Introduction
The Rac small GTPase subfamily consists of three family members (Rac1, Rac2, Rac3) which are roughly 90% homologous to each other. These small GTPases function as molecular switches that toggle between its GDP-bound inactive and GTP-bound active forms. Rac1 is the best-studied isoform likely because it is ubiquitously expressed vis-à-vis Rac2, which is primarily expressed in hematopoietic cells1. Rac1 is vital for normal cell function, which is supported by the finding that knockout of Rac1 in mice leads to embryonic lethality2. Furthermore, dysfunction of Rac1 contributes to an array of human diseases including cancer, neurodegeneration, immunological diseases, and cardiovascular diseases3. These mutations can affect several Rac1 functions including the regulation of the actin cytoskeleton4, cell adhesion, motility, and proliferation. New regulatory functions for Rac1 continue to be discovered, and several recent studies have provided key insight into Rac1’s ability to regulate macrophages. In this newsletter, we summarize these recent findings that place Rac1 as a key regulator of macrophage function in health and disease.

Rac1 regulates multiple morphologic and functional characteristics of macrophages
Macrophages are specialized immune cells that perform an array of functions in the body; for example, they perform immune surveillance, participate in wound healing, and contribute both positively and negatively to disease progression5. The Ridley group first described the role of Rac1 in bone marrow-derived macrophages using Rac1 conditional knockout macrophages to define its role6. Surprisingly, Rac1 knockout did not appear to affect macrophage migration but did affect its morphology. A recent publication studying macrophages in vivo in a zebrafish model found opposing results to this early study, whereby, chemical Rac1 inhibition significantly affected macrophage migration7. These models are quite different so additional studies may be needed to fully understand Rac1’s effect on macrophage migration. The study using zebrafish also showed that loss of Rac1 altered the macrophage’s ability to break down extracellular matrix7, and supports an earlier finding that Rac1 may be important for the macrophage's invasive properties8.
Rac1 was also shown to play a critical role in macrophage engulfment of apoptotic cells (now termed efferocytosis), as Rac1 chemical inhibition suppressed phagocytosis9. In support of this previous finding, a recent study showed that TGF-ß-signaling affected macrophage efferocytosis in a Rac1-dependent fashion10. Further links between macrophage efferocytosis and Rac1 were uncovered when a study identified that maresin-conjugates-in-tissue-regeneration (MCTRs) prime macrophages for continual efferocytosis in a Rac1-dependent fashion11. Finally, Fu et al. showed that altering Rac1 levels in a neonate macrophage model has a profound effect on the inflammatory phenotype of M1 macrophages12. Collectively, Rac1 appears to be a critical regulator of macrophage morphology and function and may be important in disease progression.
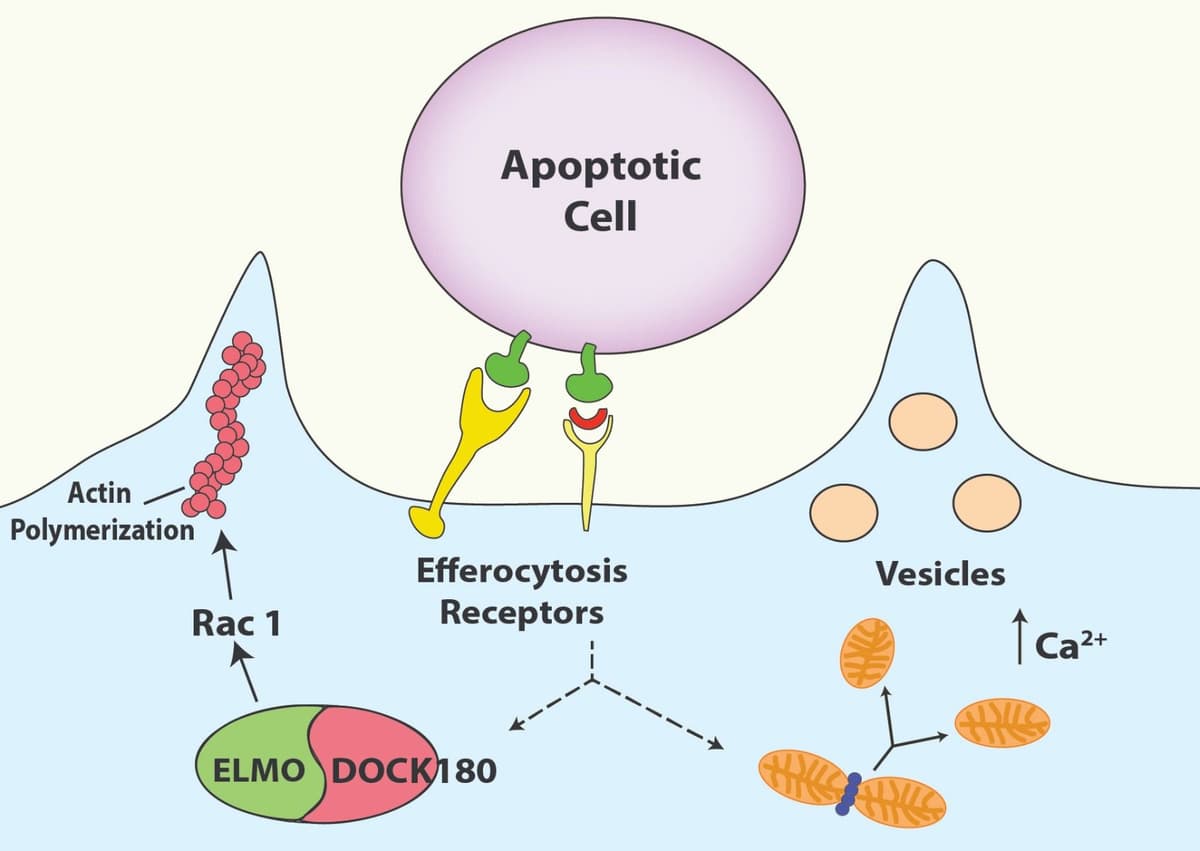
Altering Rac1 in macrophages affects cancer progression
