Introduction
The aging brain, characterized by aberrant neuronal network activity, inflammation, oxidative damage, impaired adaptive neuroplasticity, and compromised bioenergetics, is susceptible to several neurological diseases, including Alzheimer’s, Parkinson’s, and stroke(reviewed in 1).Several hallmarks of brain aging, such as aberrant neuronal network activity, mitochondrial dysfunction, and others, may be regulated by cytoskeletal function(reviewed in 2); however, a comprehensive understanding of how the cytoskeleton contributes to the aging brain is not well defined. The microtubule (MT) cytoskeleton is abundant in neurons and is an essential component of axons and dendrites(reviewed in 3). MTs form parallel bundles in axons to provide a structural backbone, while also functioning as long-distance railways for protein and organelle transport. Throughout the life of a neuron, MTs support changes in shape and transport that are important for cognitive plasticity and neuronal activity; thus, it is likely that MTs participate in the physiological aging of neurons. A recent study performed brain-wide cell-type-specific transcriptomic signatures in young versus old mice and found age-dependent decreases in genes associated with MT-based movement in tanycytes (specialized glial cells), providing evidence that MT adaptations are present in the aging brain4. This newsletter examines the role of MTs and key regulatory mechanisms in aging neurons and Alzheimer’s disease.

The Microtubule Impact on Neuronal Aging
In the aging brain, the ability of neurons to maintain their structure, which is dependent on the cytoskeleton, becomes functionally compromised; for example, there is evidence of age-related decreases in synaptic spine density, reductions in soma size, and alterations in dendrite and axon lengths5. Yet, a complete understanding of what happens to MTs during physiological aging in neurons is lacking due to limitations in experimental models for studying normal physiological aging. An early study identified a 55% reduction in microtubule density in human cortical pyramidal cell dendrites from individuals between 62- 80 years old6, suggesting that MT levels are depleted due to aging.This finding was supported by a study in rats, which showed reduced β-tubulin staining in dendrites from aged rats7.MT changes in axons are a bit more confusing, as some studies allude to a reduction in MT levels, while work on human peripheral sensory neurons reported an increase in cytoskeletal proteins with age8.With many of these examples, a reduction in MTs coincides with a reduced cellular compartment; therefore, it is difficult to discern if MT reduction leads to smaller cell compartments or if smaller cell compartments lead to MT reduction. Recently, work by Okenve-Ramos et al. utilized a Drosophila brain model to investigate the consequences of physiological aging9.Interestingly, they found that neuronal MT decay preceded the onset of other aging hallmarks like declines in axons and synaptic terminals. MT binding proteins like tau and end-binding protein 1 (EB1) were shown to be important for maintaining MT integrity, and genetic manipulation to improve MT networks was sufficient to slow the onset of other neuronal aging hallmarks [9]. In a follow-up study, the group investigated the interplay between oxidative stress, neuronal aging, and MTs10.They found that oxidative stress targets the MT binding protein EB1, which leads to MT defects and morphological transformation of axonal terminals during aging. Other studies have defined upstream regulatory mechanisms as well; for example, the Hippo pathway signaling pathway was identified as a key regulator of MT integrity in normally aged neurons from a C. elegans model11.Defining the role of MTs in neuronal aging may provide helpful strategies to enhance neuronal resilience and fitness during physiological aging and in age-related neurodegenerative diseases.
Microtubule Alterations in Aging Neurons – A Focus on Acetylation
MTs are formed by polymerizing α- and β-tubulin heterodimers which are regulated by MT-binding proteins, post-translational modifications (PTMs), and tubulin isoforms. These regulatory mechanisms contribute to MT heterogeneity across cellular compartments, and recent studies have focused on how they impact neuronal aging(reviewed in 5,12).Specifically, they identified tubulin acetylation and tau dysregulation as critical mechanisms contributing to aging in neurons. Interestingly, these mechanisms also contribute to microtubule stabilization in senescent intestinal epithelial cells13 providing support to the idea that these may be common molecular mechanisms that lead to augmented microtubule function in aging cells.
