Protocol
+3
Loading...
Cat. #PHDG1
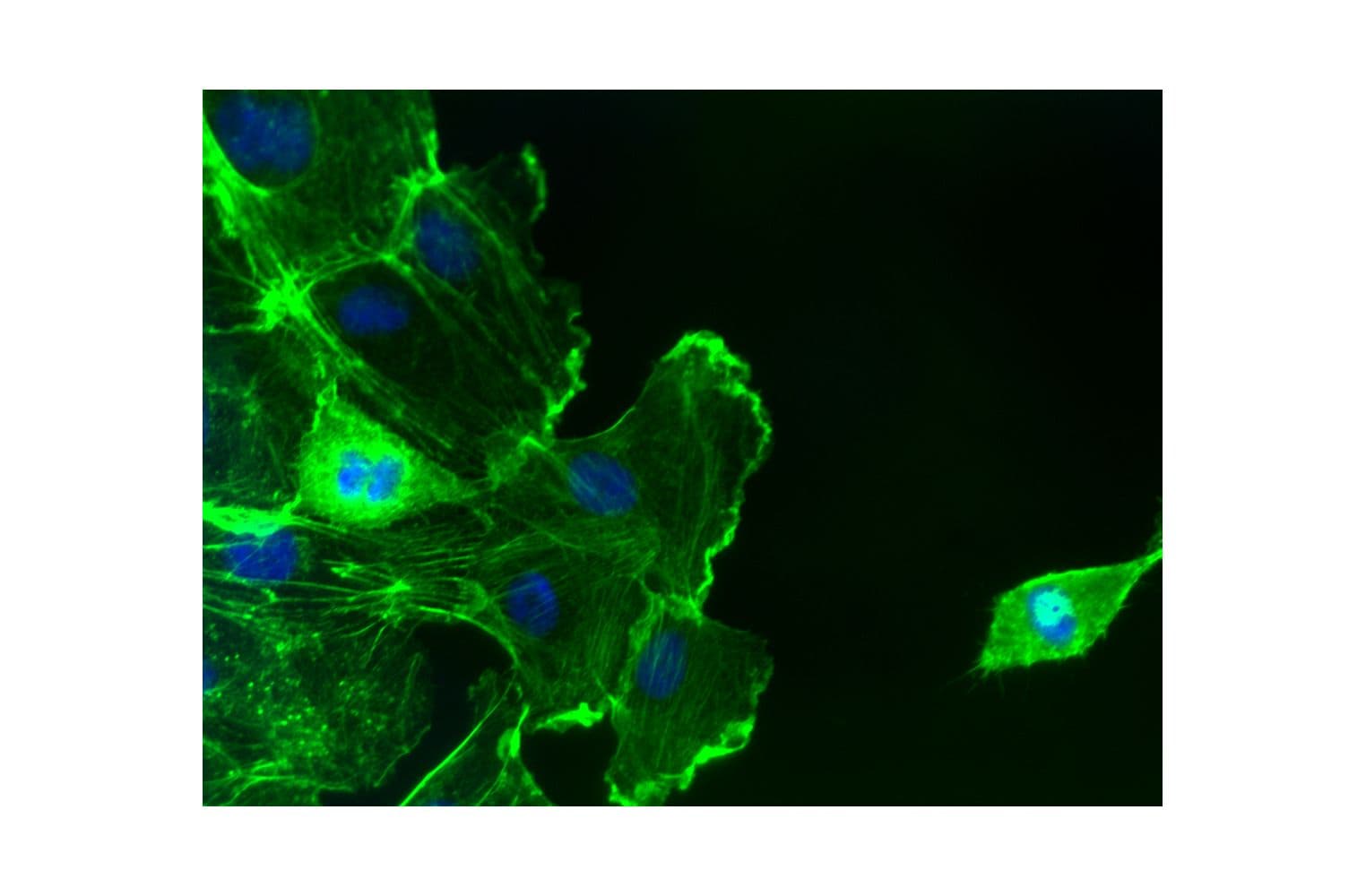
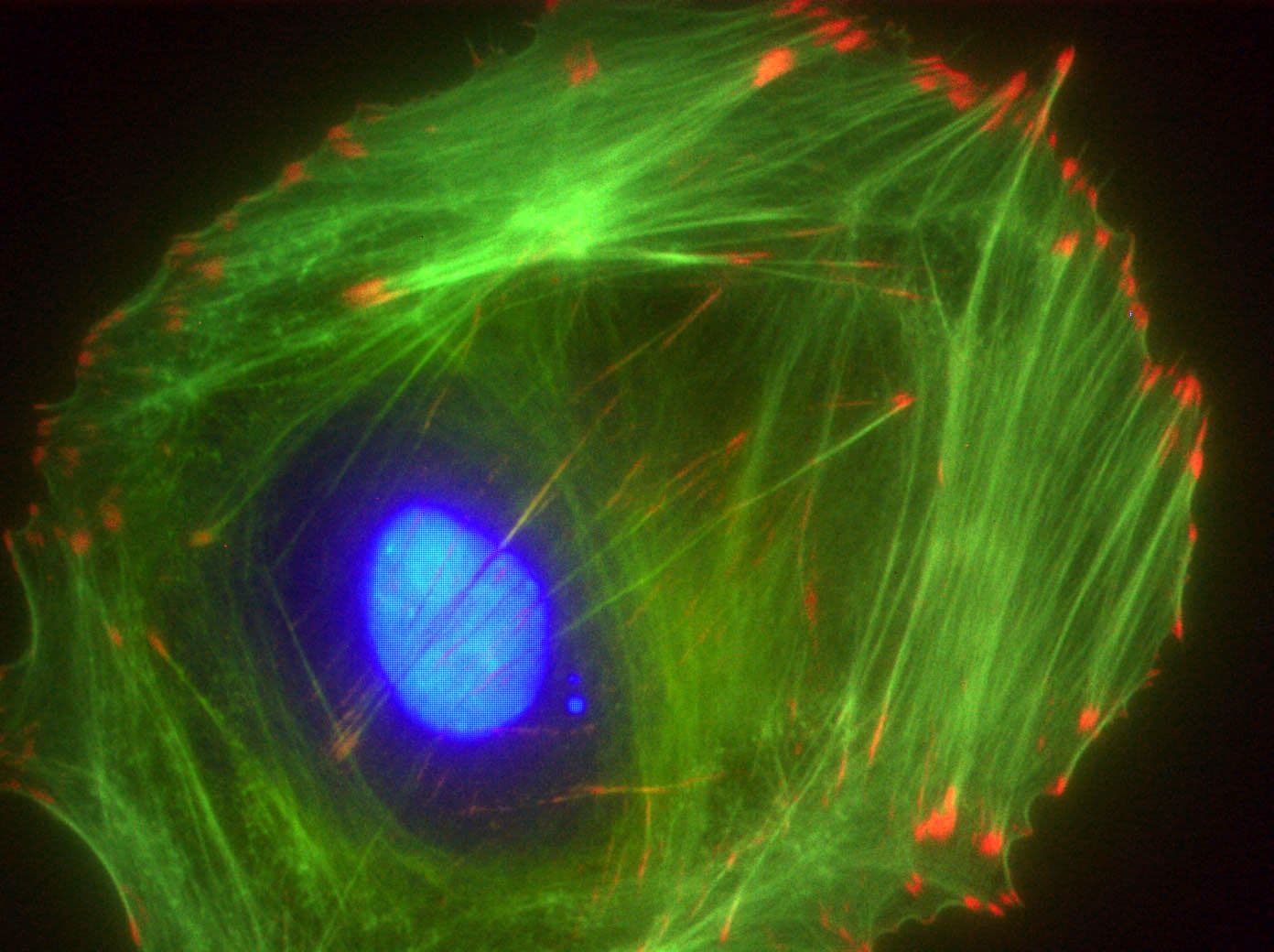
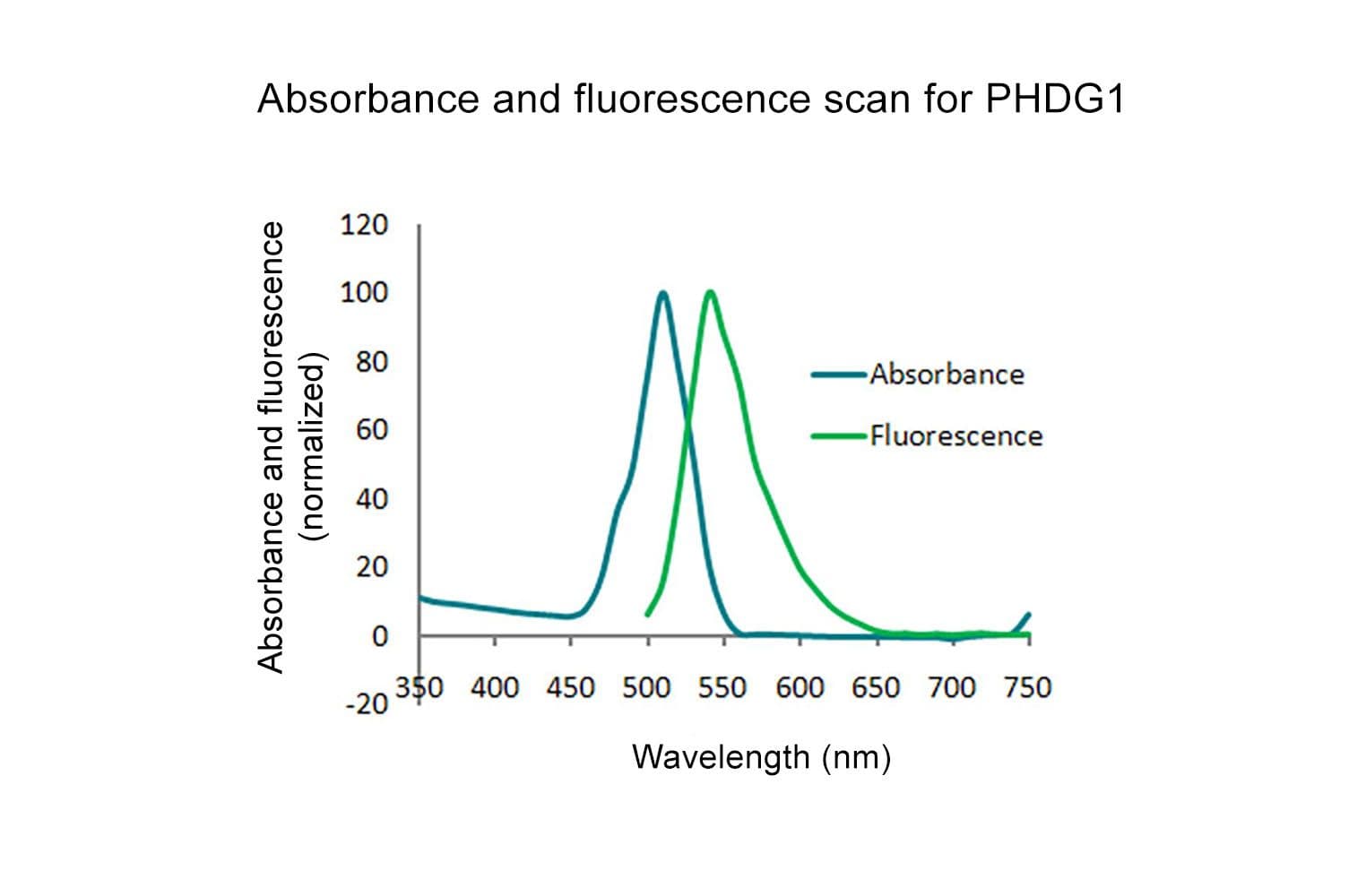

Acti-stain™488 fluorescent phalloidin is a high-affinity probe for F-actin, enabling precise visualization of the actin cytoskeleton in fixed cells and tissues. Conjugated to bright, photostable fluorophores, it delivers sharp, low-background staining ideal for fluorescence microscopy and quantitative imaging applications.
Key characteristics
≥90% by HPLC
Biological activity of Acti-stain™488 is demonstrated by the ability of phalloidin to specifically stain actin filaments in 3T3 cells.