CN04, part of the G-Switch™ line, is derived from the catalytic domain of bacterial CNF toxins and linked to a proprietary cell-penetrating moiety. It directly activates Rho, Rac, and Cdc42 GTPases by deamidating glutamine-63 (Rho) and glutamine-61 (Rac & Cdc42) in the Switch II region, converting it to glutamate and blocking GTPase activity, resulting in constitutive GTPase activation. Produced in a bacterial expression system, CN04 is a 118 kDa His-tagged recombinant protein, supplied as 20 µg per vial.
Preparation & Use:
Protein purity is assessed by scanning densitometry of Coomassie Blue-stained protein on a 4-20% polyacrylamide gel. Purity was determined to be >80%.
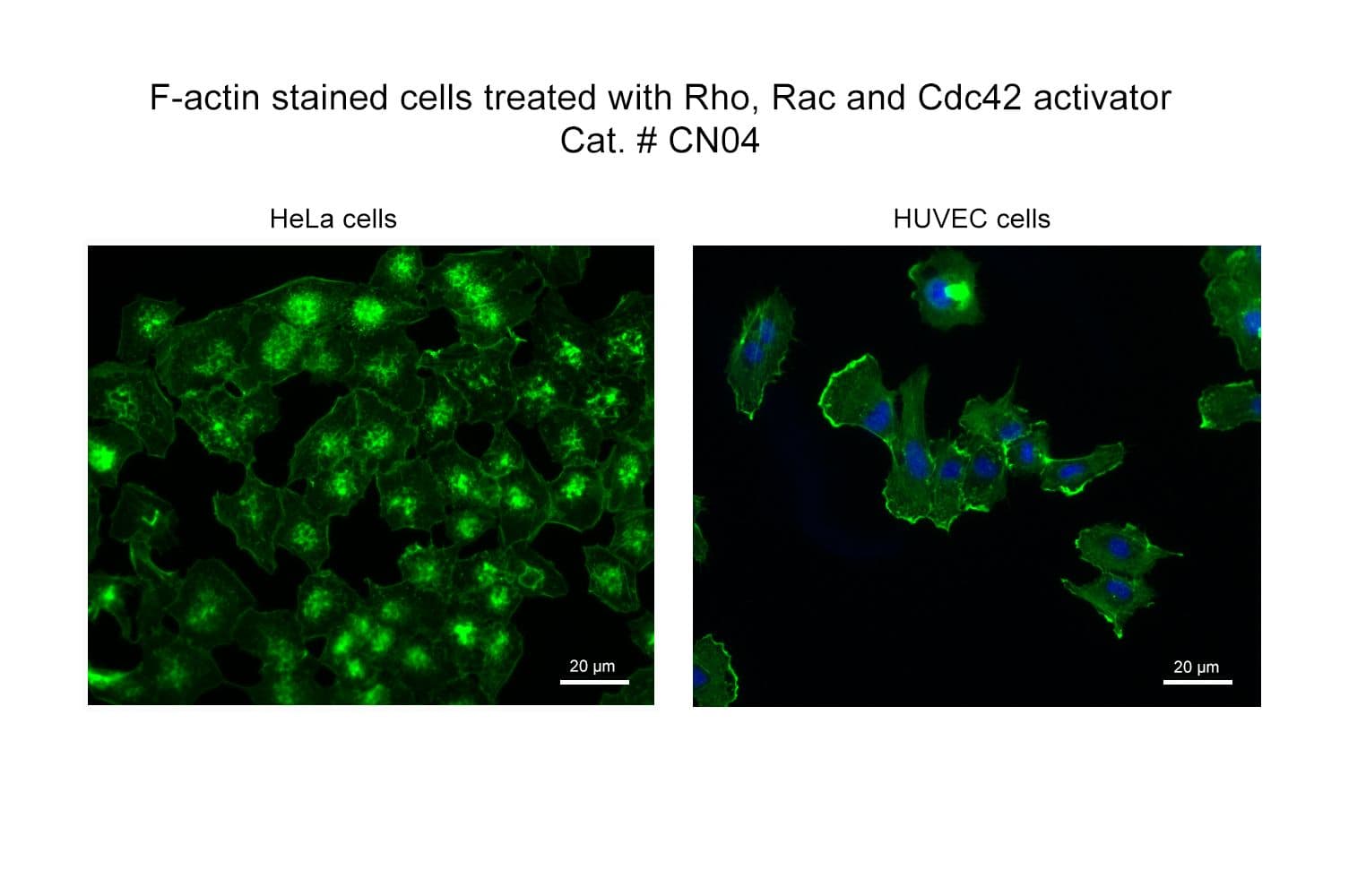
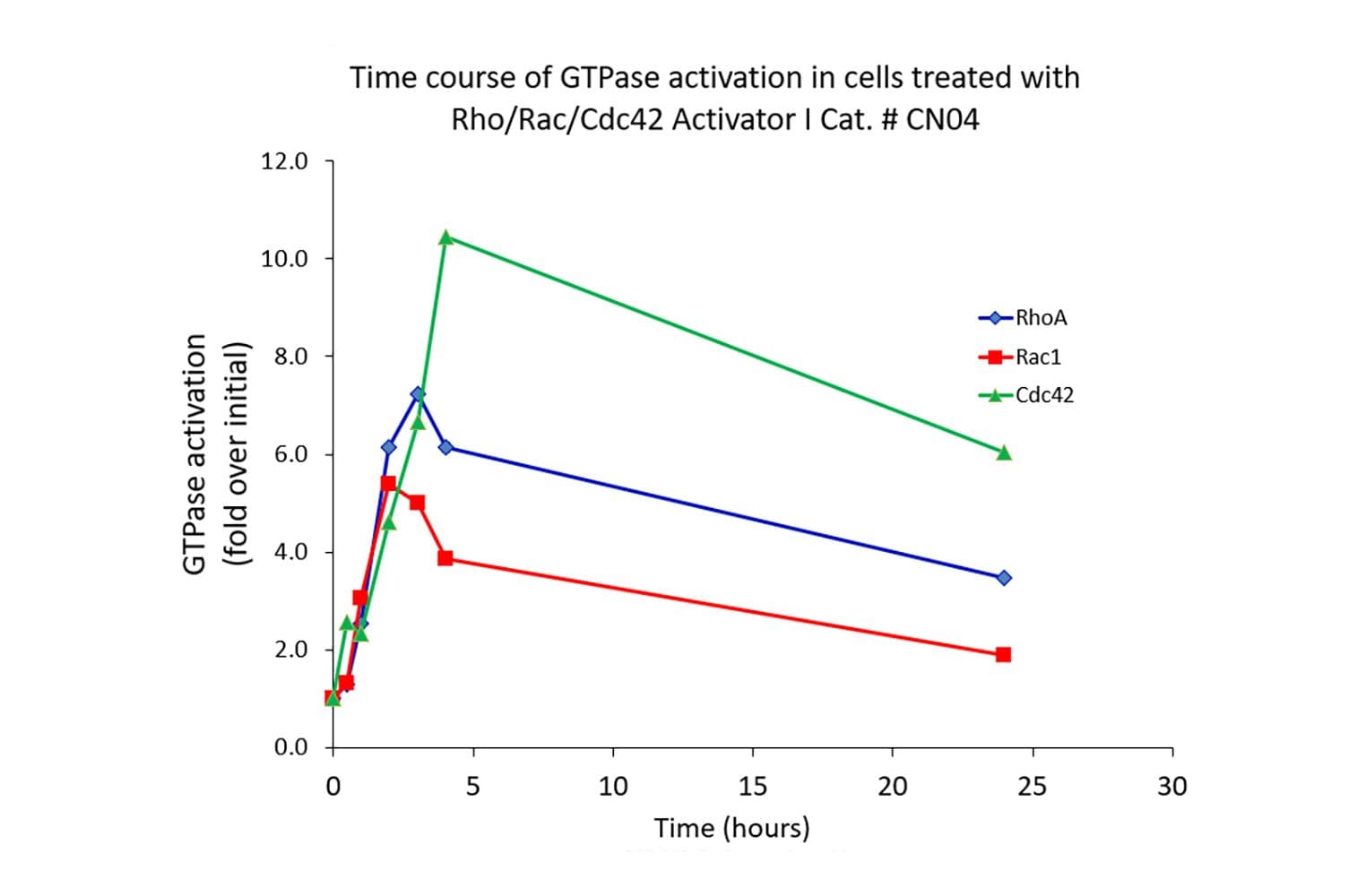
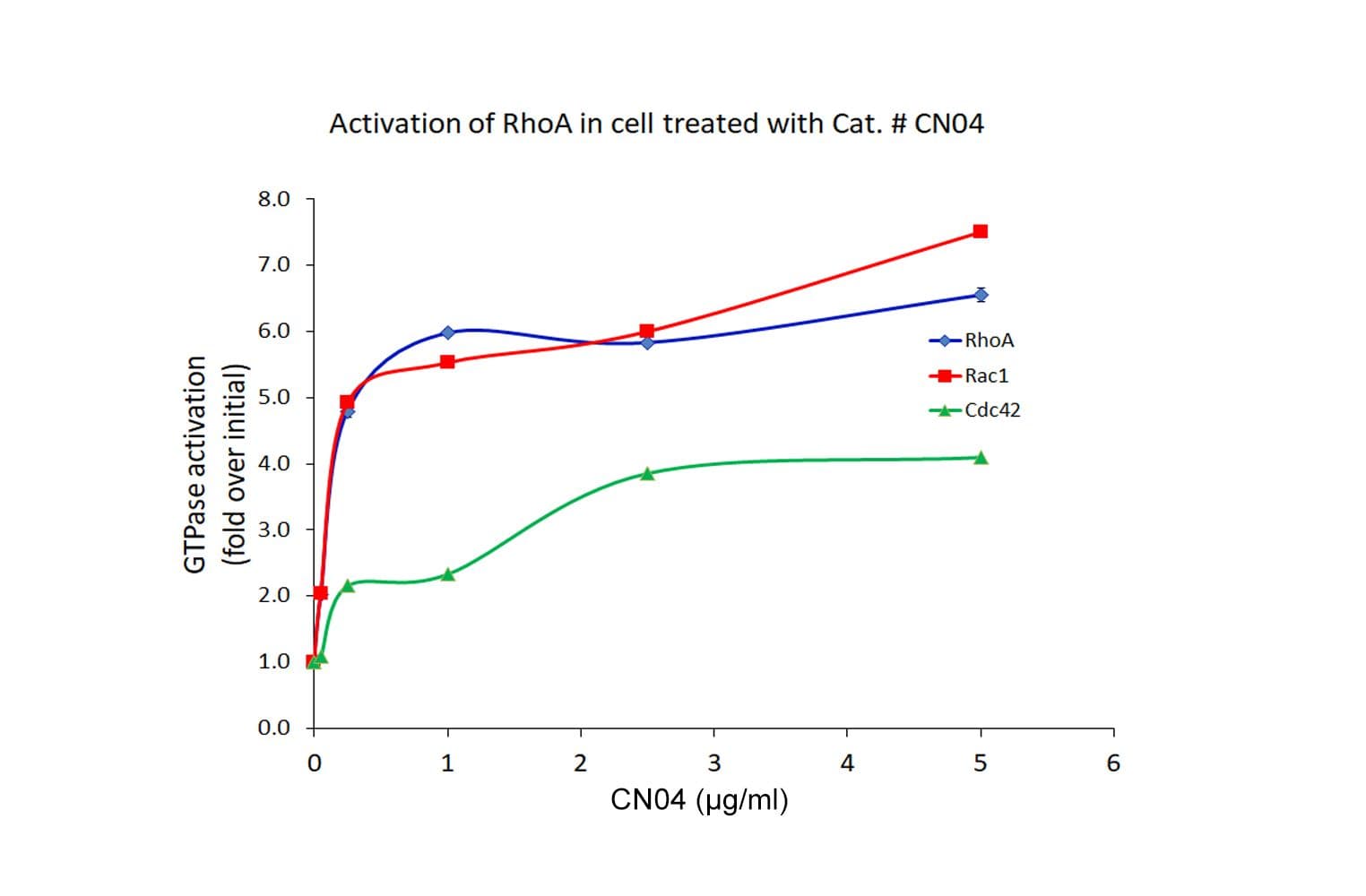
Biological activity of CN04 is demonstrated by its ability to robustly activate RhoA, Rac1 and Cdc42 in live Swiss 3T3 cells. Treatment at 1 µg/ml produces optimal activation, approximately tenfold, within 2-4 hours as measured by a RhoA BK124, Rac1 BK128 and Cdc42 BK127 G-LISA assay.
A protein containing a single point mutation that inactivates the catalytic domain of CN04 showed zero activity in a biological assay for Rho, Rac, and Cdc42 activation.
Responses to CN03 will vary depending on the cell line used.
Cat. #CN04