Tubulin acetylation of α-tubulin on the luminal surface of microtubules has been classically associated with long-lived, stable microtubules. The role of acetylation in aging neurons is still controversial, as early studies linked decreased tubulin acetylation to defective axonal transport and progression of neurodegenerative disorders like Alzheimer’s disease (AD)14,15. In support of these findings, the Sirtuin 2.3 isoform, which is a microtubule deacetylase, was shown to have accumulated in the aged mouse brain and spinal cord16. Conversely, a recent study showed that tubulin acetylation levels were elevated in post-mortem brain tissues from AD patients17, while a preprint by Shen et al. shows that hyperacetylation of microtubules acts as a biomarker during cellular senescence18. Collectively, these studies suggest that changes in tubulin acetylation occur in aging neurons; however, whether an increase or decrease in tubulin acetylation is a driving factor in neuronal aging still needs more investigation. Donker and Godinho, recently highlighted new evidence suggesting that tubulin acetylation can also occur as a dynamic modification and is a highly active stress marker that allows for cell adaptation, and may explain why we observe its accumulation in aged cells19.
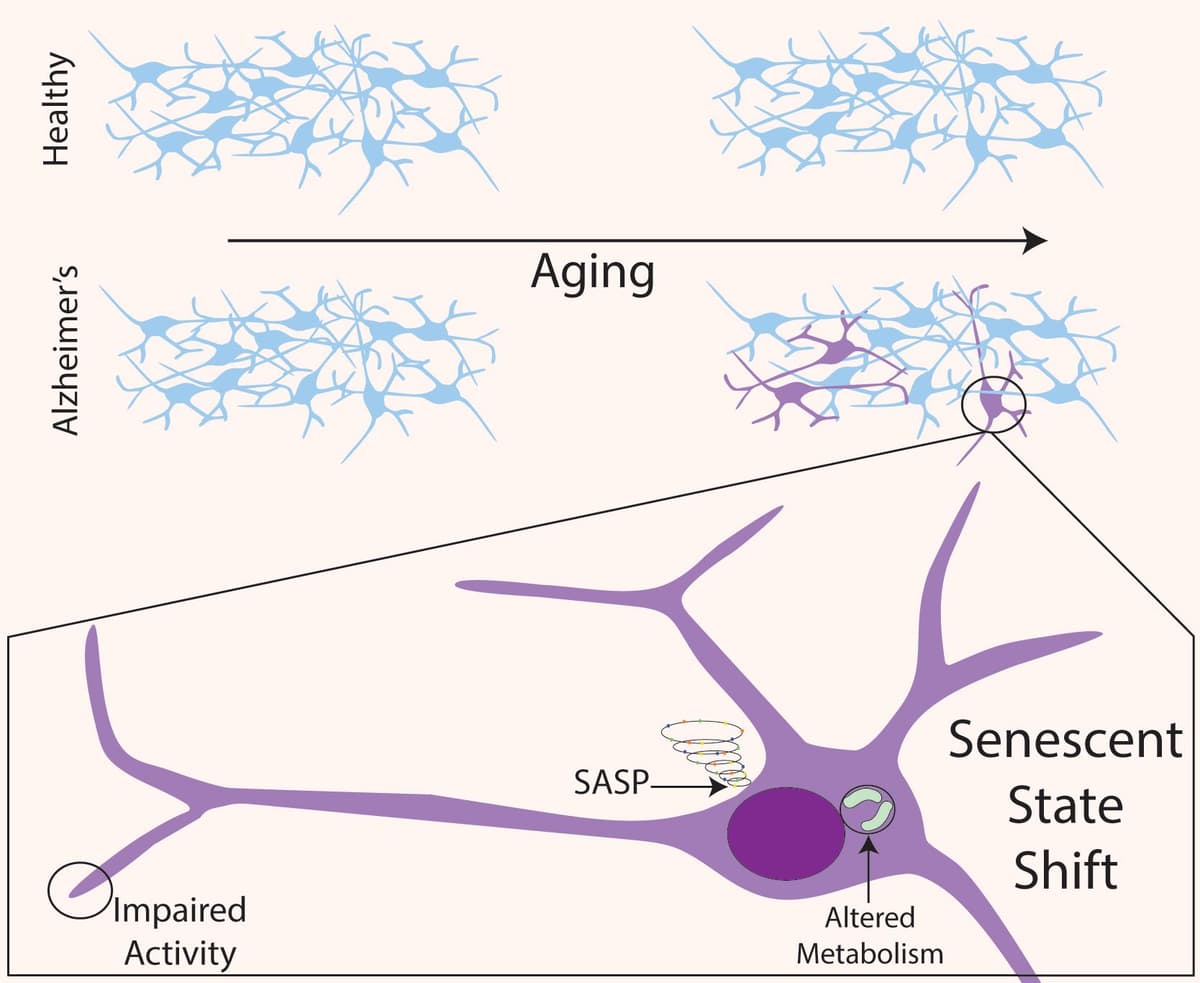
Neuronal Senescence in Alzheimer’s Disease
Senescence is an apoptosis-resistant, nonproliferative state that cells adapt to in response to stress, molecular damage, and proliferative exhaustion (reviewed in20.Senescent cells have distinct molecular profiles and may perform differently from their pre-senescent state; tissue dysfunction may occur once a critical mass of cells becomes senescent. Importantly, neurons, which are post-mitotic cells, have been shown to undergo senescence21. AD, a neurological disorder that results in dementia in older patients, is strongly linked to brain aging, with hallmarks of neurological aging having been observed in AD patients and animal models(reviewed in 22).Recently, two groups found that AD brains have higher proportions of neurons that express senescence markers23,24. They found that senescent neurons gain an inflammatory, senescence-associated secretory phenotype (see figure).
Several cytoskeletal changes associated with the aging brain are often also present in AD, the most obvious being hyperphosphorylation of tau. In both aging and AD, there is an increase in tau phosphorylation and misregulation that can lead to MT destabilization(reviewed in [5]). One of the groups that found senescent neurons in AD brains also showed that the senescent neurons contained neurofibrillary tangle tau pathology24. However, tau