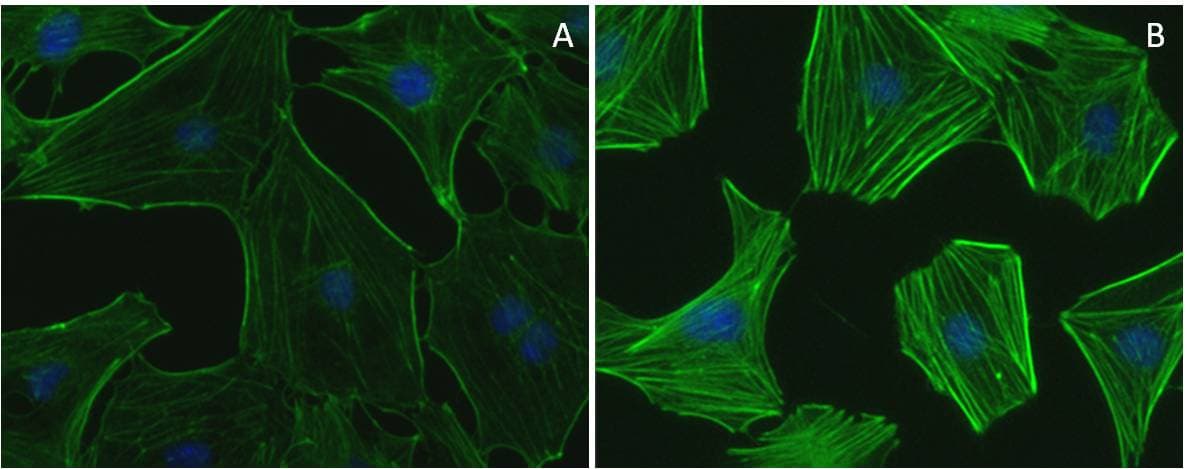
Following rotavirus infection, the expression of viral enterotoxin non-structural protein 4 (NSP4) results in mobilization of intracellular Ca2+ levels in a cell-type specific manner and cytoskeletal disorganization. To further elucidate this relationship, investigators infected MA104 cells with the DxRRV strain of rotavirus and observed disassembly of cortical actin, formation of stress fibers, and tubulin disorganization. Investigators used Cytoskeleton’s RhoA Pull-down Activation Assay (Cat.# BK036) to identify and link early activation of RhoA with a corresponding formation of stress fibers. RhoA-driven stress fiber formation was reversible by treatment of anti-rotavirus spike protein VP5* antibody, suggesting virion interaction with cell surface integrins α2β1 and α4β1 leads to RhoA activation. Importantly, treatment of infected cells with Ca2+ chelator BAPTA/AM or SiRNA-NSP4 failed to prevent stress fiber formation but did partially reverse reticulum-like reorganization of actin which suggests that RhoA activation and subsequent formation of stress-fibers occurs independently from NSP4-induced Ca2+ mobilization. Reorganization of microtubules to the cytoplasmic periphery could be partially reversed by Ca2+ chelation, with a greater reversal observed when NSP4 was silenced via SiRNA. The authors propose that cytoskeletal changes induced by rotavirus infection of intestinal absorptive cells negatively impacts the epithelial barrier function of the intestines and contributes to the development of life-threatening dehydration through diarrhea.

Products Used:
RhoA Pull-down Activation Assay Biochem Kit (bead pull-down format) - 80 Assays
Link to Citation:


