BLOG
+3
Loading...
Cat. #MG03
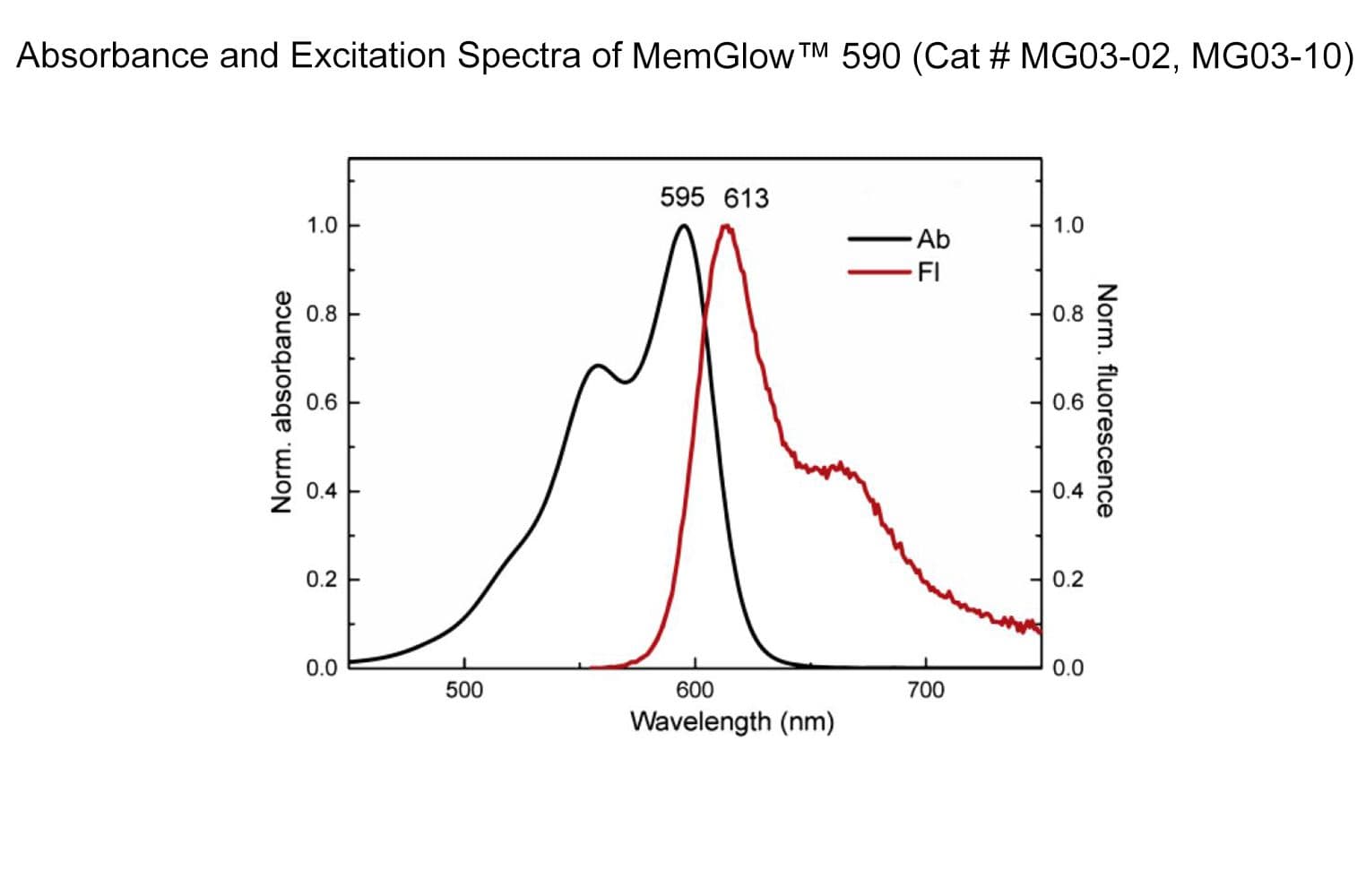
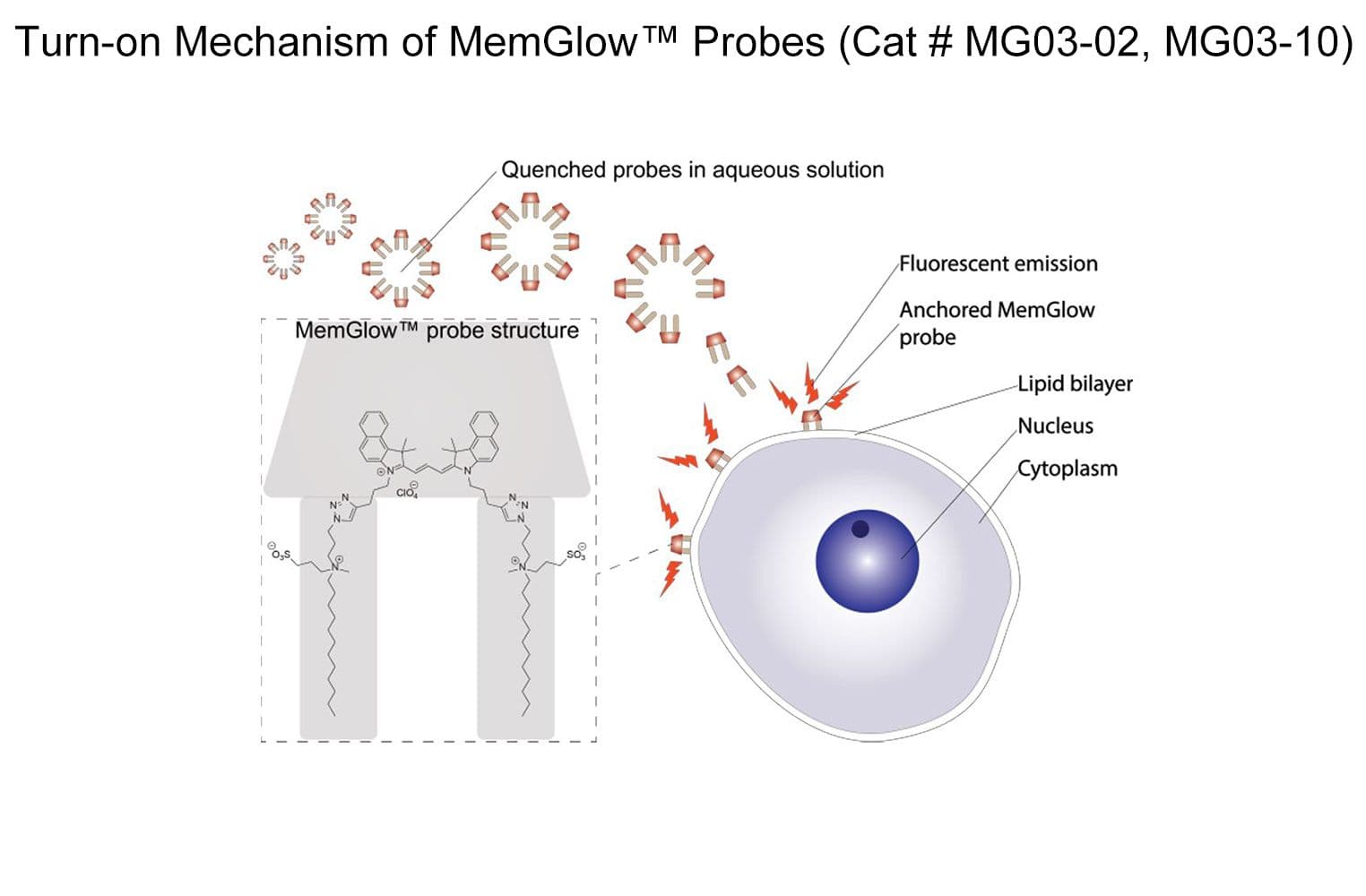
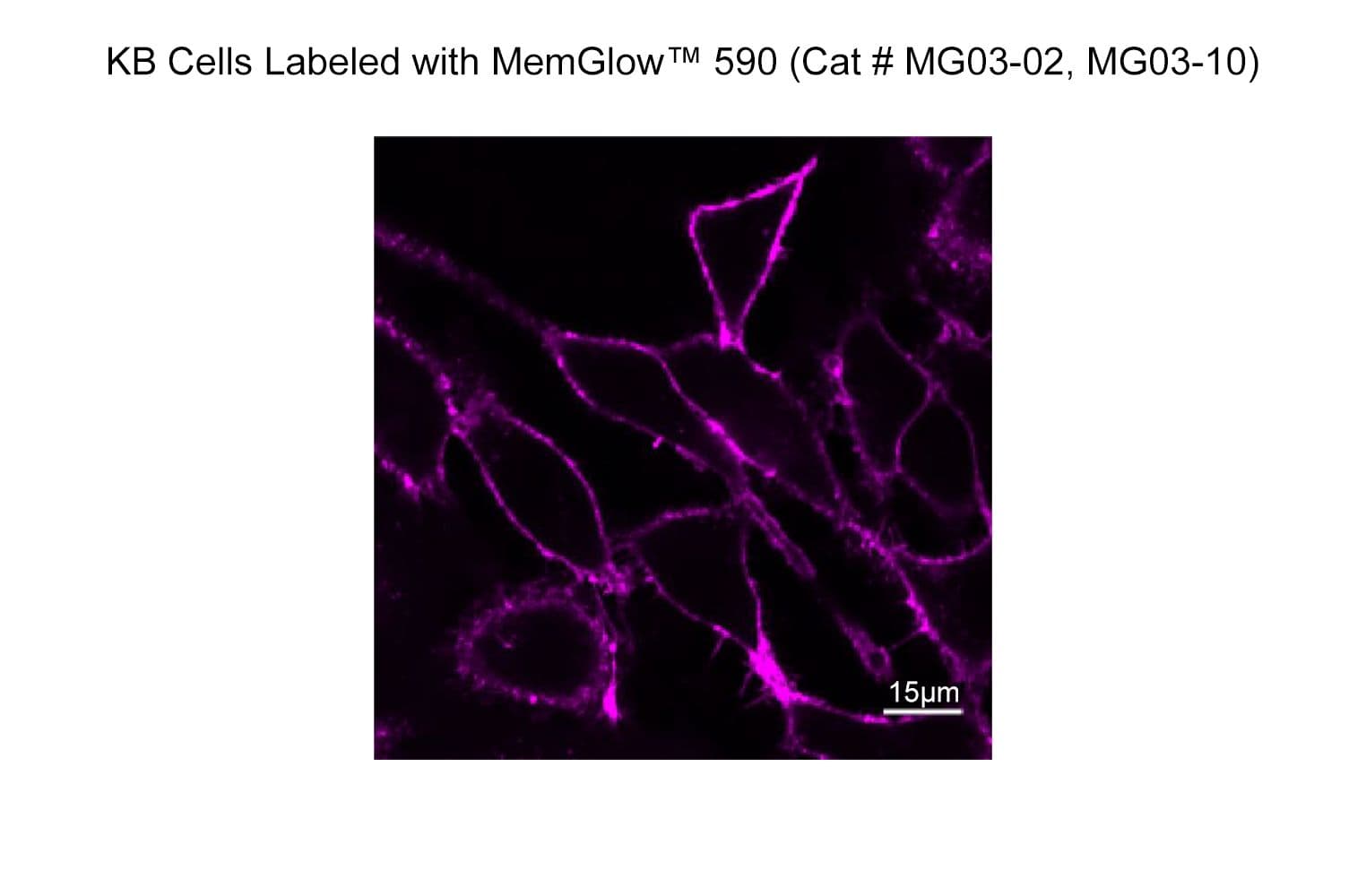

MemGlow™590 is a bright & nontoxic live cell membrane probe. It exhibits ideal microscopy characteristics, including high specificity, low background, and simple application
Key features
The biological activity of MG03 is assessed by the ability of the probe to efficiently label plasma membranes in live KB and RAW 264.7 cell cultures.