, Tang-Christensen M, Woods SC, DiMarchi RD, Tschop MH. Glucagon-like peptide 1 (GLP-1). Mol Metab. 2019;30:72-130. Epub 2019/11/27. doi: 10.1016/j.molmet.2019.09.010. PubMed PMID: 31767182; PMCID: PMC6812410.
12. Harada K, Takashima M, Kitaguchi T, Tsuboi T. F-actin determines the time-dependent shift in docking dynamics of glucagon-like peptide-1 granules upon stimulation of secretion. FEBS Lett. 2023;597(5):657-71. Epub 2023/01/25. doi: 10.1002/1873-3468.14580. PubMed PMID: 36694275.
13. Collins L, Costello RA. Glucagon-Like Peptide-1 Receptor Agonists. StatPearls. 2024 Feb 29. PubMed PMID: 31855395.