Rac1’s effect on cancer cells has been intensely investigated; for example, its been shown to play a critical role in metastasis (see previous newsletter Rac1 and Cdc42 pathways as pivotal axis in future metastatic cancer). A recent study by the Hirsch Lab showed that Rac1-NFAT signaling promotes the shift of macrophages to osteoclast differentiation and promotes bone metastasis in a lung adenocarcinoma model13. The group also showed that Inhibition of Rac1 was sufficient to alleviate bone marrow metastasis in this model. Similarly, in a colorectal cancer model, the engulfment and cell motility protein 1 (ELMO1) was shown to reprogram macrophages to tumor-associated macrophages in a Rac1-dependent fashion, which facilitates malignant cancer phenotypes14. These studies suggest that Rac1-driven cancer progression and metastasis may also depend on Rac1 effects in stromal macrophages as well as with its direct effect in the cancer cells. A peripherally related, but interesting study Rac2 was performed and the group found that hyperactive Rac2E62K mutation in human HL60-derived macrophage-like cells, or bone marrow-derived macrophages resulted in significant engulfment of T cells15. When these hyperactive-Rac2 macrophages were used in a chimeric antigen receptor-expressing macrophage setting, a significant engulfment of target cancer cells was observed. Thus, artificial regulation of the Rac family may lead to viable therapeutic strategies to treat cancer15.
Rac1 Regulates Macrophage activation in kidney disease
Macrophage accumulation is a hallmark of kidney disease, and these macrophages create an inflammatory environment that promotes fibrosis and irreparable damage to the kidney. One of the earliest studies linking Rac1 to macrophages in renal disease looked at the mechanism by which tissue-type plasminogen activator (tPA) recruited macrophages during chronic kidney disease. The group found that inhibiting NF-kß, FAK, and Rac1 all contributed to tPA-induced macrophage recruitment16. Nagase et al. further defined the importance of Rac1 in macrophages in the setting of kidney injury. They found that deletion of Rac1 in myeloid lineages protected against LPS-induced renal inflammation and injury17. Recently, a group utilized single-cell RNA sequencing to determine that macrophage efferocytosis is dysfunctional in diabetic kidney disease, and overexpression of Rac1 could enhance efferocytosis and reduce overall inflammation in this setting18. Inhibiting Rac1 to suppress macrophage migration in kidney disease also protected against kidney injury in an ischemia/reperfusion model19. Importantly, while outside the scope of this newsletter, it is important to note that Rac1 can also act on the renal cells directly in the setting of kidney disease20; nevertheless, the mounting evidence suggests that Rac1 signaling in macrophages profoundly affects kidney disease progression.
Future perspectives
The studies above support an essential role for Rac1 in macrophage morphology and function both in biology and disease. It will be interesting to determine whether Rac1’s effect on macrophages is actin-dependent or independent, what upstream signaling mechanisms activate Rac1, and whether Rac1 performs GTPase-independent functions in the macrophage. For example, a previous study showed that in macrophages, Rac1 is part of the NADPH oxidase complex that is important for defending against pathogens21. Recently the NADPH oxidase complex was solved and Rac1 in combination with p67 clamps onto Nox2 to promote its contraction and stabilization with NADPH22. Rac1 small GTPases are critical regulatory proteins with numerous, diverse functions, and having a precise understanding of its role in health and disease will provide a better rationale for Rac1 targeted therapies.
