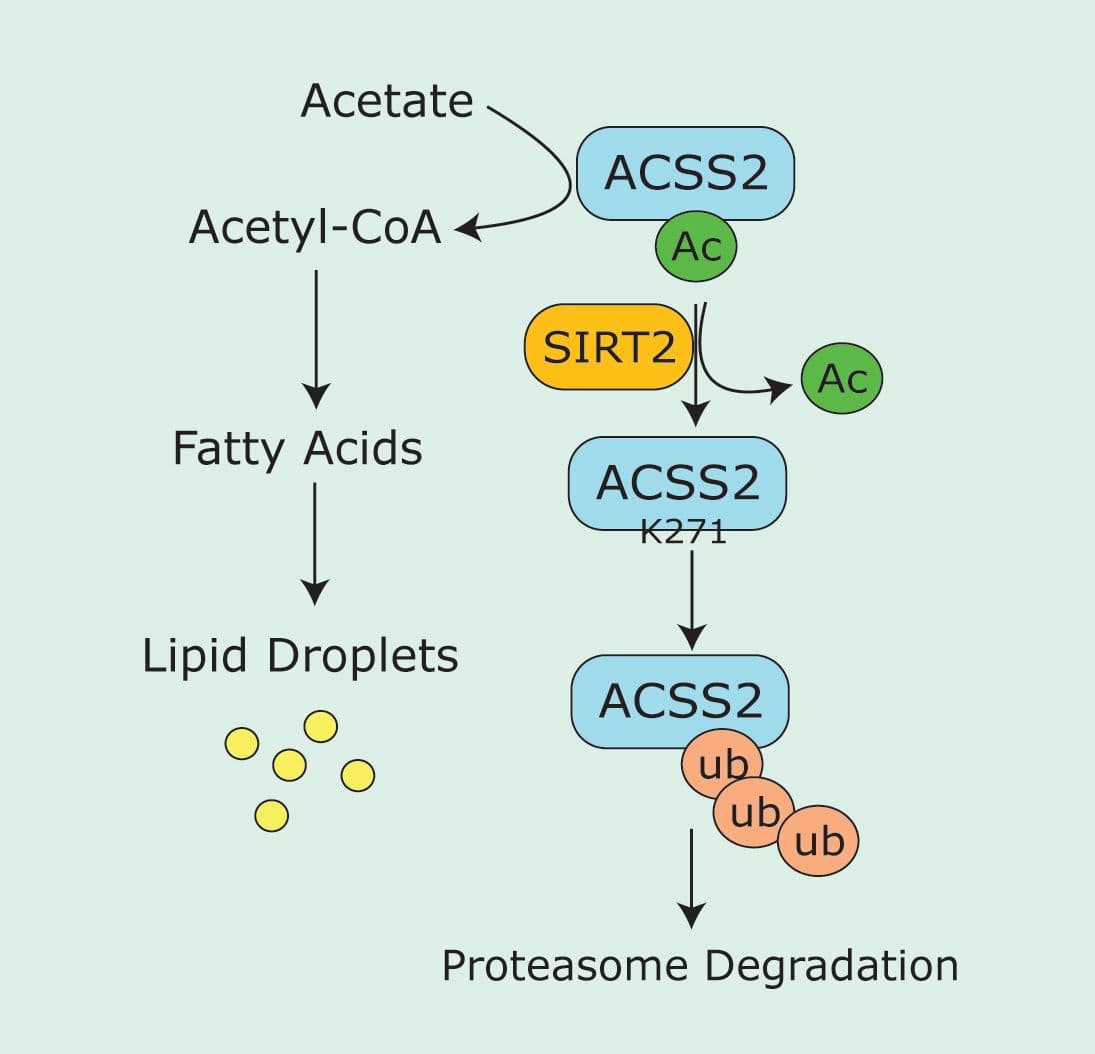
Diseases like diabetes and cancer may be impacted by cellular metabolic processes such as de novo lipogenesis (DNL), which is the process of converting excess proteins, carbohydrates, and alcohol into fatty acids. Understanding the cellular mechanisms that impact DNL is important for health and disease, and significant interest has focused on acetyl-Coenzyme A (CoA), as it is the fundamental building block for fatty acid synthesis. Recently, Karim et al. investigated mechanisms that control acetyl-CoA synthetase 2 (ACSS2), a known regulator of CoA production, during periods of nutrient stress, and identified multiple post-translational modifications (PTMs) as important events. The group chose to examine how the deacetylase, Sirtuin 2 (SIRT2), affected ACSS2 because another deacetylase, SIRT1, was previously shown to deacetylate ACSS2 and enhance its activity. Interestingly, using a heterologous cell line, the group showed that endogenous levels of ACSS2 acetylation decreased in response to amino acid deprivation (nutrient exhaustion), and this deacetylation event could be suppressed via shRNA knockdown of SIRT2. Surprisingly, under nutrient stress conditions, the knockdown of SIRT2 also produced an overall increase in ACSS2 protein levels and was reproduced in multiple cell lines. Further investigation, using a multitude of assays, determined that deacetylation of ACSS2 correlated with increased degradation, which they ultimately attributed to enhanced ubiquitination of the protein. Through mass spectrometry and mutagenesis studies, they determined that ACSS2 is both acetylated and ubiquitinated at K271, and controlling which PTM is present on the protein can dictate ACSS2 levels and activity under nutrient deprivation. This was a surprising finding as it reveals that SIRT1 and SIRT2 may have opposing effects on ACSS2, and highlights the versatility of PTMs like acetylation to regulate protein function. Finally, it was shown that in an adipocyte model, modifying ACSS2 at K271 does impact lipogenesis. Cytoskeleton Inc.’s Acetyl-Lysine Affinity Beads (Cat. # AAC04-Beads) were a critical tool used to examine the endogenous levels of ACSS2 acetylation in response to amino acid depletion. This study highlights the importance of PTM events to fine-tune critical signaling processes that control DNL, which may be important targets for diseases like cancer and obesity.
Link to Citation:
Product Used in Citation:


