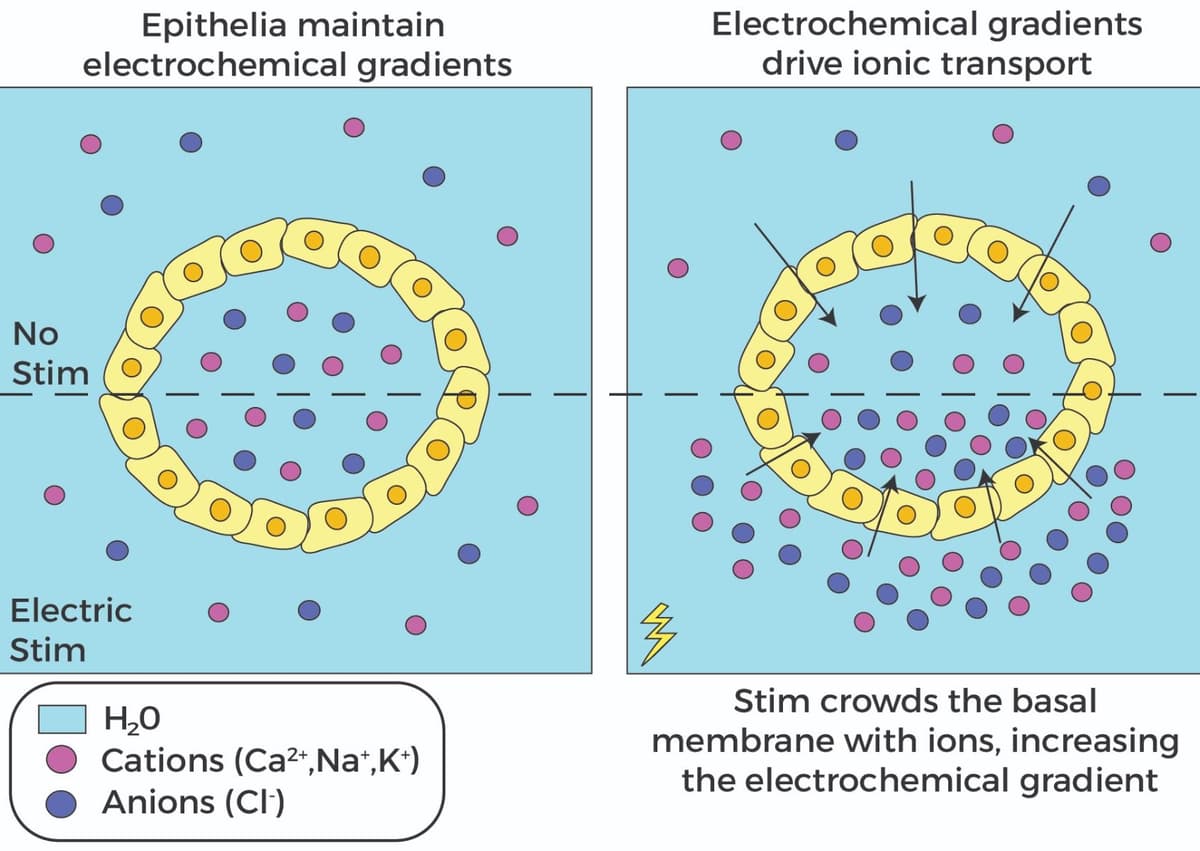
The sheet-like multicellular layer that surrounds organs and luminal structures in animals, known as the epithelia, is essential for organ development as it regulates ion and water transport, morphogenesis, cellular signaling, and cyto-mechanical events. Growing evidence shows that electrical stimulation affects epithelial function, and a recent study by Shim et al. investigated how manipulating ionic currents affect the growth and function of 3D epithelial tissue. The group used MDCK kidney cell cysts embedded in a hydrogel matrix and electrically stimulated them in their modified SCHEEPDOG electro-bioreactor, which allowed them to perform live cell imaging of tissue/organoids while controlling electrical stimulation in specified media conditions. They initially hypothesized that electrical stimulation would produce 3D electrotaxis migration, but surprisingly, they observed dramatic volumetric inflation of the MDCK cysts. This increased volume occurred in the luminal space, coincided with asymmetric deformation of the epithelium, was related to the magnitude of the electric field, and was more effective than chemical stimulants like forskolin at inducing cyst inflation. Furthermore, they determined that electrical stimulation drove ions into the lumen space of the cysts, which then recruited water to increase luminal volume, and this was confirmed with studies utilizing chemicals that inhibit ion transport. The group then assessed the role of the cytoskeleton in this phenomenon and showed that the actomyosin network worked in opposition to stabilize the cyst by establishing maximal size limits. The electro-inflation response was also studied in intestinal organoid models and further investigated using computational models to confirm these findings. Cytoskeleton Inc.’s MemGlow™ 488: Fluorogenic Membrane Probe (Cat. # MG01) was used throughout the study to effectively label the plasma membranes in MDCK cysts for effective visualization. This study demonstrated how electrical stimulation of 3D tissue promotes tissue pressurization through ion crowding mechanisms while also influencing cell migration and tissue asymmetry.
Link to Citation:
Product Used in Citation:


