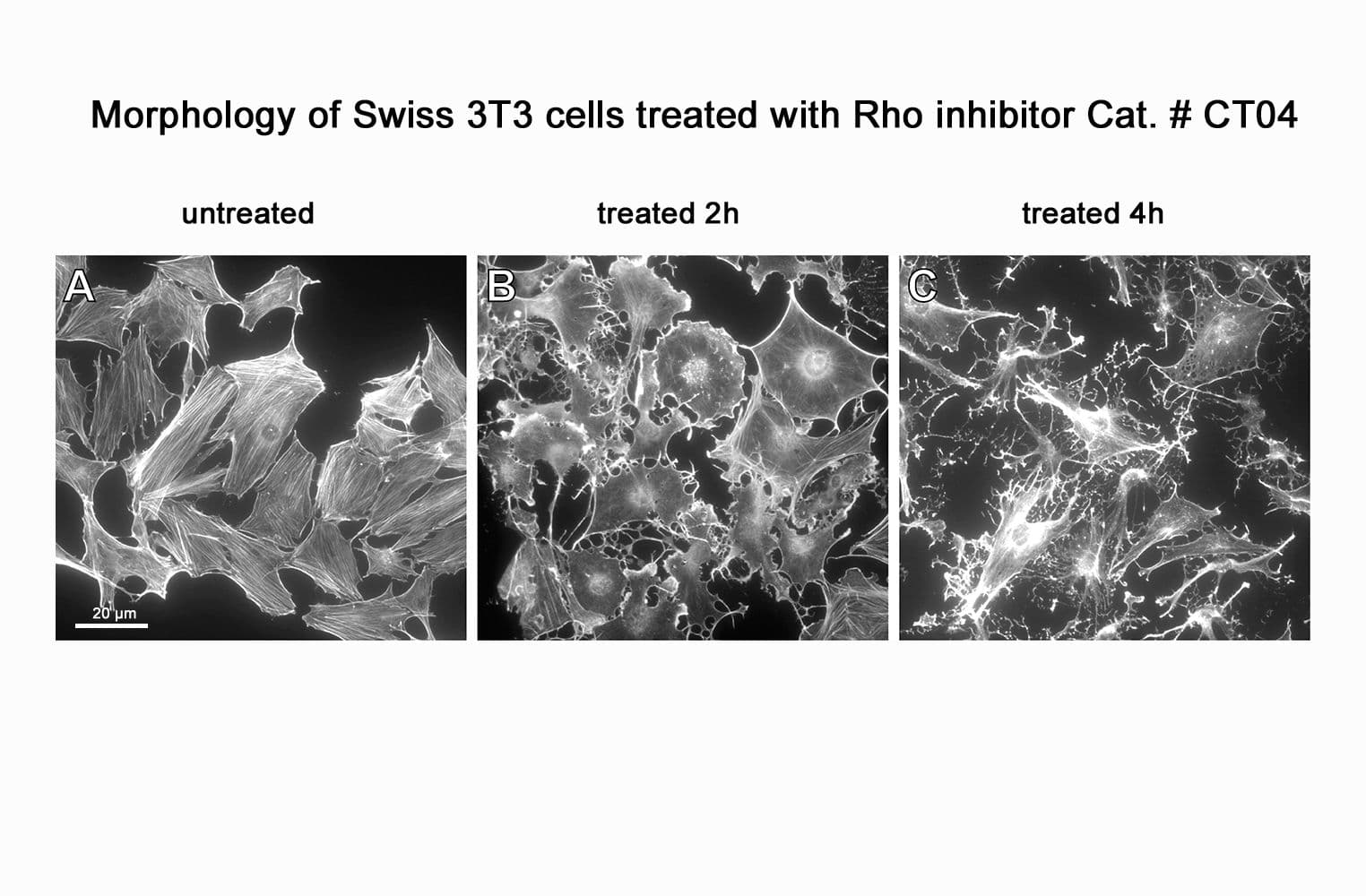
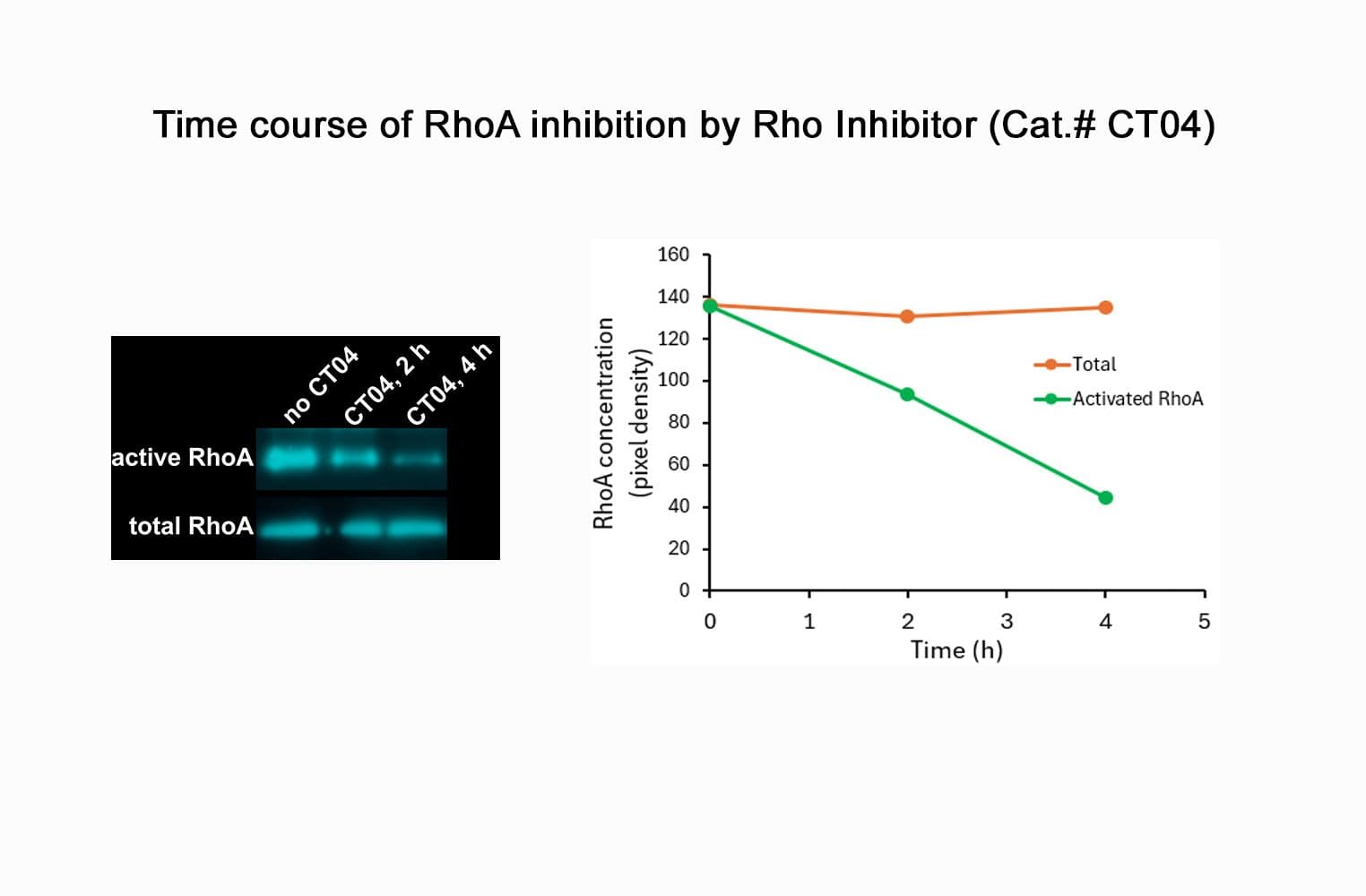
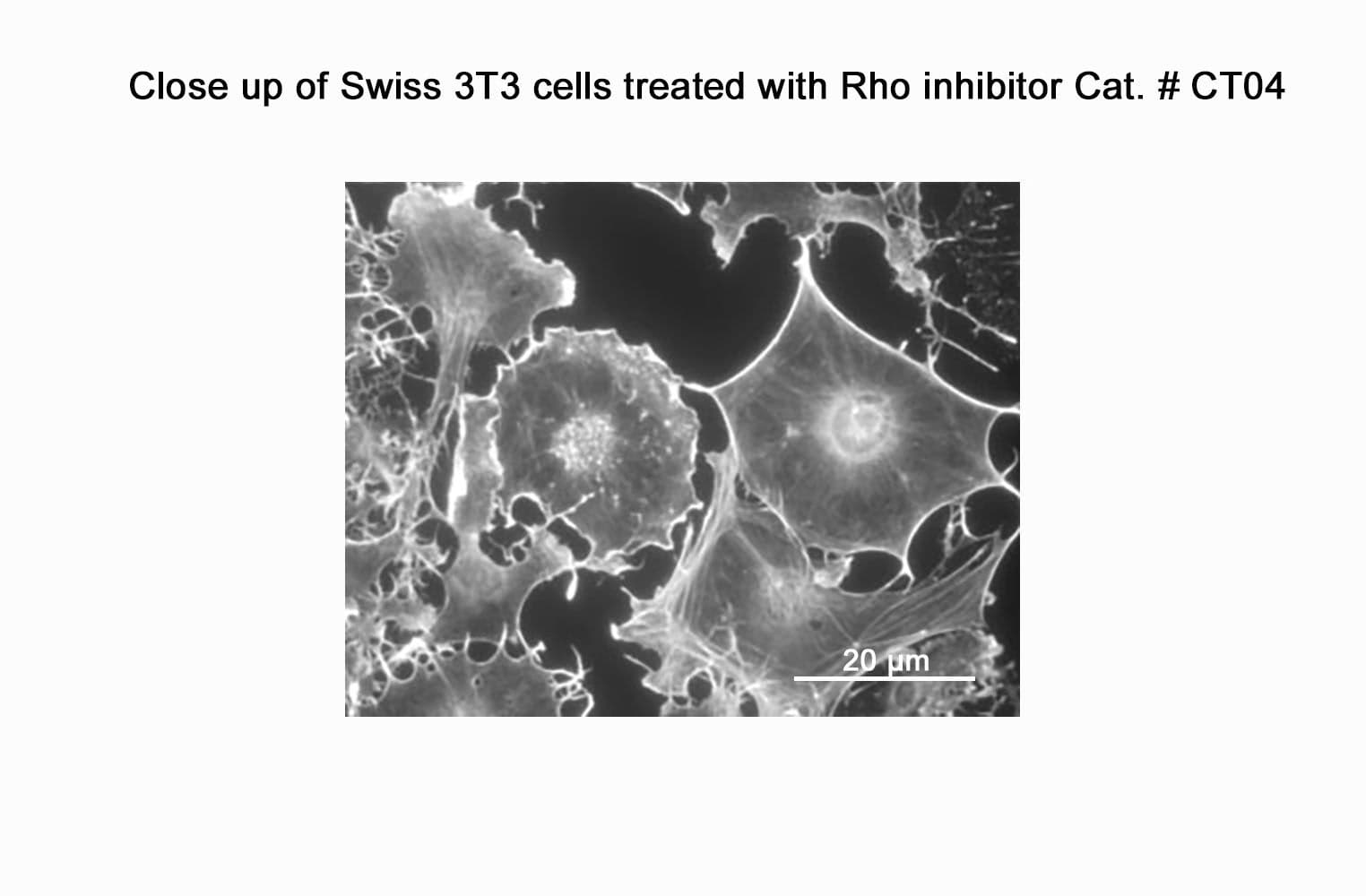
Biological activity of CT04 is demonstrated by its ability to robustly inactivate RhoA in live Swiss 3T3 cells. Treatment at 2 µg/ml for 4h produces >80% inhibition of RhoA activation by calpeptin as determined by a RhoA G-LISA assay BK124.
CT04 has been shown to inactivate Rho to an efficiency of 75-95% in fibroblasts, neurons, epithelial, endothelial, and hematopoietic cells as well as other primary and immortalized cell lines.
Responses to CT04 will vary depending on the cell line used.
- Deactivation timeline: typically 2-6 hours
- Deactivation duration: irreversible deactivation, ≥4 hours
- Typical deactivation at 4h: 55-95%