Introduction of Nuclear Actin
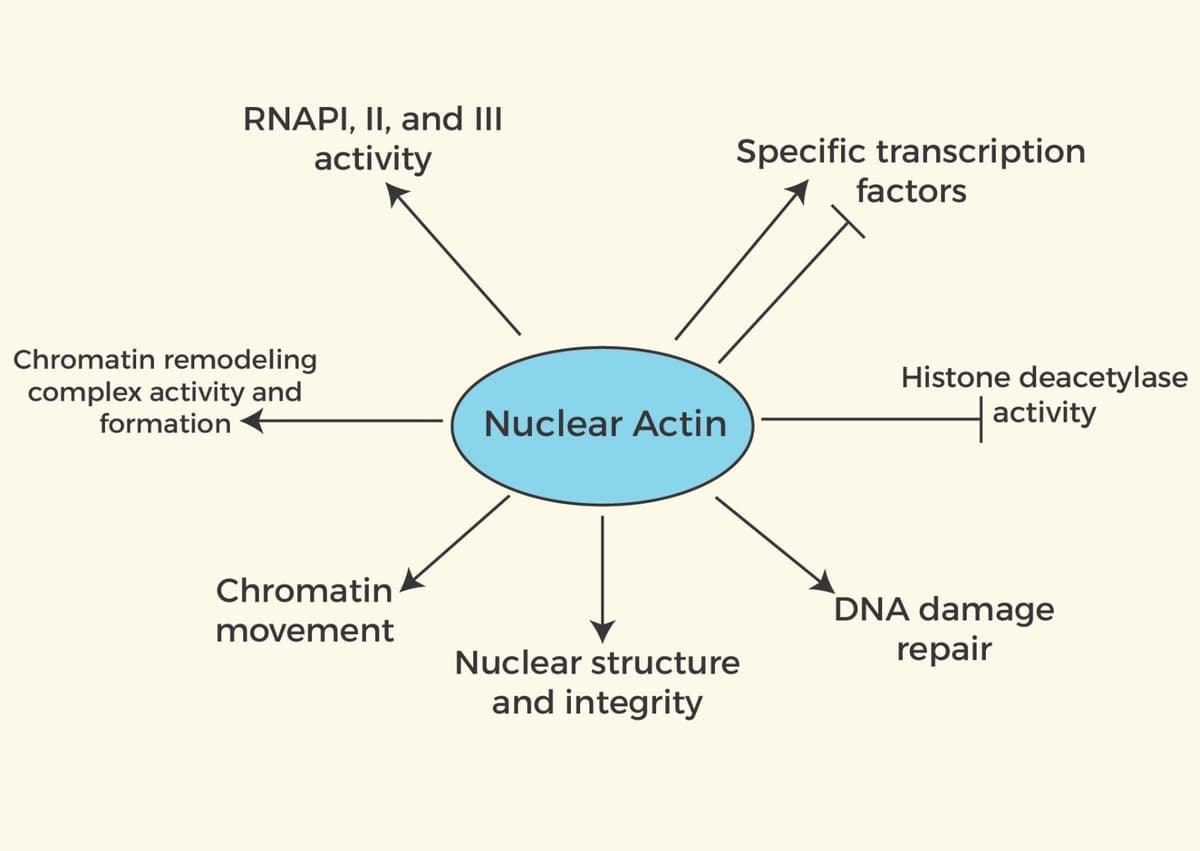
The cytoskeletal protein actin is one of the most well-studied proteins and has been shown to play an integral role in many biological processes that are essential for cell viability. While the majority of actin studies have focused on its role in the cytoplasm, several recent studies provide evidence that its function in the nucleus is also important for nuclear-specific biological processes, and disruption of nuclear actin contributes to diseases such as cancer. Early studies of nuclear actin focused on mechanisms that regulated its import and export from the nucleus, potential structures, and effects that it may have on nuclear processes (reviewed in 1). One of the earliest functions of nuclear actin was its identification as a critical component in chromatin-remodeling complexes2. Regulation of chromatin-remodeling by nuclear actin remains under intense investigation and recent work by Huang et al. showed that depletion of mitochondrial transcription factor A altered polymerization of nuclear actin, which led to a metastatic phenotype through modulated chromatin accessibility and gene expression3. Nuclear actin has also been identified as a critical player in several other nuclear processes like DNA damage repair, nuclear structure, DNA replication, cell death, and transcription (see Figure 1)(reviewed in 1, 4). This newsletter focuses on recent findings that shed light on nuclear actin's ability to regulate critical nuclear processes and its potential impact on cancer.

Click for PDF
Nuclear Actin Regulated Transcription in Cancer
Nuclear actin polymerization in response to several extracellular stimulants such as serum stimulation, hormones, and other drug stimulants was shown to have a profound effect on the regulation of transcription mechanisms(reviewed in 1). An early study by the Treisman group used serum stimulation to identify an actin treadmilling effect, whereby polymerization of cytoplasmic actin led to the release of the transcription factor Mal in the cytoplasm and its localization to the nucleus5. Once in the nucleus, it interacts with SRF (serum response factor) and promotes transcription. Interestingly, nuclear actin also inhibits serum-mediated Mal/SRF transcription, and the release of Mal by nuclear actin was also required for proper transcription6. Work by the Grosse lab showed, using an optogenetically-activated mDia formin protein, that nuclear actin polymerization was sufficient for Mal release and subsequent SRF/Mal transcription7. Similarly, several studies have reported that nuclear actin can affect general transcription through its regulation of RNA polymerase II (Pol II)(reviewed in 4), and in particular, it was shown that persistent nuclear actin filaments can have deleterious effects on Pol II transcription8. Conversely, it was shown that in response to serum stimulation, N-WASP-dependent nuclear actin polymerization led to a clustering of Pol II in a phase-separated (droplet-like) state to promote transcription9. The Horning group's recent study combined these two mechanisms and showed how the formin and actin nucleator DAAM2 associates with androgen receptors in response to dihydrotestosterone and led to the formation of transcriptional droplets that included the androgen receptors, DAAM2, and polymerized nuclear actin which interacted with active Pol II to promote hormone signaling10. Importantly, this study also determined that this nuclear actin-dependent mechanism could be important for prostate-specific antigen expression in prostate cancer. Growing evidence suggests that formin-dependent nuclear actin-driven transcription is critical from proper biological function and may be a mechanism that is disrupted in the setting of cancer.
Nuclear Actin Controls DNA Damage Response Mechanisms
There is a strong body of evidence that actin-related proteins interact with DNA damage proteins and can regulate the DNA damage processes(reviewed in 11). An early study showed that polymeric actin played a key role in DNA double-strand repair, and was important for recruiting Ku80 to damaged sites12. Another study showed that DNA damage directly generated nuclear actin filaments with assistance from key formin proteins13, and was one of the first reports to utilize advanced microscopy tools to label and track nuclear F-actin. In 2018, two groups identified a critical role for dynamic nuclear actin polymerization in the setting of homologous recombination (HR)14,15. In combination with actin-binding proteins (e.g. myosin and Arp 2/3), polymerized nuclear actin directed the movement of double-stranded break DNA to discrete sub-nuclear clusters to regulate HR repair. An interesting study found that polymerized nuclear actin contributes to DNA repair by functioning as a scaffold that promotes the localization of PML nuclear bodies to DNA lesions and that the toxic prelamin A protein can disrupt this function by localizing actin to the nuclear envelope16. Building on nuclear actin’s role in DNA damage, a recent study showed that the tumor suppressor p53, an essential protein in DNA damage response, can regulate nuclear actin filament in response to DNA damage and this crosstalk may be important in cancer cell survival17. Resistance to chemotherapy by epithelial-mesenchymal-transition (EMT) has been observed, but the mechanisms for the resistance were not well defined. Work by Debaugnies et al. showed that the small GTPase RhoJ is specifically upregulated in cancer cells that have undergone EMT which allowed them to activate DNA damage response mechanisms in response to chemotherapy18. Interestingly, RhoJ did not affect traditional DNA damage checkpoint proteins, but was found to regulate nuclear actin by specifically interacting with and modulating actin-interacting proteins18. RhoJ’s impact on nuclear actin led to enhanced DNA damage repair in response to chemotherapy, which could be reversed in RhoJ cells with the F-actin inhibitor latrunculin B18. Nuclear actin plays a fundamental role in DNA damage responses and appears to be an important target modulated by cancer cells for their survival.
Replication Stress Repair Involves Nuclear Actin
In its simplest terms, replication stress (RS) is when the DNA replication process is disrupted due to exogenous insults or endogenous mechanisms. In the setting of cancer, RS is of great interest because it promotes genomic instability, is highly prevalent in tumor cells, and may be a mechanism to evade therapeutic response (reviewed in 19). Initial studies with nuclear actin showed that its dynamicity and regulation by formins were important for DNA replication20. In 2020, the Cesare group directly linked nuclear actin to RS; specifically, they found that polymerized nuclear F-Actin suppressed nuclear deformation via increased nuclear volume, mobilized stressed-replication foci to the nuclear periphery, and directed movements along nuclear filaments with the assistance of myosin II21. These mechanisms collectively resulted in RS repair and were shown to be active in xenograft tumor models after treatment with RS-inducing chemotherapeutics21. Recently, it was shown that nuclear actin-dependent RS remodeling functioned by interacting with replication factories and limited PrimPol activity in response to genotoxic treatments22. These results were interesting as PrimPol, a DNA primase and DNA polymerase, was shown to rescue fork degradation and promote DNA synthesis in response to repeated doses of chemotherapeutics as a mechanism for cancer cell survival23. Collectively, these studies suggest that nuclear actin may be a critical mechanism to regulate RS and may be an important target to control RS in cancer.
