Cdc42 protein: His tagged: human wild type
Cat. #CD01
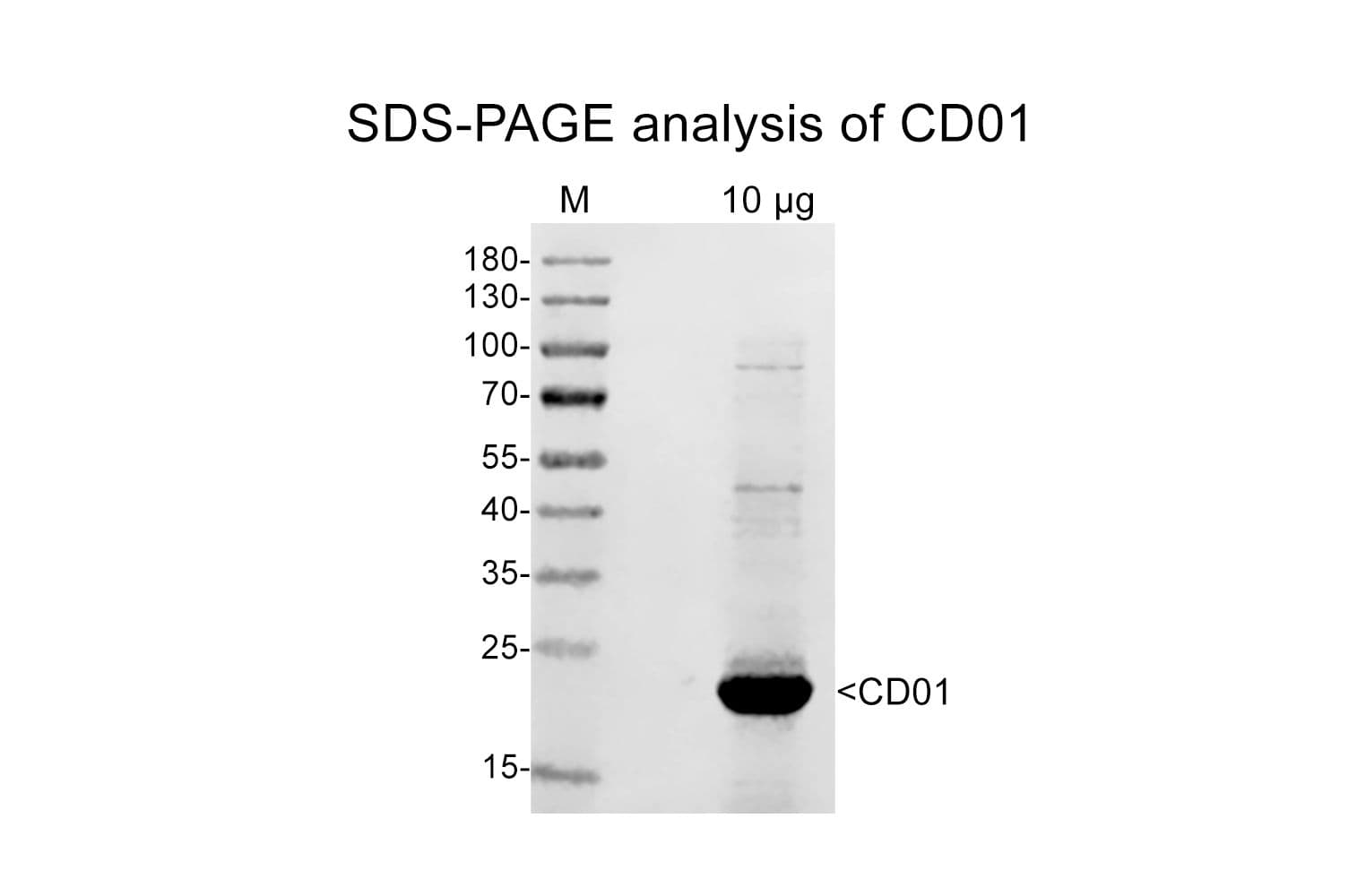
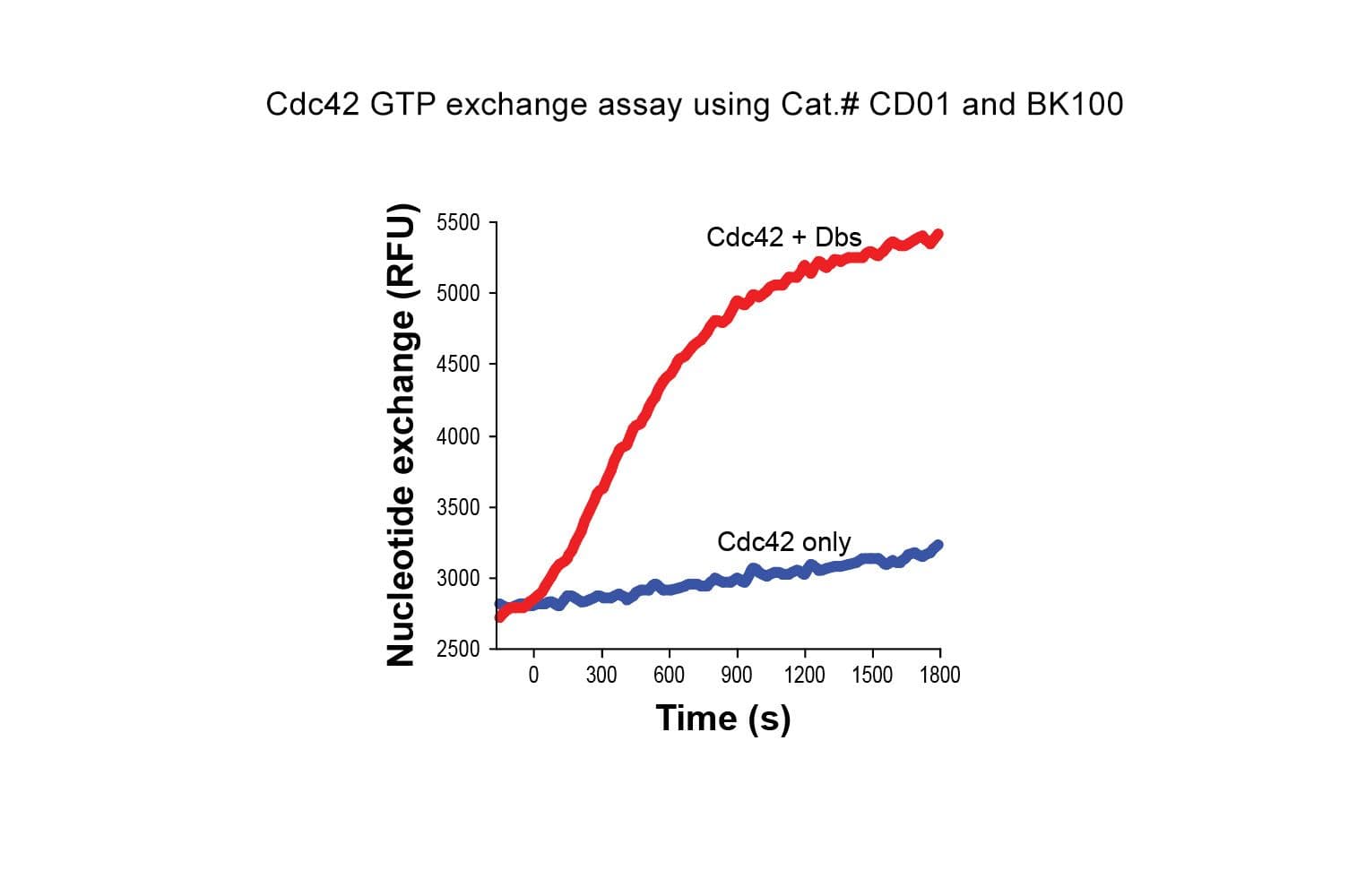

Cat. #CD01



Cdc42 is a small GTP-binding protein in the Rho family that regulates cell polarity, cytoskeletal organization, and signal transduction. It is best known for controlling the formation of filopodia—thin, actin-rich projections that help cells sense their environment and guide directional movement.
Wild-type human Cdc42 protein is produced in a bacterial expression system.
Protein purity is assessed by scanning densitometry of Coomassie Blue-stained protein on a 4-20% polyacrylamide gel. Purity was determined to be ≥90% pure.
The biological activity of CD01 is measured by its ability to exchange GTP. A standard assay uses GST-tagged PAK-1 PBD beads Cat. # PAK02 to detect GTP-bound Cdc42-His preloaded with GTPγS or GDP. The PAK CRIB domain specifically binds the active (GTP-bound) form of Cdc42. Quality control ensures ≥70% binding with GTPγS-loaded Cdc42 and ≤10% binding with GDP-loaded Cdc42.