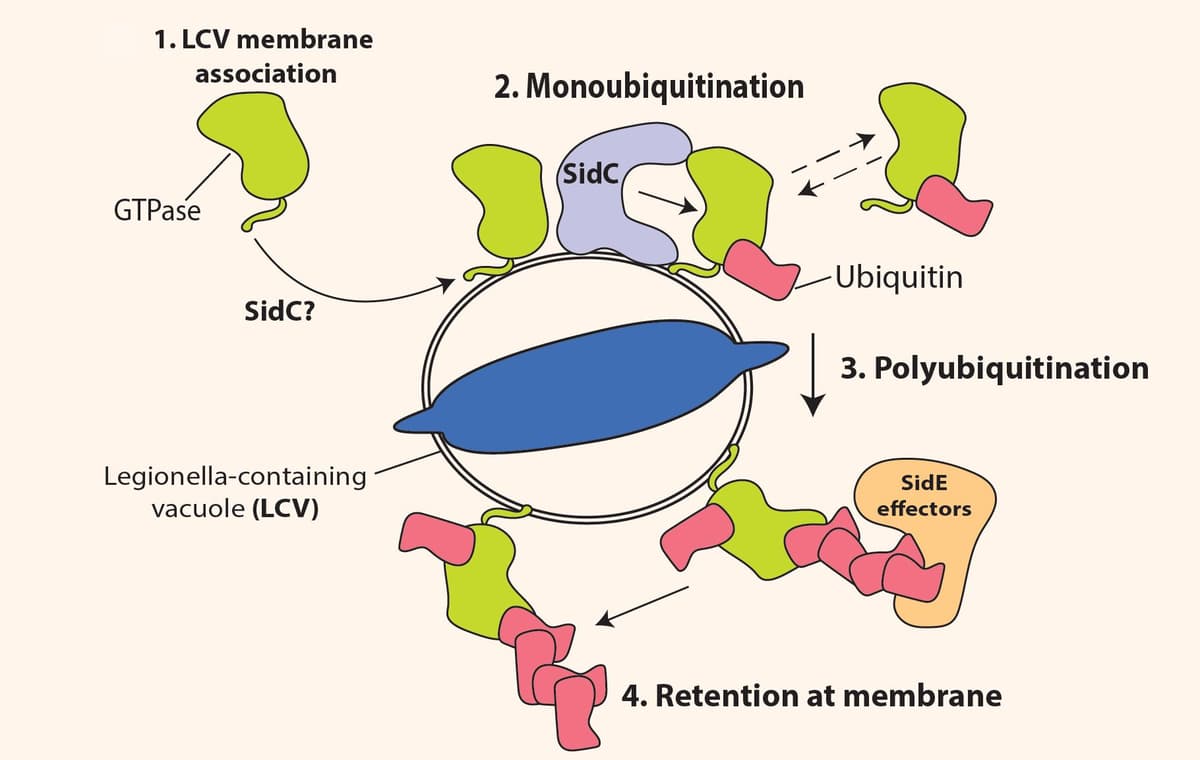
Legionella pneumophila (L.p.), the pathogen that causes Legionnaires disease, utilizes several of its bacterial proteins to hijack an array of host target processes including autophagy, translation, and membrane trafficking as a mechanism to promote pathogenesis. Several signaling pathways are known to be regulated by L.p. infection, and small GTPase signaling, which has been linked to the formation of the Legionella-containing vacuole (LCV), is of particular interest. Recent work by Steinbach et al. performed unbiased and temporal ubiquitin analysis to show that L.p. regulates an array of small GTPases through specific mechanisms. To understand L.p.’s effect on host ubiquitination, the group performed unbiased-protein-ubiquitination mass spectrometry on HEK293 FcgR cells infected with L.p. at multiple time points. Nearly 500 unique proteins showed increased ubiquitination in response to L.p. infection; remarkably, roughly 15% of identification sites belonged to small GTPases in the Ras superfamily. The group then utilized western blot approaches to validate these findings and to gain more insight into the ubiquitin chain length modifications, because that information is lost during diGly ubiquitinomic studies. They found that ubiquitination of these small GTPases were confirmed by western blot experiments, bug the results also revealed that many small GTPases were being mono- and poly-ubiquitinated. This result was very interesting, as the small GTPase protein levels were quite steady, which suggested that the ubiquitination was likely non-degradative. The group then turned their focus to specific Rab family members and found that their ubiquitination depended on recruitment to the LCV membrane, and, in particular, Rab 5 ubiquitination was regulated by the effector proteins SidC and SdcA. Several other Ras family members and Sid effector proteins were studied, and the overall findings point to a critical interaction between small GTPases and Sid proteins in the recruitment, ubiquitination, and retention of small GTPases at the LCV in response to L.p. infection. Cytoskeleton Inc.’s ubiquitination detection kit (Cat. # BK161) was used to measure the mono- and poly-ubiquitination of small GTPases in this study.
Link to Citation:
Steinbach A. et al. Cross-family small GTPase ubiquitination by the intracellular pathogen Legionella pneumophila. Mol Biol Cell. 2024 Mar 1;35(3):ar27. doi: 10.1091/mbc.E23-06-0260.
Product Used in Citation:
Signal-Seeker Ubquitination Detection Kit (30 assays) (Cat # BK161)