References
1. Didsbury, J., et al., rac, a novel ras-related family of proteins that are botulinum toxin substrates. J Biol Chem, 1989. 264(28): p. 16378-82.
2. Sugihara, K., et al., Rac1 is required for the formation of three germ layers during gastrulation. Oncogene, 1998. 17(26): p. 3427-33.
3. Marei, H. and A. Malliri, Rac1 in human diseases: The therapeutic potential of targeting Rac1 signaling regulatory mechanisms. Small GTPases, 2017. 8(3): p. 139-163.
4. Ridley, A.J., et al., The small GTP-binding protein rac regulates growth factor-induced membrane ruffling. Cell, 1992. 70(3): p. 401-10.
5. Austermann, J., J. Roth, and K. Barczyk-Kahlert, The Good and the Bad: Monocytes' and Macrophages' Diverse Functions in Inflammation. Cells, 2022. 11(12).
6. Wells, C.M., et al., Rac1-deficient macrophages exhibit defects in cell spreading and membrane ruffling but not migration. J Cell Sci, 2004. 117(Pt 7): p. 1259-68.
7. Travnickova, J., et al., Macrophage morphological plasticity and migration is Rac signalling and MMP9 dependant. Sci Rep, 2021. 11(1): p. 10123.
8. Wheeler, A.P., et al., Rac1 and Rac2 regulate macrophage morphology but are not essential for migration. J Cell Sci, 2006. 119(Pt 13): p. 2749-57.
9. Nakaya, M., et al., Spatiotemporal activation of Rac1 for engulfment of apoptotic cells. Proc Natl Acad Sci U S A, 2008. 105(27): p. 9198-203.
10. Liao, Z., et al., Myofiber directs macrophages IL-10-Vav1-Rac1 efferocytosis pathway in inflamed muscle following CTX myoinjury by activating the intrinsic TGF-beta signaling. Cell Commun Signal, 2023. 21(1): p. 168.
11. Koenis, D.S., et al., Efferocyte-Derived MCTRs Metabolically Prime Macrophages for Continual Efferocytosis via Rac1-Mediated Activation of Glycolysis. Adv Sci (Weinh), 2024. 11(7): p. e2304690.
12. Fu, H., et al., Interfering with Rac1-activation during neonatal monocyte-macrophage differentiation influences the inflammatory responses of M1 macrophages. Cell Death Dis, 2023. 14(9): p. 619.
13. Zhang, X., et al., RAC1 inhibition ameliorates IBSP-induced bone metastasis in lung adenocarcinoma. Cell Rep, 2024. 43(8): p. 114528.
14. Wen, B., et al., Engulfment and cell motility protein 1 fosters reprogramming of tumor-associated macrophages in colorectal cancer. Cancer Sci, 2023. 114(2): p. 410-422.
15. Mishra, A.K., et al., Hyperactive Rac stimulates cannibalism of living target cells and enhances CAR-M-mediated cancer cell killing. Proc Natl Acad Sci U S A, 2023. 120(52): p. e2310221120.
16. Lin, L., et al., Myeloid-derived tissue-type plasminogen activator promotes macrophage motility through FAK, Rac1, and NF-kappaB pathways. Am J Pathol, 2014. 184(10): p. 2757-67.
17. Nagase, M., et al., Deletion of Rac1GTPase in the Myeloid Lineage Protects against Inflammation-Mediated Kidney Injury in Mice. PLoS One, 2016. 11(3): p. e0150886.
18. Song, Y., et al., Single-Cell RNA Sequencing Reveals RAC1 Involvement in Macrophages Efferocytosis in Diabetic Kidney Disease. Inflammation, 2024. 47(2): p. 753-770.
19. Park, Y.R., et al., Rac1 inhibition protects the kidney against kidney ischemia/reperfusion through the inhibition of macrophage migration. Korean J Physiol Pharmacol, 2023. 27(3): p. 257-265.
20. Shibata, S., et al., Modification of mineralocorticoid receptor function by Rac1 GTPase: implication in proteinuric kidney disease. Nat Med, 2008. 14(12): p. 1370-6.
21. Zhao, X., K.A. Carnevale, and M.K. Cathcart, Human monocytes use Rac1, not Rac2, in the NADPH oxidase complex. J Biol Chem, 2003. 278(42): p. 40788-92.
22. Liu, X., et al., Structure of human phagocyte NADPH oxidase in the activated state. Nature, 2024. 627(8002): p. 189-195.


