+3
Loading...
© 2026 Cytoskeleton, Inc All Rights Reserved.
Validated for:
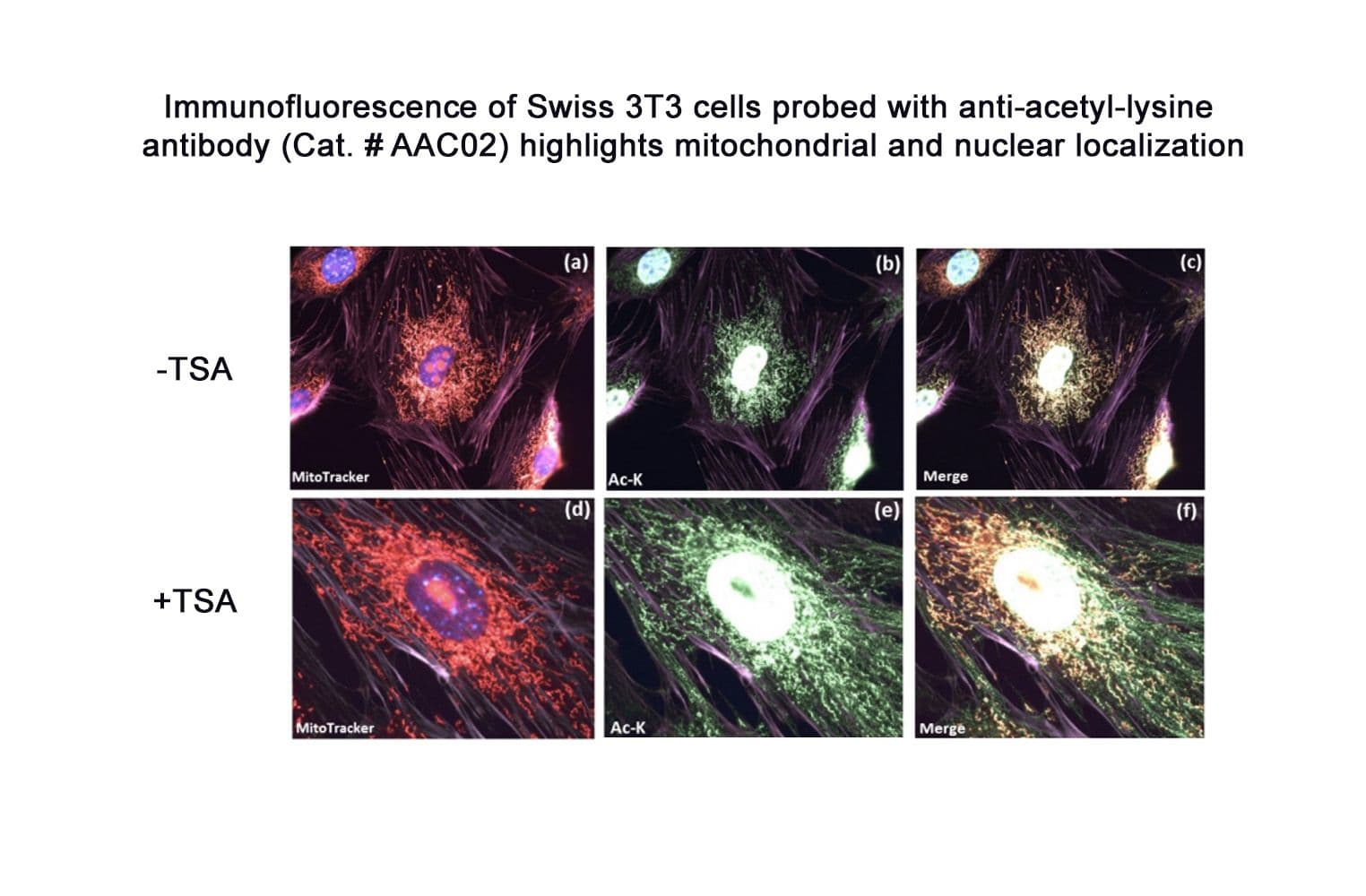
Anti-acetyl lysine antibody (AAC02) was raised against a proprietary mix of acetylated proteins to ensure broad acetyl lysine recognition. It detects a wide range of acetylated proteins, including tubulin, histones, and the mitochondrial protein pyruvate dehydrogenase (PDHE1). AAC02 shows some specificity towards acetylated histones and mitochondrial proteins in IF applications. It is purified via Protein G chromatography.
Host/isotype/clone:
Reactivity:
Amount of material:
Working concentrations:
Formulation
Sensitivity:
Specificity:
Cat. #AAC02