Future Insights
Similarly to actin’s role in the cytoplasm, it appears that nuclear actin's dynamic ability to form filamentous structures is essential for an array of processes in the nucleus. Its ability to organize critical complexes spatially and temporally is critical to control nuclear processes like those discussed above, and disruption or augmentation of nuclear actin polymerization appears to be a mechanism for cancer progression. The evidence above suggests that many of these mechanisms are regulated through actin-binding proteins; however, it will be interesting to determine if other mechanisms such as post-translational modifications also play a role in defining unique populations of actin that perform discrete functions. Identifying these unique features is important if the goal is to target specific actin-dependent nuclear mechanisms for therapeutic intervention. The novel functions of nuclear actin continue to grow, and Cytoskeleton Inc. is proud to provide critical tools to support this growing area of research.
References
- Kelpsch, D.J. and T.L. Tootle, Nuclear Actin: From Discovery to Function. Anat Rec (Hoboken), 2018. 301(12): p. 1999-2013.
- Zhao, K., et al., Rapid and phosphoinositol-dependent binding of the SWI/SNF-like BAF complex to chromatin after T lymphocyte receptor signaling. Cell, 1998. 95(5): p. 625-36.
- Huang, Q., et al., TFAM loss induces nuclear actin assembly upon mDia2 malonylation to promote liver cancer metastasis. EMBO J, 2022. 41(11): p. e110324.
- Ulferts, S., et al., Nuclear actin dynamics and functions at a glance. J Cell Sci, 2024. 137(6).
- Miralles, F., et al., Actin dynamics control SRF activity by regulation of its coactivator MAL. Cell, 2003. 113(3): p. 329-42.
- Vartiainen, M.K., et al., Nuclear actin regulates dynamic subcellular localization and activity of the SRF cofactor MAL. Science, 2007. 316(5832): p. 1749-52.
- Baarlink, C., H. Wang, and R. Grosse, Nuclear actin network assembly by formins regulates the SRF coactivator MAL. Science, 2013. 340(6134): p. 864-7.
- Serebryannyy, L.A., et al., Persistent nuclear actin filaments inhibit transcription by RNA polymerase II. J Cell Sci, 2016. 129(18): p. 3412-25.
- Wei, M., et al., Nuclear actin regulates inducible transcription by enhancing RNA polymerase II clustering. Sci Adv, 2020. 6(16): p. eaay6515.
- Knerr, J., et al., Formin-mediated nuclear actin at androgen receptors promotes transcription. Nature, 2023. 617(7961): p. 616-622.
- Caridi, C.P., et al., Nuclear actin filaments in DNA repair dynamics. Nat Cell Biol, 2019. 21(9): p. 1068-1077.
- Andrin, C., et al., A requirement for polymerized actin in DNA double-strand break repair. Nucleus, 2012. 3(4): p. 384-95.
- Belin, B.J., T. Lee, and R.D. Mullins, DNA damage induces nuclear actin filament assembly by Formin -2 and Spire-(1/2) that promotes efficient DNA repair. [corrected]. Elife, 2015. 4: p. e07735.
- Schrank, B.R., et al., Nuclear ARP2/3 drives DNA break clustering for homology-directed repair. Nature, 2018. 559(7712): p. 61-66.
- Caridi, C.P., et al., Nuclear F-actin and myosins drive relocalization of heterochromatic breaks. Nature, 2018. 559(7712): p. 54-60.
- Cobb, A.M., et al., Filamentous nuclear actin regulation of PML NBs during the DNA damage response is deregulated by prelamin A. Cell Death Dis, 2022. 13(12): p. 1042.
- Torii, T., et al., Loss of p53 function promotes DNA damage-induced formation of nuclear actin filaments. Cell Death Dis, 2023. 14(11): p. 766.
- Debaugnies, M., et al., RHOJ controls EMT-associated resistance to chemotherapy. Nature, 2023. 616(7955): p. 168-175.
- Macheret, M. and T.D. Halazonetis, DNA replication stress as a hallmark of cancer. Annu Rev Pathol, 2015. 10: p. 425-48.
- Parisis, N., et al., Initiation of DNA replication requires actin dynamics and formin activity. EMBO J, 2017. 36(21): p. 3212-3231.
- Lamm, N., et al., Nuclear F-actin counteracts nuclear deformation and promotes fork repair during replication stress. Nat Cell Biol, 2020. 22(12): p. 1460-1470.
- Palumbieri, M.D., et al., Nuclear actin polymerization rapidly mediates replication fork remodeling upon stress by limiting PrimPol activity. Nat Commun, 2023. 14(1): p. 7819.
- Quinet, A., et al., PRIMPOL-Mediated Adaptive Response Suppresses Replication Fork Reversal in BRCA-Deficient Cells. Mol Cell, 2020. 77(3): p. 461-474 e9.