is not the only MT-associated protein that affects AD, as the kinesin, KIF9, was also shown to contribute to AD through regulating the transport of lysosomes presumably along MTs25.These data collectively suggest that regulation of MTs through altered MT-associated protein function has an impact on AD progression. However, can directly manipulating MTs alter AD progression? The premise that destabilized MTs contribute to AD was tested by treating AD mice with MT-stabilizing drugs26,27. Interestingly, a brain-penetrating MT-stabilizing drug improved cognitive function while ameliorating neuritic pathology, which suggests that MT dysfunction is a contributing factor in AD.
Summary and Future Perspective
These studies reveal an underlying common theme pointing to the importance of MTs in aging neuronal cells that can contribute to neurodegenerative disease progression like AD. Furthermore, regulatory mechanisms like PTMs and MT-associated proteins modify MTs in aging cells. It is not clear whether these events are adaptations to other stress events or causal mechanisms that drive MTs to a dysfunctional state that contributes to disease progression and thus warrant further investigation. A related area of interest to MTs in neuronal aging is how MTs contribute to axon regeneration in injured neurons. A recent study by Costa et al. utilized a novel in vitro model for dorsal root ganglion axonal asymmetry and found that the peripheral and central axon branches have distinct MT population patterns that produce differential regenerative capacities, and were regulated by key MT-regulatory proteins like tau,spastin, and CRMP528.These studies cooperatively highlight how important MTs are for neuronal cell form and function throughout their lifespan.
References
1. Mattson, M.P. and T.V. Arumugam, Hallmarks of Brain Aging: Adaptive and Pathological Modification by Metabolic States. Cell Metab, 2018. 27(6): p. 1176-1199.
2. Zia, A., et al., Molecular and cellular pathways contributing to brain aging. Behav Brain Funct, 2021. 17(1): p. 6.
3. Pena-Ortega, F., A.A. Robles-Gomez, and L. Xolalpa-Cueva, Microtubules as Regulators of Neural Network Shape and Function: Focus on Excitability, Plasticity and Memory. Cells, 2022. 11(6).
4. Jin, K., et al., Brain-wide cell-type-specific transcriptomic signatures of healthy ageing in mice. Nature, 2025. 638(8049): p. 182-196.
5. Richardson, B., et al., How do neurons age? A focused review on the aging of the microtubular cytoskeleton. Neural Regen Res, 2024. 19(9): p. 1899-1907.
6. Cash, A.D., et al., Microtubule reduction in Alzheimer's disease and aging is independent of tau filament formation. Am J Pathol, 2003. 162(5): p. 1623-7.
7. Filipek, A., et al., Age-dependent changes in neuronal distribution of CacyBP/SIP: comparison to tubulin and the tau protein. J Neural Transm (Vienna), 2008. 115(9): p. 1257-64.
8. Metzner, K., et al., Age-dependent increase of cytoskeletal components in sensory axons in human skin. Front Cell Dev Biol, 2022. 10: p. 965382.
9. Okenve-Ramos, P., et al., Neuronal ageing is promoted by the decay of the microtubule cytoskeleton. PLoS Biol, 2024. 22(3): p. e3002504.
10. Shields, S., et al., Oxidative Stress Promotes Axonal Atrophy through Alterations in Microtubules and EB1 Function. Aging Dis, 2025.
11. Lee, H., et al., Neuroprotective role of Hippo signaling by microtubule stability control in Caenorhabditis elegans. Elife, 2025. 13.
12. Brill, M.S., C. Fassier, and Y. Song, Editorial: Cytoskeletal alterations in aging and disease. Front Cell Dev Biol, 2023. 11: p. 1359465.
13. Chu, S., et al., Multiple pathways promote microtubule stabilization in senescent intestinal epithelial cells. NPJ Aging, 2022. 8(1): p. 16.
14.Dompierre, J.P., et al., Histone deacetylase 6 inhibition compensates for the transport deficit in Huntington's disease by increasing tubulin acetylation. J Neurosci, 2007. 27(13): p. 3571-83.
15. Li, L., et al., MEC-17 deficiency leads to reduced alpha-tubulin acetylation and impaired migration of cortical neurons. J Neurosci, 2012. 32(37): p. 12673-83.
16. Maxwell, M.M., et al., The Sirtuin 2 microtubule deacetylase is an abundant neuronal protein that accumulates in the aging CNS. Hum Mol Genet, 2011. 20(20): p. 3986-96.
17. Martinez-Hernandez, J., et al., Crosstalk between acetylation and the tyrosination/detyrosination cycle of alpha-tubulin in Alzheimer's disease. Front Cell Dev Biol, 2022. 10: p. 926914.
18. Shen, Y., et al., Microtubule acetylation is a biomarker of cytoplasmic health during cellular senescence. bioRxiv, 2025.
19. Donker, L. and S.A. Godinho, Rethinking tubulin acetylation: From regulation to cellular adaptation. Curr Opin Cell Biol, 2025. 94: p. 102512.
20. Herdy, J.R., J. Mertens, and F.H. Gage, Neuronal senescence may drive brain aging. Science, 2024. 384(6703): p. 1404-1406.
21. Paramos-de-Carvalho, D., et al., Targeting senescent cells improves functional recovery after spinal cord injury. Cell Rep, 2021. 36(1): p. 109334.
22. Liu, Y., et al., The interaction between ageing and Alzheimer's disease: insights from the hallmarks of ageing. Transl Neurodegener, 2024. 13(1): p. 7.
23. Herdy, J.R., et al., Increased post-mitotic senescence in aged human neurons is a pathological feature of Alzheimer's disease. Cell Stem Cell, 2022. 29(12): p. 1637-1652 e6.
24. Dehkordi, S.K., et al., Profiling senescent cells in human brains reveals neurons with CDKN2D/p19 and tau neuropathology. Nat Aging, 2021. 1(12): p. 1107-1116.
25. Wang, M., et al., KIF9 Ameliorates Neuropathology and Cognitive Dysfunction by Promoting Macroautophagy in a Mouse Model of Alzheimer's Disease. Aging Cell, 2025: p. e14490.
26. Fernandez-Valenzuela, J.J., et al., Enhancing microtubule stabilization rescues cognitive deficits and ameliorates pathological phenotype in an amyloidogenic Alzheimer's disease model. Sci Rep, 2020. 10(1): p. 14776.
27. Brunden, K.R., et al., Epothilone D improves microtubule density, axonal integrity, and cognition in a transgenic mouse model of tauopathy. J Neurosci, 2010. 30(41): p. 13861-6.
28. Costa, A.C., et al., Axon-specific microtubule regulation drives asymmetric regeneration of sensory neuron axons. Elife, 2025. 13.


